Abstract
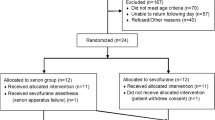
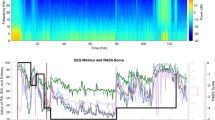
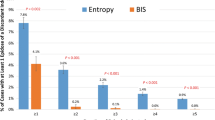
Ketamine may affect the reliability of electroencephalographic (EEG) depth-of-hypnosis indices as it affects power in high-frequency EEG components. The purpose of this study was to compare the effects of ketamine on three commonly-used depth-of-hypnosis indices by extending our EEG simulator to allow replay of previously-recorded EEG. Secondary analysis of previously-collected data from a randomized controlled trial of intravenous anesthesia with ketamine: Group 0.5 [ketamine, 0.5 mg kg−1 bolus followed by a 10 mcg kg−1 min−1 infusion], Group 0.25 [ketamine, 0.25 mg kg−1 bolus, 5 mcg kg−1 min−1 infusion], and Control [no ketamine]. EEG data were replayed to three monitors: NeuroSENSE (WAV), Bispectral Index (BIS), and Entropy (SE). Differences in depth-of-hypnosis indices during the initial 15 min after induction of anesthesia were compared between monitors, and between groups. Monitor agreement was evaluated using Bland–Altman analysis. Available data included 45.6 h of EEG recordings from 27 cases. Ketamine was associated with higher depth-of-hypnosis index values measured at 10 min (BIS, χ2 = 8.01, p = 0.018; SE, χ2 = 11.44, p = 0.003; WAV, χ2 = 9.19, p = 0.010), and a higher proportion of index values > 60 for both ketamine groups compared to the control group. Significant differences between monitors were not observed, except between BIS and SE in the control group. Ketamine did not change agreement between monitors. The ketamine-induced increase in depth-of-hypnosis indices was observed consistently across the three EEG monitoring algorithms evaluated. The observed increase was likely caused by a power increase in the beta and gamma bands. However, there were no lasting differences in depth-of-hypnosis reported between the three compared indices.




Similar content being viewed by others
References
Fahy BG, Chau DF. The technology of processed electroencephalogram monitoring devices for assessment of depth of anesthesia. Anesth Analg. 2018;126:111–7.
Purdon PL, Pierce ET, Mukamel EA, Prerau MJ, Walsh JL, Wong KFK, et al. Electroencephalogram signatures of loss and recovery of consciousness from propofol. Proc Natl Acad Sci. 2013;110:E1142–E1151151.
Purdon PL, Sampson A, Pavone KJ, Brown EN. Clinical electroencephalography for anesthesiologists: part I: background and basic signatures. Anesthesiology. 2015;123:937–60.
Luginbühl M, Wüthrich S, Petersen-Felix S, Zbinden AM, Schnider TW. Different benefit of bispectal index (BIS) in desflurane and propofol anesthesia. Acta Anaesthesiol Scand. 2003;47:165–73.
Gan TJ, Glass PS, Windsor A, Payne F, Rosow C, Sebel P, et al. Bispectral index monitoring allows faster emergence and improved recovery from propofol, alfentanil, and nitrous oxide anesthesia. BIS Utility Study Group Anesthesiol. 1997;87:808–15.
Petersen CL, Görges M, Massey R, Dumont GA, Ansermino JM. A procedural electroencephalogram simulator for evaluation of anesthesia monitors. Anesth Analg. 2016;123:1136–40.
van Heusden K, Cooke E, Brodie S, West N, Görges M, Dumont GA, et al. Effect of ketamine on the NeuroSENSE WAVCNS during propofol anesthesia; a randomized feasibility trial. J Clin Monit Comput. 2020. https://doi.org/10.1007/s10877-020-00511-0.
Blain-Moraes S, Lee U, Ku S, Noh G, Mashour GA. Electroencephalographic effects of ketamine on power, cross-frequency coupling, and connectivity in the alpha bandwidth. Front Syst Neurosci. 2014;8:114.
Marchant N, Sanders R, Sleigh J, Vanhaudenhuyse A, Bruno M-A, Brichant JF, et al. How electroencephalography serves the anesthesiologist. Clin EEG Neurosci. 2014;45:22–32.
