Abstract
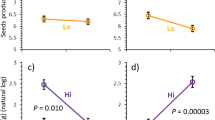
In a greenhouse study, we evaluated the effect of the light environment and support availability on the induction of tropane alkaloids (TAs) after leaf damage in the twining vine Convolvulus arvensis. We also tested whether leaf damage modifies the phenotypic responses of the plant to shade and physical support. We found a consistent pattern of induction of TAs after leaf damage in each environmental condition. The induction of TAs was differentially affected by combinations of support and light treatments. In the sun, prostrate and climbing vines exhibited similar induced responses. In the shade, prostrate vines showed greater induced responses. Thus, vines showed the greatest chemical induction when damage occurred in a resource-poor environment (shade), and there was no cue (support) of future increase in resource uptake. Damaged plants showed reduced plasticity to shading in leaf shape and internodes and petiole length in comparison with control, undamaged plants. Herbivory and/or induced responses to herbivory may limit adaptive plant responses to the environment. Therefore, the negative consequences of herbivory on plant fitness might be magnified in a context of changing environments.


Similar content being viewed by others
References
Agrawal, A. A. 2005. Future directions in the study of induced plant responses to herbivory. Entomol. Exp. Appl. 115:97–105.
Cipollini, D. F. 2004. Stretching the limits of plasticity: Can a plant defend against both competitors and herbivores? Ecology 85:28–37.
Cipollini, D. F. and Bergelson, J. 2001. Plant density and nutrient availability constrain the constitutive and wound-induced expression of trypsin inhibitors in Brassica napus L. J. Chem. Ecol. 27:593–610.
Collantes, H. G., Gianoli, E., and Niemeyer, H. M. 1997. Effect of defoliation on the patterns of allocation of a hydroxamic acid in rye (Secale cereale L.). Environ. Exp. Bot. 38:231–235.
Costa-Arbulú, C., Gianoli, E., Gonzales, W. L., and Niemeyer, H. M. 2001. Feeding by the aphid Sipha flava produces a reddish spot on leaves of Sorghum halepense: An induced defence? J. Chem. Ecol. 27:273–283.
Darwin, C. R. 1875. The Movements and Habits of Climbing Plants. John Murray, London.
Den Dubbelden, K. C. and Oosterbeek, B. 1995. The availability of external support affects allocation patterns and morphology of herbaceous climbing plants. Funct. Ecol. 9:628–634.
Fordyce, J. A. 2001. The lethal plant defense paradox remains: Inducible host-plant aristolochic acids and the growth and defense of the pipevine swallowtail. Entomol. Exp. Appl. 100:339–346.
Gentry, A. H. 1991. The distribution and evolution of climbing plants, pp. 3–49, in F. E. Putz, and H. A. Mooney (eds.) The Biology of Vines. Cambridge University Press, Cambridge.
Gianoli, E. 2001. Lack of differential plasticity to shading of internodes and petioles with growth habit in Convolvulus arvensis (Convolvulaceae). Int. J. Plant Sci. 162:1247–1252.
Gianoli, E. 2002. Maternal environmental effects on the phenotypic responses of the twining vine Ipomoea purpurea to support availability. Oikos 99:324–330.
Gianoli, E. 2003. Phenotypic responses of the twining vine Ipomoea purpurea (Convolvulaceae) to physical support availability in sun and shade. Plant Ecol. 165:21–26.
Gianoli, E. 2004. Evolution of a climbing habit promotes diversification in flowering plants. Proc. R. Soc. Lond. B 271:2011–2015.
Gianoli, E. and Molina-Montenegro, M. A. 2005. Leaf damage induces twining in a climbing plant. New Phytol. 167:385–390.
Gianoli, E. and Niemeyer, H. M. 1996. Environmental effects on the induction of wheat chemical defences by aphid infestation. Oecologia 107:549–552.
Gianoli, E. and Niemeyer, H. M. 1997. Lack of costs of herbivory-induced defenses in a wild wheat: Integration of physiological and ecological approaches. Oikos 80:269–275.
González, A. V. and Gianoli, E. 2004. Morphological plasticity in response to shading in three Convolvulus species of different ecological breadth. Acta Oecol. 26:185–190.
Heil, M. 2002. Ecological costs of induced resistance. Curr. Opin. Plant Biol. 5:345–350.
Heil, M. 2004. Induced indirect defences benefit Lima bean (Phaseolus lunatus) in nature. J. Ecol. 92:527–536.
Heil, M. and Baldwin, I. T. 2002. Costs of induced resistance: Emerging experimental support for a slippery concept. Trends Plant Sci. 7:61–67.
Izaguirre, M. M., Scopel, A. L., Baldwin, I. T., and Ballaré, C. L. 2003. Convergent responses to stress. Solar ultraviolet-B radiation and Manduca sexta herbivory elicit overlapping transcriptional responses in field-grown plants of Nicotiana longiflora. Plant Physiol. 132:1755–1767.
