Abstract
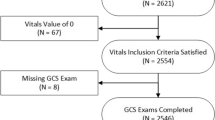
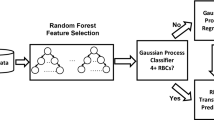
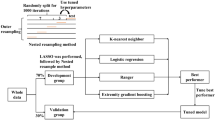
Cardiac aortic surgery is an extremely complicated procedure that often requires large volume blood transfusions during the operation. Currently, it is not possible to accurately estimate the intraoperative blood transfusion volume before surgery. Therefore, in this study, to determine the clinically precise usage of blood for intraoperative blood transfusions during aortic surgery, we established a predictive model based on machine learning algorithms. We performed a retrospective analysis on 4,285 patients who received aortic surgery in Beijing Anzhen Hospital between January 2018 and September 2022. Ultimately, 3,654 patients were included in the study, including 2,557 in the training set and 1,097 in the testing set. By utilizing 13 current mainstream models and a large-scale cardiac aortic surgery dataset, we built a novel machine learning model for accurately predicting intraoperative red blood cell transfusion volume. Based on the transfusion-related risk factors that the model identified, we also established the relevant variables that affected the results. The results revealed that decision tree models were the most suitable for predicting the blood transfusion volume during aortic surgery. In particular, the mean absolute error for the best-performing extremely randomized forest model was 1.17 U, while the R2 value was 0.50. Further exploration into intraoperative blood transfusion during aortic surgery identified erythrocytes, estimated operation duration, body weight, sex, red blood cell count, and D-dimer as the six most significant risk factors. These factors were subsequently analyzed for their influence on intraoperative blood transfusion volume in relevant patients, as well as the protective threshold for prediction. The novel intraoperative blood transfusion prediction model for cardiac aorta surgery in this study effectively assists clinicians in accurately calculating blood transfusion volumes and achieving effective utilization of blood resources. Furthermore, we utilize interpretability technology to reveal the influence of critical risk factors on intraoperative blood transfusion volume, which provides an important reference for physicians to provide timely and effective interventions. It also enables personalized and precise intraoperative blood usage.




Similar content being viewed by others
Data availability
The dataset used and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Rogers MA, Blumberg N, Saint S, et al. Hospital variation in transfusion and Infection after cardiac Surgery: a cohort study. BMC Med. 2009;7:37.
Stover EP, Siegel LC, Parks R, et al. Variability in transfusion practice for coronary artery bypass surgery persists despite national consensus guidelines: a 24-institution study. Institutions of the multicenter study of perioperative ischemia research group. Anesthesiology. 1998;88:327–33.
Snyder-Ramos SA, Mohnle P, Weng YS, et al. The ongoing variability in blood transfusion practices in cardiac Surgery. Transfusion. 2008;48:1284–99.
Michalopoulos A, Tzelepis G, Dafni U, et al. Determinants of hospital mortality after coronary artery bypass grafting. Chest. 1999;115:1598–603.
Koch CG, Li L, Duncan AI, et al. Morbidity and mortality risk associated with red blood cell and blood-component transfusion in isolated coronary artery bypass grafting. Crit Care Med. 2006;34:1608–16.
Murphy GJ, Reeves BC, Rogers CA, et al. Increased mortality, postoperative morbidity, and cost after red blood cell transfusion in patients having cardiac Surgery. Circulation. 2007;116:2544–52.
Marik PE, Corwin HL. Efficacy of red blood cell transfusion in the critically ill: a systematic review of the literature. Crit Care Med. 2008;36:2667–74.
Engoren MC, Habib RH, Zacharias A, et al. Effect of blood transfusion on long-term survival after cardiac operation. Ann ThoracSurg. 2002;74:1180–6.
Leal-Noval SR, Rincon-Ferrari MD, Garcia-Curiel A, et al. Transfusion of blood components and postoperative Infection in patients undergoing cardiac Surgery. Chest. 2001;119:1461–8.
Hemli JM, Ducca EL, Chaplin WF, et al. Transfusion in root replacement for aortic dissection: The STS adult cardiac surgery database analysis. Ann Thorac Surg. 2022;114(6):2149–56.
Feng Y-N. Intelligent prediction of RBC demand in trauma patients using decision tree methods. Mil Med Res. 2021;8(1):1–12.
Jiang Z. An explainable machine learning algorithm for risk factor analysis of in-hospital mortality in sepsis survivors with ICU readmission. Comput Method Progr Biomed. 2021;204:106040.
Levi R, Carli F. Artificial intelligence-based prediction of transfusion in the intensive care unit in patients with gastrointestinal bleeding. BMJ Health Care Inform. 2021;28(1):e100245.
Kang S. Machine Learning Model for the Prediction of Hemorrhage in Intensive Care Units. Healthc Inform Res. 2022;28(4):364–75.
Liu S. Machine learning models to predict red blood cell transfusion in patients undergoing mitral valve surgery. Ann Transl Med. 2021;9(7):530.
Hitinder S. A random forest based risk model for reliable and accurate prediction of receipt of transfusion in patients undergoing percutaneous coronary intervention. PloS one. 2014;9(5):e96385.
Lundberg Scott M. Explainable machine-learning predictions for the prevention of hypoxaemia during surgery. Nat Biomed Eng. 2018;2(10):749–60.
Camilo A, BChir, et al. The Effect of BloodTransfusion on outcomes inAortic. Surg Int J Angiol. 2017;26:135–42.
Funding
The authors have not disclosed any funding.
Author information
Authors and Affiliations
Contributions
CJ and YB contributed to study conception and design, acquisition of data, statistical analysis, interpretation of data, drafting the manuscript, and critical revision of the manuscript for important intellectual content. XY, WL, CY, HJ and ZH contributed to acquisition of data, statistical analysis, interpretation of data, and critical revision of the manuscript for important intellectual content. CY, HJ and ZH contributed to interpretation of data and critical revision of the manuscript for important.
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing interests.
Ethical approval
This study protocol was approved by the Ethics Committee of Beijing Anzhen Hospital, Capital Medical University.
Consent to participate
This study protocol was approved by the Ethics Committee of Beijing Anzhen Hospital, Capital Medical University.
Consent for publication
Not applicable.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Che, J., Yang, B., Xie, Y. et al. A precise blood transfusion evaluation model for aortic surgery: a single-center retrospective study. J Clin Monit Comput (2023). https://doi.org/10.1007/s10877-023-01112-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10877-023-01112-3