Abstract
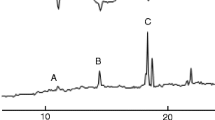
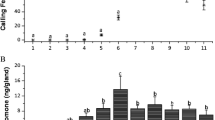
The southern cowpea weevil, Callosobruchus chinensis (Coleoptera: Bruchidae), is a major pest of stored legumes in warm temperate and tropical climates. The female sex attractant pheromone was extracted from filter-paper shelters taken from containers that housed virgin females. The extracts were purified by various chromatographic techniques, and the biologically active components in the fractions were screened by gas chromatographic–electroantennographic detection analysis with male antennae. Two compounds that elicited electrophysiological responses were isolated, and gas chromatography–mass spectrometry, nuclear magnetic resonance, and micro-chemical analyses suggested that the active compounds were homofarnesals, (2Z,6E)- and (2E,6E)-7-ethyl-3,11-dimethyl-2,6,10-dodecatrienals. Males of C. chinensis were significantly attracted to filter paper discs loaded with the synthetic compounds at 0.01–0.1 ng compared to solvent control in a Y-tube olfactometer assay. These pheromone components represent unique chemical structures within the genus Callosobruchus.






Similar content being viewed by others
References
Attygalle, A. B. 1998. Microchemical techniques, pp. 207–294, in J. G. Millar, and K. F. Haynes (eds.). Methods in Chemical Ecology: Vol. 1, Chemical Methods. Kluwer, Norwell, MA.
Beroza, M., and Bierl, B. A. 1966. Apparatus for ozonolysis of microgram to milligram amounts of compound. Anal. Chem. 38:1976–1977.
Beroza, M., and Bierl, B. A. 1967. Rapid determination of olefin position in organic compounds in microgram range by ozonolysis and gas chromatography. Anal. Chem. 39:1131–1135.
Borowiec, L. 1987. The genera of seed-beetles (Coleoptera: Bruchidae). Pol. Pis. Entomol. 57:3–207.
Burkholder, W. E., and Ma, M. 1985. Pheromones for monitoring and control of stored-product insects. Annu. Rev. Entomol. 30:257–272.
Cardé, R. T., and Bell, W. J. (eds.). 1995. Chemical Ecology of Insects. Chapman & Hall, New York.
Cork, A., Hall, D. R., Blaney, W. M., and Simmonds, M. S. J. 1991. Identification of a component of the female sex pheromone of Callosobruchus analis (Coleoptera: Bruchidae). Tetrahedron Lett. 32:129–132.
Fujii, K., Gatehouse, A. M. R., Johnson, C. D., Mitchell, R., and Yoshida, T. (eds.). 1990. Bruchids and Legumes: Economics, Ecology and Coevolution. Kluwer, Dordrecht.
Hariri, G. 1981. Distribution and importance of bruchid attacks on different species of pulses consumed in the Near East, pp. 215–221, in V. Labeyrie (ed.). The Ecology of Bruchids Attacking Legumes (Pulses). Junk, The Hague.
Heaps, J. W. (ed.). 2006. Insect Management for Food Storage and Processing, 2nd Edition. AACC International, St. Paul, MN.
Honda, H., and Ohsawa, K. 1990. Chemical ecology for stored product insects. J. Pesticide Sci. 15:263–270 (in Japanese with English abstract).
Ignacimuthu, S., Wäckers, F. L., and Dorn, S. 2000. The role of chemical cues in host finding and acceptance by Callosobruchus chinensis. Entomol. Exp. Appl. 96:213–219.
Imura, O. 1990. Life histories of stored-products insects, pp. 257–270, in K. Fujii, A. M. R. Gatehouse, C. D. Johnson, R. Mitchell, and T. Yoshida (eds.). Bruchids and Legumes: Economics, Ecology and Coevolution. Kluwer, Dordrecht.
Labeyrie, V. (ed.). 1981. The Ecology of Bruchids Attacking Legumes (Pulses). Junk, The Hague.
Leal, W. S., Kuwahara, Y., Suzuki, T., and Kurosa, K. 1989. The alarm pheromone of the mite Suidasia medanensis Oudemans, 1924 (Acariformes, Suidasiidae). Agric. Biol. Chem. 10:2703–2709.
Matsumoto, H., Tebayashi, S., Kuwahara, Y., Matsuyama, S., Suzuki, T., and Fujii, K. 1994. Identification of taxifolin present in the azuki bean as an oviposition stimulant of the azuki bean weevil. J. Pesticide Sci. 19:181–186.