Bland JM, Altman DG. Statistical methods for assessing agreement between two methods of clinical measurement. Lancet (London, England). 1986;1:307–10.
Bland JM, Altman DG. Agreement between methods of measurement with multiple observations per individual. J Biopharm Stat. 2007;17:571–82.
Lin LI. A concordance correlation coefficient to evaluate reproducibility. Biometrics. 1989;45:255–68.
Hayashi K, Tsuda N, Sawa T, Hagihira S. Ketamine increases the frequency of electroencephalographic bicoherence peak on the alpha spindle area induced with propofol. Br J Anaesth. 2007;99:389–95.
Maksimow A, Särkelä M, Långsjö JW, Salmi E, Kaisti KK, Yli-Hankala A, et al. Increase in high frequency EEG activity explains the poor performance of EEG spectral entropy monitor during S-ketamine anesthesia. Clin Neurophysiol. 2006;117:1660–8.
Kelley SD. Monitoring consciousness using the bispectral IndexTM (BISTM) during anesthesia: a pocket guide for clinicians [Internet]. [cited 2020 Jan 13]. Available from: https://www.medtronic.com/covidien/en-us/clinical-education/catalog/monitoring-consciousness-using-bispectral-index-during-anesthesia-bis-pocket-guide-clinicians.html
GE Healthcare. Quick guide: entropy monitoring: a valuable tool for guiding delivery of anesthesia [Internet]. [cited 2020 Jan 13]. Available from: https://www.gehealthcare.com/quick-guide/entropy-monitoring-a-valuable-tool-for-guiding-delivery-of-anesthesia
Bibian S, Zikov T. NeuroSENSE monitor with WAVcns cortical quantifier: a deterministic approach to EEG analysis (white paper) [Internet]. 2018 [cited 2020 Mar 11]. Available from: https://www.neurowavesystems.com/pdfs/NeuroSENSE_White_Paper_September_2018.pdf
Yeung JK, Zed PJ. A review of etomidate for rapid sequence intubation in the emergency department. CJEM. 2002;4:194–8.
Eagleman SL, Drover DR. Calculations of consciousness: electroencephalography analyses to determine anesthetic depth. Curr Opin Anaesthesiol. 2018;31:431–8.
Bojak I, Day HC, Liley DTJ. Ketamine, propofol, and the EEG: a neural field analysis of HCN1-mediated interactions. Front Comput Neurosci. 2013;7:1–14.
Acknowledgements
The authors would like to thank the clinical staff at the Eagle Ridge Hospital and the research team who conducted the original study, from which the data for this study was extracted.
Funding
This study was supported by internal funds. NeuroWave Systems Inc. provided loan equipment and sensors for the original study.
Author information
Authors and Affiliations
Contributions
SS contributed to the study design and data analysis. CP contributed to the development of the EEG replay technology. NW contributed to data analysis. JMA contributed to study design. RNM contributed to study design. MG contributed to study design and data analysis. All authors contributed to preparation of this manuscript and read and approved the final version.
Corresponding author
Ethics declarations
Conflicts of interest
JMA has consulted for NeuroWave Systems Inc. JMA, RNM and MG are party to a licensing agreement between the University of British Columbia and NeuroWave Systems Inc. for control technology. JMA and RNM have received funds for research sponsored by NeuroWave Systems Inc. The remaining authors have no interests to declare. This study was supported by internal funds. NeuroWave Systems Inc. provided loan equipment and sensors for the original study.
Ethical approval
The Fraser Health research ethics board approval [FHREB 2016–054] approved the randomized ketamine trial (clinicaltrials.gov NCT02908945), which allowed secondary analysis of the EEG data collected, and the UBC C&W Research Ethics Board (H15-03244) granted permission for de-identified DoH monitor comparisons using EEG data replay.
Informed consent
Written informed consent was obtained from all individual participants in the original study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Schüler, S.S., Petersen, C.L., West, N.C. et al. The effect of ketamine on depth of hypnosis indices during total intravenous anesthesia—a comparative study using a novel electroencephalography case replay system. J Clin Monit Comput 35, 1027–1036 (2021). https://doi.org/10.1007/s10877-020-00565-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10877-020-00565-0