Izaguirre, M. M., Mazza, C. A., Biondini, M., Baldwin, I. T., and Ballaré, C. L. 2006. Remote sensing of future competitors: Impacts on plant defenses. Proc. Natl. Acad. Sci. USA 103:7170–7174.
Karban, R. and Baldwin, I. T. 1997. Induced Responses to Herbivory. University of Chicago Press, Chicago.
Khan, M. B. and Harborne, J. B. 1990. Induced alkaloid defence in Atropa acuminata in response to mechanical and herbivore leaf damage. Chemoecology 1:77–80.
Krug, E. and Proksch, P. 1993. Influence of dietary alkaloids on survival and growth of Spodoptera littoralis. Biochem. Syst. Ecol. 21:749–756.
Kurashige, N. S. and Agrawal, A. A. 2005. Phenotypic plasticity to shading and herbivory in Chenopodium album. Am. J. Bot. 92:21–26.
Lee, D. W. 1988. Simulating forest shade to study the developmental ecology of tropical plants: Juvenile growth in three vines in India. J. Trop. Ecol. 4:281–292.
Marquis, R. J. 1992. The selective impact of herbivores, pp. 301–325, in R. S. Fritz and E. L. Simms (eds.). Plant Resistance to Herbivores and Pathogens. Ecology, Evolution and Genetics. University of Chicago Press, Chicago.
McGuire, R. and Agrawal, A. A. 2005. Trade-offs between the shade-avoidance response and plant resistance to herbivores? Tests with mutant Cucumis sativus. Funct. Ecol. 19:1025–1031.
Peñalosa, J. 1983. Shoot dynamics and adaptive morphology of Ipomoea phillomega (Vell.) House (Convolvulaceae), a tropical rainforest liana. Ann. Bot. 52:737–754.
Putz, F. E. 1984. The natural history of lianas on Barro Colorado Island, Panamá. Ecology 65:1713–1724.
Putz, F. E. and Mooney, H. A. 1991. The Biology of Vines. Cambridge University Press, Cambridge.
Quezada, I. M. and Gianoli, E. 2006. Simulated herbivory limits phenotypic responses to drought in Convolvulus demissus (Convolvulaceae). Pol. J. Ecol. 54:499–503.
Rausher, M. D., Iwao, K., Simms, E. L., Ohsaki, N., and Hall, D. 1993. Induced resistance in Ipomoea purpurea. Ecology 74:20–29.
Ray, T. S. 1992. Foraging behaviour in tropical herbaceous climbers (Araceae). J. Ecol. 80:189–203.
Rosenthal, S. S. and Hostettler, N. 1980. Galeruca rufa (Col., Chrysomelidae) seasonal life history and the effect of its defoliation on its host plant, Convolvulus arvensis (Convolvulaceae). Entomophaga 25:381–388.
Schierenbeck, K. A., Mack, R. N., and Sharitz, R. R. 1994. Effects of herbivory on growth and biomass allocation in native and introduced species of Lonicera. Ecology 75:1661–1672.
Strauss, S. Y. and Agrawal, A. A. 1999. Ecology and evolution of plant tolerance to herbivory. Trends Ecol. Evol. 14:179–185.
Strauss, S. Y., Rudgers, J. A., Lau, J. A., and Irwin, R. E. 2002. Direct and ecological costs of resistance to herbivory. Trends Ecol. Evol. 17:278–285.
Todd, F. G., Stermitz, F. R., Knight, A. P., and Traub-Dargatz, J. 1995. Tropane alkaloids and toxicity of Convolvulus arvensis. Phytochemistry 39:301–303.
Weaver, S. E. and Riley, W. R. 1982. The biology of Canadian weeds. 53. Convolvulus arvensis L. Can. J. Plant Sci. 62:461–472.
Wright, S. J., Calderón, O., Hernández, A., and Paton, S. 2004. Are lianas increasing in importance in tropical forests? A 17-year record from Panama. Ecology 85:484–489.
Zangerl, A. R. 2003. Evolution of induced plant responses to herbivores. Basic Appl. Ecol. 4:91–1003.
Zangerl, A. R. and Bazzaz, F. A. 1992. Theory and pattern in plant defense allocation, pp. 363–391, in R. S. Fritz and E. L. Simms (eds.). Plant Resistance to Herbivores and Pathogens. Ecology, Evolution and Genetics. University of Chicago Press, Chicago.
Acknowledgement
We thank Z. Rozas, S. Meneses, and V. Hernández for assistance during the chemical analysis, and M. González and P. Torres for help in the greenhouse. M. Heil and an anonymous reviewer made helpful suggestions for improvement of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gianoli, E., Molina-Montenegro, M.A. & Becerra, J. Interactive Effects of Leaf Damage, Light Intensity and Support Availability on Chemical Defenses and Morphology of a Twining Vine. J Chem Ecol 33, 95–103 (2007). https://doi.org/10.1007/s10886-006-9215-8
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10886-006-9215-8