Mbata, G. N., Shu, S., and amaswamy, S. B. 2000. Sex pheromones of Callosobruchus subinnotatus and C. maculatus (Coleoptera: Bruchidae): congeneric responses and role of air movement. Bull. Entomol. Res. 90:147–154.
Minks, A. K., and Cardé, R. T. 1997. Use of pheromones in direct control, pp. 333–459, in R. T. Cardé, and A. K. Minks (eds.). Insect Pheromone Research, New Directions. Chapman & Hall, New York.
Morgan, E. D. 1999. Terpenes, pp. 85–103, in E. D. Morgan (ed.). Biosynthesis in InsectsThe Royal Society of Chemistry. Milton Road, Cambridge.
Mori, K. 1972. Synthesis of compounds with juvenile hormone activity—XII. A stereoselective synthesis of 6-ethyl-10-methyldodeca-5-trans, 9-cis-dien-2-one, a key intermediate in the synthesis of C18-Cecropia juvenile hormone. Tetrahedron 28:3747–3756.
Mori, K. 2005. Synthetic examination of incorrectly proposed structures of biomolecules. Chem. Rec. 5:1–16.
Mori, K., Mitsui, T., Fukami, J., and Ohtaki, T. 1971. Synthesis of compounds with juvenile hormone activity part VII. A convenient non-stereoselective synthesis of the C18-Cecropia juvenile hormone and its analogues; effect of the terminal alkyl substituents on biological activity. Agri. Biol. Chem. 35:1116–1127.
Nojima, S., Kiemle, D. J., Webster, F. X., and Roelofs, W. L. 2004. Submicro scale NMR sample preparation for volatile chemicals. J. Chem. Ecol. 30:2153–2161.
Nojima, S., Linn, C. Jr., Morris, B., Zhang, A., and Roelofs, W. L. 2003. Identification of host fruit volatiles from hawthorn (Crataegus spp.) attractive to hawthorn-origin Rhagoletis pomonella flies. J. Chem. Ecol. 29:321–336.
Nojima, S., Shimomura, K., Honda, H., Yamamoto, I., and Ohsawa, K. 2007. Contact sex pheromone components of the cowpea weevil, Callosobruchus maculatus. J. Chem. Ecol. 33:923–933.
Oshima, K., Honda, H., and Yamamoto, I. 1973. Isolation of an oviposition marker from azuki bean weevil, Callosobruchus chinensis (L.). Agri. Biol. Chem. 37:2679–2680.
Phillips, T. W. 1997. Semiochemicals of stored-product insects: research and applications. J. Stored Prod. Res. 33:17–30.
Phillips, T. W., Phillips, J. K., Webster, F. X., Tang, R., and Burkholder, W. E. 1996. Identification of sex pheromones from cowpea weevil, Callosobruchus maculatus, and related studies with C. analis (Coleoptera: Bruchidae). J. Chem. Ecol. 22:2233–2249.
Rees, D. P. 1995. Coleoptera, pp. 1–40, in B. Subramanyam, and D. W. Hagstrum (eds.). Integrated Management of Insects in Stored Products. Marcel Dekker, Madison Avenue, New York.
Sakai, A., Honda, H., Oshima, K., and Yamamoto, I. 1986. Oviposition marking pheromone of two bean weevils, Callosobruchus chinensis and Callosobruchus maculatus. J. Pesticide Sci. 11:163–168.
Shinoda, K., and Yoshida, T. 1985. Ecology of azuki weevil in azuki field. I. Emergent prevalence and damage of azuki in azuki field on autumn. Jpn. Appl. Entmol. Zool. 29:14–20 (in Japanese with English summary).
Shinoda, K., and Yoshida, T. 1990. Life history of the azuki bean weevil, Callosobruchus chinensis (Coleoptera: Bruchidae), in the field, pp. 149–159, in K. Fujii, A. M. R. Gatehouse, C. D. Johnson, R. Mitchell, and T. Yoshida (eds.). Bruchids and Legumes: Economics, Ecology and Coevolution. Kluwer, Dordrecht.
Shinoda, K., Yoshida, T., and Igarashi, H. 1992. Population ecology of the azuki bean beetle, Callosobruchus chinensis (L.), (Coleoptera: Bruchidae) on two wild leguminous hosts, Vigna angularis var. nipponensis and Dunbaria villosa. Appl. Entmol. Zool. 27:311–318.
Shinoda, K., Yoshida, T., and Okamoto, T. 1991. Two wild leguminous host plants of the azuki bean weevil, Callosobruchus chinensis (L.) (Coleoptera: Bruchidae). Appl. Entmol. Zool. 26:91–98.
