Abstract
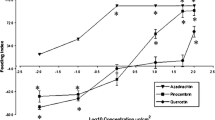
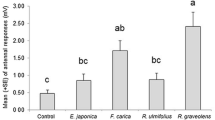
We examined the effects of the flavonoids pinocembrin and quercetin on the feeding behavior, survival, and development of the Cucurbitaceae pest Epilachna paenulata (Coleoptera: Coccinellidae). In no-choice experiments, 48 hr-consumption of Cucurbita maxima Duch. leaves treated with pinocembrin at 1, 5, and 50 μg/cm2 was less than one third of that for leaves treated with 0.1 μg/cm2 of pinocembrin or untreated leaves. Larvae stopped feeding after 9 days of high doses of pinocembrin (5 and 50 μg/cm2), and larval weight and survival were negatively affected by pinocembrin at 1–50 μg/cm2. Delayed mortality in comparison to food-deprived larvae suggests that the mechanism of action for pinocembrin is chronic intoxication, rather than simple starvation from antifeedant effects. In contrast, leaf consumption and larval weight were not significantly affected by quercetin (at 0.1, 1, 5, and 50 μg/cm2) while mortality rates were only slightly increased. The response of E. paenulata larvae in a choice-test to combinations of pinocembrin at antifeedant doses (5 and 50 μg/cm2) and quercetin at phagostimulant doses (0.01 and 0.1 μg/cm2) indicated that the feeding deterrent activity of the former completely overshadowed the stimulant activity of the latter. These results demonstrate the different responses of one insect species to two widely distributed plant flavonoids. Pinocembrin strongly affected survival of E. paenulata while quercetin had only a weak effect without major consequences on the insect life-cycle.




Similar content being viewed by others
References
Abou-Zaid, M. M., Beninger, C. W., Arnason, J. T., and Nozzolillo, C. 1993. The effect of one flavone, two catechins and four flavonols on mortality and growth of the European corn borer (Ostrinia nubilalis Hubner). Biochem. Syst. Ecol. 21: 415–420.
Adelmann, J., Passos, M., Breyer, D. H., Rocha Dos Santos, M. H., Lenz, C., Ferreira Leite, N., Lanças, F. M., and Fontana, J. D. 2007. Exotic flora dependence of an unusual Brazilian propolis: The pinocembrin biomarker by capillary techniques. J. Pharm. Biomed. Anal. 43:174–178.
Bernays, E. A., and Chapman, R. F. 2000. A neurophysiological study of sensitivity to a feeding deterrent in two sister species of Heliothis with different diet breadths. J. Insect. Physiol. 46:905–912.
Bernays, E. A., and Chapman, R. F. 2001. Taste cell responses in the polyphagous arctiid, Grammia geneura: towards a general pattern for caterpillars. J. Insect. Physiol. 47:1029–1043.
Bick, I. R. C., Brown, R. B., and Hillis, W. E. 1972. Three flavanones from leaves of Eucalyptus sieberi. Aust. J. Chem. 25:449–451.
Camarano, S., González, A., and Rossini, C. 2009. Biparental endowment of endogenous defensive alkaloids in Epilachna paenulata. J. Chem. Ecol. 35:1–7.
Carpinella, C., Ferrayoli, C., Valladares, G., Defagó, M., and Palacios, S. M. 2002. Potent limonoid insect antifeedant from Melia azedarach. Biosci. Biotechnol. Biochem. 66:1731–1736.
Carpinella, M. C., Defagó, M. T., Valladares, G., and Palacios S. M. 2003. Antifeedant and insecticide properties of a limonoid from Melia azedarach (Meliaceae) with potential use for pest management. J. Agric. Food Chem. 51:369–374.
Chapman, R. F. 2003. Contact chemoreception in feeding by phytophagous insects. Annu. Rev. Entomol. 48: 455–484.
Defagó, M. T., Valladares, G., Palacios, S. M., and Carpinella, M. C. 2009. Melia azedarach extracts: A potential tool for insect pest management, pp 17–33, in J. N. GOVIL and V. K. SINGH (eds.). Recent Progress in Medicinal Plants. Studium Press LLC, Houston.
Diaz Napal, G., Carpinella, M. C., and Palacios, S. M. 2009. Antifeedant activity of ethanolic extract from Flourensia oolepis and isolation of pinocembrin as its active principle. Bioresour. Technol. 100:3669–3673.
Dillon, M. O., and Mabry, T. J. 1977. Flavonoid aglycones from Flourensia. Phytochemistry, 16:1318–1319.
Green, P. W. C., Stevenson, P. C., Simmonds, M. S. J., and Sharma, H. C. 2003. Phenolic compounds on the pod-surface of pigeonpea, Cajanus cajan, mediate feeding behavior of Helicoverpa armigera larvae. J. Chem. Ecol. 29: 811–821.
