Abstract
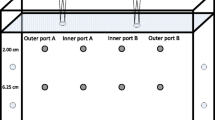
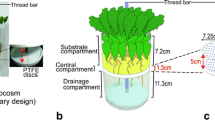
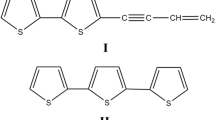
The difficulties of monitoring allelochemical concentrations in soil and their dynamics over time have been a major barrier to testing hypotheses of allelopathic effects. Here, we evaluate three diffusive sampling strategies that employ polydimethylsiloxane (PDMS) sorbents to map the spatial distribution and temporal dynamics of root-exuded thiophenes from the African marigold, Tagetes erecta. Solid phase root zone extraction (SPRE) probes constructed by inserting stainless steel wire into PDMS tubing were used to monitor thiophene concentrations at various depths beneath marigolds growing in PVC pipes. PDMS sheets were used to map the distribution of thiophenes beneath marigolds grown in thin glass boxes. Concentrations of the two major marigold thiophenes measured by these two methods were extremely variable in both space and time. Dissection and analysis of roots indicated that distribution of thiophenes in marigold roots also was quite variable. A third approach used 1 m lengths of PDMS microtubing placed in marigold soil for repeated sampling of soil without disturbance of the roots. The two ends of the tubing remained out of the soil so that solvent could be washed through the tubing to collect samples for HPLC analysis. Unlike the other two methods, initial experiments with this approach show more uniformity of response, and suggest that soil concentrations of marigold thiophenes are affected greatly even by minimal disturbance of the soil. Silicone tube microextraction gave a linear response for α-terthienyl when maintained in soils spiked with 0–10 ppm of this thiophene. This method, which is experimentally simple and uses inexpensive materials, should be broadly applicable to the measurement of non-polar root exudates, and thus provides a means to test hypotheses about the role of root exudates in plant-plant and other interactions.





Similar content being viewed by others
References
Arthur, C., and Pawliszyn, J. 1990. Solid-phase microextraction with thermal desorption using fused-silica optical fibers. Anal. Chem. 62:2145–2148.
Bakker, J., Gommers, F., Nieuwenhuis, I., and Wynberg, H. 1979. Photoactivation of the nematicidal compound α-terthienyl from roots of marigolds (Tagetes spp.). J. Biol. Chem. 254:1841–1844.
Baltussen, E., Sandra, P., David, F., and Cramers, C. 1999. Stir bar sorptive extraction (SBSE), a novel extraction technique for aqueous samples: theory and principles. J. Microcolumn Sep. 11:737-747.
Barto, K. and Cipollini, D. 2009. Half-lives and field soil concentrations of Alliaria petiolata secondary metabolites. Chemosphere 76:71–75.
Bohlmann, F., Burkhardt, T., and Zdero, C. 1973. Naturally occurring acetylenes. Academic, London.
Bruheim, I., Liu, X., and Pawliszyn, J., 2003. Thin-film microextraction. Anal. Chem. 75:1002–1010.
Campbell, G., Lambert, J., Arnason, T., and Towers, G. 1982. Allelopathic properties of α-terthienyl and phenylheptatriyne, naturally occurring compounds from species of Asteraceae. J. Chem. Ecol. 8:961–972.
Cornelissen, G., Pettersen, A., Broman, D., Mayer, P., and Breedveld, G. 2008. Field testing of equilibrium passive samplers to determine freely dissolved native polycyclic aromatic hydrocarbon concentrations. Environ. Toxicol. Chem. 27:499–508.
Croes, A., Van Den Berg, A., Bosveld, M., Breteler, H., and Wullems, G. 1989. Thiophene accumulation in relation to morphology in roots of Tagetes patula. Planta 179:43–50.
Croes, A., Jacobs, J., Arroo, R. and Wullems, G. 1994. Thiophene biosynthesis in Tagetes roots: Molecular vs. metabolic regulation. Plant Cell Tiss. Org. 38:159–165.
Downum, K., and Towers, G. 1983. Analysis of thiophenes in the Tageteae (Asteraceae) by HPLC. J. Nat. Prod. 46:98–103.