Shu, S., Mbata, G. N., Cork, A., and Ramaswamy, S. B. 1998. Female sex pheromone in Callosobruchus subinnotatus (Coleoptera: Bruchidae): Production and male responses. Ann. Entomol. Soc. Am. 91:840–844.
Shu, S., Mbata, G. N., Cork, A., and Ramaswamy, S. B. 1999. Sex pheromone of Callosobruchus subinnotatus. J. Chem. Ecol. 25:2715–2727.
Siegel, S. 1956. The one-sample case, pp. 35–60, in S. Siegel (ed.). Nonparametric Statistics for the Behavioral Science. McGraw-Hill, New York.
Subramanyam, B., and Hagstrum, D. W. (eds.). 2000. Alternatives to Pesticides in Stored-product IPM. Kluwer, Boston, MA.
Tanaka, K., Ohsawa, K., Honda, H., and Yamamoto, I. 1981. Copulation release pheromone, Erectin, from the azuki bean weevil (Callosobruchus chinensis L.). J. Pesticide Sci. 6:75–82.
Tanaka, K., Ohsawa, K., Honda, H., and Yamamoto, I. 1982. Synthesis of Eectin, a copulation release pheromone of the azuki bean weevil, Callosobruchus chinensis L. J. Pesticide Sci. 7:535–537.
Tebayashi, S., Matsuyama, S., Suzuki, T., Kuwahara, Y., Nemoto, T., and Fujii, K. 1995. Quercimertrin: the third oviposition stimulant of the azuki bean weevil from the host azuki bean. J. Pesticide Sci. 20:299–305.
Tuda, M., Chou, L.-Y., Niyomdham, C., Buranapanichpan, S., and Tateishi, Y. 2005. Ecological factors associated with pest status in Callosobruchus (Coleoptera: Bruchidae): high host specificity of non-pests to Cajaninae (Fabaceae). J. Stored Pro. Res. 41:31–45.
Tuda, M., Rönn, J., Buranapanichpan, S., Wasano, N., and Arnqvist, G. 2006. Evolutionary diversification of the bean beetle genus Callosobruchus (Coleoptera: Bruchidae): traits associated with stored-product pest status. Mol. Ecol. 15:3541–3551.
Ueno, T., Kuwahara, Y., Fujii, K., Taper, M. L., Toquenaga, Y., and Suzuki, T. 1990. D-Catechin: an oviposition stimulant of azuki bean weevil Callosobruchus chinensis in the host azuki bean. J. Pesticide Sci. 15:573–578.
Watanabe, N. 1990. Diversity in life cycle patterns of bruchids occurring in Japan (Coleoptera: Bruchidae), pp. 141–147, in K. Fujii, A. M. R. Gatehouse, C. D. Johnson, R. Mitchell, and T. Yoshida (eds.). Bruchids and Legumes: Economics, Ecology and Coevolution. Kluwer, Dordrecht.
White, N. D. G., and Leesch, J. G. 1996. Chemical Control, pp. 287–330, in B. Subramanyam, and D. W. Hagstrum (eds.). Integrated Management of Insects in Stored Products. Marcel Dekker, Madison Avenue, New York.
Yoneda, M., Kaneda, M., and Akiyama, H. 1990. Plant quarantine and fumigation of imported grain legumes in Japan, pp. 115–120, in K. Fujii, A. M. R. Gatehouse, C. D. Johnson, R. Mitchell, and T. Yoshida (eds.). Bruchids and Legumes: Economics, Ecology and Coevolution. Kluwer, Dordrecht.
Zagatti, P., Kunesch, G., amiandrasoa, F., Malosse, C., Hall, D. R., Lester, R., and Nesbitt, B. F. 1987. Sex pheromones of rice moth, Corcyra cephalonica Staomtpm I. Identification of male pheromone. J. Chem. Ecol. 13:1561–1573.
Acknowledgments
The authors are grateful to Dr. Kaoru Tanaka for his early efforts in this project in our laboratory and valuable comments on the study. We thank Drs. Izuru Yamamoto and Hiroshi Honda for helpful advice. We also thank Drs. Goro Yabuta and Arata Yajima for help with the chemical synthesis. We also express our gratitude to Dr. Coby Schal for reviewing the manuscript. Finally, we are also thankful to two anonymous reviewers and an associate editor (Dr. Steven J. Seybold) for their help on the previous draft of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Shimomura, K., Nojima, S., Yajima, S. et al. Homofarnesals: Female Sex Attractant Pheromone Components of the Southern Cowpea Weevil, Callosobruchus chinensis . J Chem Ecol 34, 467–477 (2008). https://doi.org/10.1007/s10886-008-9451-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10886-008-9451-1