Harborne, J. B., and Williams, Ch. A. 2000. Advances in flavonoid research since 1992. Phytochemistry 55:481–504.
Harnly, J. M., Doherty, R. F., Beecher, G. R., Holden, J. M., Haytowitz, D. B., Bhagwat, S., and Gebhardt, S. 2006. Flavonoid content of US fruits, vegetables, and nuts. J. Agric. Food. Chem. 54:9966–9977.
Harwood, M., Danielewska-Nikiel, B., Borzelleca, J. F., Flamm, G. W., Williams, G. M., and Lines, T. C. 2007. A critical review of the data related to the safety of quercetin and lack of evidence of in vivo toxicity, including lack of genotoxic/carcinogenic properties. Food. Chem. Toxicol. 45:2179–2205.
Lattanzio, V., Arpaia, S., Cardinali, A., Di Venere, D., and Linsalata, V. 2000. Role of endogenous flavonoids in resistance mechanism of Vigna to aphids. J. Agric. Food Chem. 48:5316–5320.
Levy, E. C., Ishaaya, I., Gurevitz, E., Cooper, R., and Lavie, D. 1974. Isolation and identification of host compounds eliciting attraction and bite stimuli in the fruit tree bark beetle, Scolytus mediterraneus. J. Agric. Food Chem. 22:376–379.
Lin, L. Z., Mukhopadhyay, S., Robbins, R. J., and Harnly, J. M. 2007 Identification and quantification of flavonoids of Mexican oregano (Lippia graveolens) by LC-DAD-ESI/MS analysis. J. Food Compos. Anal. 20:361–369.
Maggi, M. E., Mangeaud, A., Carpinella, M. C., Ferrayoli, C. G., Valladares, G. R., and Palacios, S. M. 2005. Laboratory evaluation of Artemisia annua L. extract and artemisinin activity against Epilachna paenulata and Spodoptera eridania. J. Chem. Ecol. 31:1527–1536.
Mazoir, N., Benharref, A., Bailén, M., Reina, M., and González Coloma, A. 2008. Bioactive triterpene derivatives from latex of two Euphorbia species. Phytochemistry 69:1328–1338.
Morimoto, M., Kumeda, S., and Komai, K. 2000. Insect antifeedant flavonoids from Gnaphalium affine D. Don. J. Agric. Food Chem. 48:1888–1891.
Neacsu, M., Eklund, P. C., Sjöholm, R. E., Pietarinen, S. P., Ahotupa, M. O., Holmbom, B. R., and Willför, S. M. 2007. Antioxidant flavonoids from knotwood of Jack pine and European aspen. Holz. Roh. Werkst. 65:1–6.
Palacios, S. M., Maggi, M. E., Bazán, C. M., Carpinella, M. C., Turco, M., Muñoz, A., Alonso, R. A., Nuñez, C., Cantero, J. J., Defagó, M. T., Ferrayoli, C. G., and Valladares, G. R. 2007. Screening of some Argentinean medicinal plants for pesticide activity. Fitoterapia 78:580–584.
Schoonhoven, L. M., and Van Loon, J. J. A. 2002. An inventory of taste in caterpillars: each species its own key. Acta Tech. Acad. Sci. Hung. 48:215–263.
Simmonds, M. S. J. 2001. Importance of flavonoids in insect-plant interactions: feeding and oviposition. Phytochemistry 56:245–252.
Simmonds, M. S. J. 2003. Flavonoid-insect interactions: recent advances in our knowledge. Phytochemistry 64:21–30.
Stuart, R. J., and Polavarapu, S. 1998. Oviposition preferences of the polyphagous moth Choristoneura parallela (Lepidoptera: Tortricidae): effects of plant species, leaf size, and experimental design. Environ. Entomol. 27:102–109.
Tewtrakul, S., Subhadhirasakul, S., Puripattanavong, J., and Panphadung, T. 2003. HIV-1 protease inhibitory substances from the rhizomes of Boesenbergia pandurata Hollt. Songklanakarin. J. Sci. Technol. 25:503–508.
Treutter, D. 2006. Significance of flavonoids in plant resistance: A review. Environ. Chem. Lett. 4:147–157.
Van Loon, J. J. A. 1990. Chemoreception of phenolic acids and flavonoids in larvae of two species of Pieris. J. Comp. Physiol. A 166:889–899.
Acknowledgments
This work was supported by a Grant from FONCyT- PICT 2005 and PICTO-CRUP 2005. G.D.N. gratefully acknowledges receipt of a fellowship from CONICET. We thank Joss Heywood for revising the English language.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Diaz Napal, G.N., Defagó, M.T., Valladares, G.R. et al. Response of Epilachna paenulata to Two Flavonoids, Pinocembrin and Quercetin, in a Comparative Study. J Chem Ecol 36, 898–904 (2010). https://doi.org/10.1007/s10886-010-9823-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10886-010-9823-1