Jacobs, J., Croes, A. Engelberts, A. and Wullems G. 1994. Thiophene biosynthesis and distribution in young developing plants of Tagetes patula and T. erecta. J. Exp. Bot. 45:1459–1466.
Kwon, J. -H., Wuethrich, T., Mayer, P. and ESCHER., B. 2009. Development of a dynamic delivery method for in vitro bioassays. Chemosphere 76:83–90.
Martin, J., and Weidenhamer, J. 1995. Potassium deficiency increases thiophene production in Tagetes erecta. Curr. Top. Plant Physiol. 15:277–279.
Mayer, P., and Holmstrup, M. 2008. Passive dosing of soil invertebrates with polycyclic aromatic hydrocarbons: Limited chemical activity explains toxicity cutoff. Environ. Sci. Technol. 42:7516–7521.
Mayer, P., Vaes, W., Wijnker, F., LeGierse, K., Kraaij, R., Tolls, J., and Hermens, J. 2000. Sensing dissolved sediment porewater concentrations of persistent and bioaccumulative pollutants using disposable solid-phase microextraction fibers. Environ. Sci. Technol. 34:5177–5183.
Mayer, P., Tolls, J., Hermens, J., and Mackay, D. 2003. Equilibrium sampling devices. Environ. Sci. Technol. 37:184A–191A.
Mayer, P., Toräng, L., Glæsner, N., and Jönsson, J. 2009. Silicone membrane equilibrator: Measuring chemical activity of nonpolar chemicals with poly(dimethylsiloxane) microtubes immersed directly in tissue and lipids. Anal. Chem. 81:1536–1542.
Ooki, A., and Yokouchi, Y. 2008. Development of a silicone membrane equilibrator for measuring partial pressures of volatile organic compounds in natural water. Environ. Sci. Technol. 42:5706–5711.
Reichenberg, F. and Mayer, P. 2006. Two complementary sides of bioavailability: Accessibility and chemical activity of organic contaminants in sediments and soils. Environ. Toxicol. Chem. 25:1239–1245.
Tang, C., Wat, C., and Towers, G. 1987. Thiophenes and benzofurans in the undisturbed rhizosphere of Tagetes patula L. Plant and Soil 98:93–97.
Weidenhamer, J. 1996. Distinguishing resource competition and chemical interference: Overcoming the methodological impasse. Agron. J. 88:866–875.
Weidenhamer, J. 2005. Biomimetic measurement of allelochemical dynamics in the rhizosphere. J. Chem. Ecol. 31:221–236.
Weidenhamer, J., Boes, P., and Wilcox, D. 2009. Solid-phase root zone extraction (SPRE): A new methodology for measurement of allelochemical dynamics in soil. Plant and Soil 322:177–186. doi: 10.1007/s11104-009-9905-4.
Williamson, G. B., and Weidenhamer, J. D. 1990. Bacterial degradation of juglone: Evidence against allelopathy? J. Chem. Ecol. 16: 1739–1742.
Zhang, H., Davison, W., Miller, S., and Tych, W. 1995. In situ high resolution measurements of fluxes of Ni, Cu, Fe, and Mn and concentrations of Zn and Cd in porewaters by DGT. Geochim. Cosmochim. Acta 59:4181–4192.
Zhang, H., Zhao, F. -J., Sun, B., Davison, W., and McGrath, S. 2001. A new method to measure effective soil solution concentration predicts copper availability to plants. Environ. Sci. Technol. 35: 2602–2607.
Acknowledgments
This research was supported by a grant from the National Science Foundation (DEB 0515826) and from the European Commission (MODELPROBE, no. 213161). Philip Boes assisted with isolation and characterization of BBT. Udo Blum and two anonymous reviewers provided helpful comments on the manuscript. Kelly Harrison assisted with greenhouse cultivation of plants.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mohney, B.K., Matz, T., LaMoreaux, J. et al. In Situ Silicone Tube Microextraction: A New Method for Undisturbed Sampling of Root-exuded Thiophenes from Marigold (Tagetes erecta L.) in Soil. J Chem Ecol 35, 1279–1287 (2009). https://doi.org/10.1007/s10886-009-9711-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10886-009-9711-8