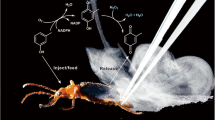
Abstract
Mandelonitrile benzoate, a minor defense component produced by polydesmoid millipedes, is produced in large amounts together with hydrogen cyanide following shake-disturbances administered to individuals of Nedyopus tambanus tambanus, Parafontaria tonominea, Epanerchodus sp., and Epanerchodus fulvus. These species commonly produce mandelonitrile and benzoyl cyanide (the oxidized product after discharge). The newly generated mandelonitrile benzoate was identified as a product of post secretion Schotten-Baumann reaction under basic conditions of bled bodily fluids (pH ca. 9.0), and was not an enzymatic reaction product. The reaction occurred in vitro even under less basic conditions [1M Tris-HCl buffer (pH 8.0)], and could be defined as a new mechanism of hydrogen cyanide release occurring in roughly half of polydesmoid millipedes. Species possessing no benzoyl cyanide, such as Oxidus gracilis and Cryptocorypha sp., could also produce mandelonitrile benzoate under conditions in which benzoyl cyanide was exogenously provided.






Similar content being viewed by others
References
Conner, W. E., Jones, T. H., Eisner, T., and Meinwald, J. 1977. Benzoyl cyanide in the defensive secretion of polydesmoid millipedes. Experientia 33:206–207.
Duffey, S. S., and Towers, G. H. N. 1978. On the biochemical basis of hydrocyanic acid production in the millipede Harpaphe haydeniana (Xystodesmidae: Polydesmida). Can. J. Zool. 56:7–16.
Duffey, S. S., Underhill, E. W., and Towers, G. H. N. 1974. Intermediates in the biosynthesis of hydrogen cyanide and benzaldehyde by a polydesmid millipede Harpaphe haydeniana. Comp. Biochem. Physiol., Part B: Biochem. Mol. Biol. 47(B):753–766.
Duffey, S. S., Blum, M. S., Fales, H. M., Evans, S. L., Roncadori, R. W., Tiemann, D. L., and Nakagawa, Y. 1977. Benzoyl cyanide and mandelonitrile benzoate in the defensive secretions of millipedes. J. Chem. Ecol. 3:101–113.
Eisner, T., Alsop, D., Hicks, K., and Meinwald, J. 1978. Defensive secretions of millipedes, pp. 41–72, in S. Bettini (ed.). Handbuch der Experimentellen Pharmakologie 48 (Arthropod Venoms). Springer-Verlag, Berlin.
Kuerti, L., and Czako, B. 2005. Strategic Applications of Named Reactions in Organic Synthesis, Background and Detailed Mechanism. Elsevier Inc. Burlington (Translated version into Japanese, by TOMIOKA, K., Kagaku-dojin, 2006).
Kuwahara, Y. 1999. Chemical defenses of millipedes, pp. 291–298, in T. Hidaka, Y. Matsumoto, K. Honda, H. Honda, and S. Tatsuki (eds.). Environmental Entomology, Behavior, Physiology and Chemical Ecology. The University of Tokyo Press, Tokyo, Japan (written in Japanese).
Kuwahara, Y., Suzuki, H., Matsumoto, K., and Wada, Y. 1983. Pheromone study on acarid mites XI. Function of mite body as geometrical isomerization and reduction of citral (the alarm pheromone). Appl. Entomol. Zool. 18:30–39.
Kuwahara, Y., Omura, H., and Tanabe, T. 2002. 2-Nitroethenylbenzene as natural products in millipede defense secretions. Naturwissenschaften 89:308–310.
Kuwahara, Y., Mori, N., Sakuma, M., and Tanabe, T. 2003. (1Z)- and (1E)-2-Nitroethenylbenzenes, and 2-nitroethylbenzene as natural products in defense secretions of a millipede Thelodesmus armatus Miyosi (Polydesmida: Pyrgodesmidae). Jpn. J. Environ. Entomol. Zool. 14:149–155.
Kuwahara, Y., Shimizu, N., Sakamoto, F., and Tanabe, T. 2008. Mandelonitrile as the defense secretion, and phenylacetonitrile as its biosynthetic precursor in Nedyopus tambanus tambanus (Paradoxomatidae, Polydesmida). Jpn. J. Environ. Entomol. Zool. 19:79–84.
Mori, N., Kuwahara, K., Yoshida, T., and Nishida, R. 1994. Identification of benzaldehyde, phenol and mandelonitrile from Epanerchodes japonicus Carl (Polidesmida: Polydesmidae) as possible defense substances. Appl. Entomol. Zool. 29:517–522.
Mori, N., Kuwahara, Y., Yoshida, T., and Nishida, R. 1995. Mandelonitrile: major defense cyanogen from Parafontaria laminata armigera Verhoeff (Xystodesmidae: Polydesmida). Appl. Entomol. Zool. 30:197–202.
Noge, K., Kato, M., Mori, N., Kataoka, M., Tanaka, C., Yamasue, Y., Nishida, R., and Kuwahara, Y. 2008. Geraniol dehydrogenase, the key enzyme in biosynthesis of the alarm pheromone, from the astigmatid mite Carpoglyphus lactis (Acari: Carpoglyphidae). FEBS J. 275:2807–2817.
Noguchi, S., Mori, N., Higa, Y., and Kuwahara, Y. 1997a. Identification of mandelonitrile as a major secretory compound from Chamberlinius hualienensis Wang (Polydesmida: Paradoxosomatidae). Jpn. J. Environ. Entomol. Zool. 8:208–214.
Noguchi, S., Mori, N., Higa, Y., and Kuwahara, Y. 1997b. Identification of Nedyopus patrioticus patrioticus (Attems, 1898) (Polydesmida: Paradoxosomatidae) secretions as possible defense substances. Appl. Entomol. Zool. 32:447–452.
Ômura, H., Kuwahara, Y., and Tanabe, T. 2002a. 1-Octen-3-ol together with geosmin: new secretion compounds from a polydesmid millipede, Niponia nodulosa. J. Chem. Ecol. 28:2601–2612.
Ômura, H., Kuwahara, Y., and Tanabe, T. 2002b. Species-specific chemical compositions of defense secretions from Parafontaria tonominea Attems and Riukiaria semicircularis semicircularis Takakuwa (Polydesmida: Xystodesmidae). Appl. Entomol. Zool. 37:73–78.
Taira, J., Nakamura, K., and Higa, Y. 2003. Identification of secretory compounds from the millipede, Oxidus gracilis C. L. Koch (Polydesmida: Paradoxosomatidae) and their variation in different habitats. Appl. Entomol. Zool. 38:401–404.
Wu, X., Burden, D. W., and Attygalle, A. B. 2007. Hydroquinones from defensive secretion of a giant pacific millipede, Acladocricus setigericus (Diplopoda: Spirobolida). Chemoecology 17:131–138.
Zagrobeiny, M., Bak, S., and Moller, B. L. 2008. Cyanogenesis in plants and arthropods. Phytochemistry 67:1457–1468.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kuwahara, Y., Shimizu, N. & Tanabe, T. Release of Hydrogen Cyanide via a Post-secretion Schotten-Baumann Reaction in Defensive Fluids of Polydesmoid Millipedes. J Chem Ecol 37, 232–238 (2011). https://doi.org/10.1007/s10886-011-9920-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10886-011-9920-9