Abstract
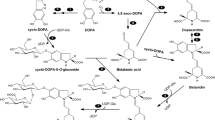
l-3,4-Dihydroxyphenylalanine (l-dopa) is one of the few allelochemicals in which the phytotoxic action mechanism has been studied. Excess exogenous l-dopa suppresses root elongation in some plant species, and the inhibitory action is species-selective. The main factor of phytotoxicity of l-dopa is considered to be oxidative damage by reactive oxygen species (ROS) and/or free radical species (FRS). This study was performed to elucidate the mechanism of species-selective phytotoxicity. The involvement of ROS/FRS and polyphenol oxidase (PPO) in species-selective phytotoxicity was examined with barnyardgrass (Echinochloa crus-galli L.) and lettuce (Lactuca sativa L.), tolerant and susceptible species, respectively. Lipid peroxidation and melanin accumulation correlated with growth inhibition by L-dopa. Antioxidants, ascorbic acid and α-tocopherol, decreased lipid peroxidation and melanin accumulation and rescued lettuce root from growth inhibition. The oxidation of L-dopa by PPO was much greater in lettuce than in barnyardgrass. From these results, the phytotoxicity of L-dopa is considered due to the oxidative damage caused by ROS/FRS generated from the melanin synthesis pathway. PPO activity might be involved in the mechanism of species-selective phytotoxicity between barnyardgrass and lettuce.





Similar content being viewed by others
References
Albrecht, C., and Kohlenbach, H. W. 1990. l-dopa content, per-oxidase activity, and response H2O2 of Vicia fava L. and V. narbonensis L. in situ and in vitro. Protoplasma 154:144–150.
Basma, A. N., Morris, E. J., Nicklas, W. J., and Geller, H. M. 1995. l-Dopa cytotoxicity to PC12 cells in culture is via its autoxidation. J. Neurochem 64:85–832.
Beckman, J. S., Minor, R. I., JR, White, C. W., Repine, J. E., Rosen, G. M., and Freeman, B. A. 1988. Superoxide dismutase and catalase conjugated to polyethylene glycol increases endothelial enzyme activity and oxidant resistance. J. Biol. Chem. 263:825–832.
Buckley, B. J., Tanswell, A. K., and Freeman, B. A. 1987. Liposome-mediated augmentation of catalase in alveolar type II cells protects against H2O2 injury. J. Appl. Physiol. 63:359–367.
Fujii, Y. 1994. Screening of allelopathic candidates by new specific discrimination, and assessment methods for allelopathy, and the identification of l-dopa as the allelopathic substance from the most promising velvetbean (Mucuna pruriens). Bull. Natl. Inst. Agro.-Environ. Sci. 10:115–218.
Graham, D. G. 1978. Oxidative pathways for catecholamines in the genesis of neuromelanin and cytotoxic quinines. Mol. Phalmacol. 14:633–643.
Hachinohe, M., and Matsumoto, H. 2005. Involvement of reactive oxygen species generated from melanin synthesis pathway in phytotoxicity of l-dopa. J. Chem. Ecol. 31:237–246.
Hachinohe, M., Sunohara, Y., and Matsumoto, H. 2004. Absorption, translocation and metabolism of l-dopa in barnyardgrass and lettuce: their involvement in species-selective phytotoxic action. Plant Growth Regul. 43:237–243.
Haque, E., Asunuma, M., Higashi, Y., Miyazaki, I., Tanaka, K., and Ogawa, N. 2003. Apotosis-inducting neurotoxicity of dopamine and its metabolites via reactive quinone generation in neuroblastma cells. Biochem. Biophys. Acta 1619:39–52.
Kruk, L., Lichszteld, K., Bounias, M., Kladna, A., and Krubera-Nowakowska L. 1999. Formation of active oxygen species during autoxidation of dopa. Chemosphere 39:443–453.
Lai, C. T., and Yu, P. H. 1997. Dopamine- and l-beta-3,4-dihydroxyphenylalnine hydrochloride (l-dopa)-induced cytotoxicity towards catecholaminergic neuroblastma SH-SY5Y cells. Effects of oxidative stress and autoxidative factors. Biochem. Pharmacol. 53:363–372.
Nakajima, N., Hiradate, S., and Fujii, Y. 1999. Characteristics of growth inhibitory effect of L-3, 4-dihydroxyphenylalanine (l-dopa) on cucumber seedling (in Japanese). J. Weed. Sci. Technol. 44:132–138.
Parson, P. G. 1985. Modification of dopa toxicity in human tumour cells. Biochem. Pharmacol. 34:1801–1807.
Pattison, I. D., Dean, T. R., and Davies. 2002. Oxidation of DA, proteins and lipids by dopa, protein-bound DOPA, and related catechol(amine)s. Toxicology 177:23–37.
Sherman, T. D., Vaughn, K. C., and Duke, S. O. 1991. A limited survey of the phylogenetic distribution of polyphenol oxidase. Phytochemistry 30:2499–2506.
Vaughn, K. C., Lax, A. R., and Duke, S. O. 1988. Polyphenol oxidase: The chloroplast oxidase with no established function. Physiol. Plantarum 72:659–665.
Velikova, V., Yordanov, I., and Edreva, A. 2000. Oxidative stress and some antioxidant systems in acid rain-treated bean plants. Protective role of exogenous polyamines. Plant Sci.151:59–66.
Wakamatsu, K., and Ito, S. 2000. Advanced chemical methods in melanin determination. Pigment Cell Res. 15:174–183.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hachinohe, M., Matsumoto, H. Mechanism of Selective Phytotoxicity of l-3,4-Dihydroxyphenylalanine (l-Dopa) in Barnyardglass and Lettuce. J Chem Ecol 33, 1919–1926 (2007). https://doi.org/10.1007/s10886-007-9359-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10886-007-9359-1