Abstract
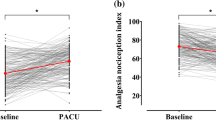
The aim of this study was to evaluate the performance of surgical pleth index (SPI) measured before arousal from general anaesthesia for prediction of immediate postoperative pain and postoperative opioid requirement during postoperative 48 h. After obtaining ethical approval and written informed consent, we enrolled 51 patients undergoing liver resection under isoflurane based general anaesthesia using laryngeal mask airway in this prospective observational study. Data relating to SPI values were recorded every 30 s for the last 3 min of surgery (bispectral index < 60 at all times). Postoperative pain intensity was assessed using a 0–10 numerical rating scale (NRS) every 10 min in the recovery room. The relationships between SPI with postoperative pain score and opioid requirement were analysed. A receiver-operating characteristic curve (ROC) was used to evaluate the performance of SPI to predict NRS ≥ 5. SPI value was significantly associated with the highest pain score in the recovery room (r = 0.63, p < 0.001). An SPI value of 60, which showed the highest sensitivity and specificity, was defined post hoc as the cut-off for moderate-severe pain (NRS ≥ 5). When compared the patients who showed SPI value over 60 or not, there was significant difference in the amount of fentanyl consumption during postoperative 48 h (1093 ± 406 µg vs. 766 ± 369 µg, p = 0.014; SPI ≥ 60 vs. SPI < 60). SPI measured before arousal after inhalation anaesthesia was associated with immediate postoperative pain and postoperative opioid consumption.


Similar content being viewed by others
References
Rawal N. Current issues in postoperative pain management. Eur J Anaesthesiol. 2016;33:160–71. https://doi.org/10.1097/eja.0000000000000366.
Ledowski T, Pascoe E, Ang B, Schmarbeck T, Clarke MW, Fuller C, Kapoor V. Monitoring of intra-operative nociception: skin conductance and surgical stress index versus stress hormone plasma levels. Anaesthesia. 2010;65:1001–6. https://doi.org/10.1111/j.1365-2044.2010.06480.x.
Wang L, Chang Y, Kennedy SA, Hong PJ, Chow N, Couban RJ, McCabe RE, Bieling PJ, Busse JW. Perioperative psychotherapy for persistent post-surgical pain and physical impairment: a meta-analysis of randomised trials. Br J Anaesth. 2018;120:1304–14. https://doi.org/10.1016/j.bja.2017.10.026.
Kehlet H, Jensen TS, Woolf CJ. Persistent postsurgical pain: risk factors and prevention. Lancet. 2006;367:1618–25. https://doi.org/10.1016/s0140-6736(06)68700-x.
Argoff CE. Recent management advances in acute postoperative pain. Pain Pract. 2014;14:477–87. https://doi.org/10.1111/papr.12108.
Grant MC, Sommer PM, He C, Li S, Page AJ, Stone AB, Hobson D, Wick E, Wu CL. Preserved analgesia with reduction in opioids through the use of an acute pain protocol in enhanced recovery after surgery for epen hepatectomy. Reg Anesth Pain Med. 2017;42:451–7. https://doi.org/10.1097/aap.0000000000000615.
Park JH, Kim DH, Yoo SK, Lim HJ, Lee JW, Ahn WS, Lee EK, Choi BM, Noh GJ. The analgesic potency dose of remifentanil to minimize stress response induced by intubation and measurement uncertainty of Surgical Pleth Index. Minerva Anestesiol. 2018;84:546–55. https://doi.org/10.23736/s0375-9393.17.12065-1.
Ledowski T, Burke J, Hruby J. Surgical pleth index: prediction of postoperative pain and influence of arousal. Br J Anaesth. 2016;117:371–4. https://doi.org/10.1093/bja/aew226.
Chooi CS, White AM, Tan SG, Dowling K, Cyna AM. Pain vs comfort scores after caesarean section: a randomized trial. Br J Anaesth. 2013;110:780–7. https://doi.org/10.1093/bja/aes517.
Funcke S, Sauerlaender S, Pinnschmidt HO, Saugel B, Bremer K, Reuter DA, Nitzschke R. Validation of innovative techniques for monitoring nociception during general anesthesia: a clinical study using tetanic and intracutaneous electrical stimulation. Anesthesiology. 2017;127:272–83. https://doi.org/10.1097/aln.0000000000001670.
Gruenewald M, Ilies C, Herz J, Schoenherr T, Fudickar A, Hocker J, Bein B. Influence of nociceptive stimulation on analgesia nociception index (ANI) during propofol-remifentanil anaesthesia. Br J Anaesth. 2013;110:1024–30. https://doi.org/10.1093/bja/aet019.
Shoushtarian M, Sahinovic MM, Absalom AR, Kalmar AF, Vereecke HE, Liley DT, Struys MM. Comparisons of electroencephalographically derived measures of hypnosis and antinociception in response to standardized stimuli during target-controlled propofol-remifentanil anesthesia. Anesth Analg. 2016;122:382–92. https://doi.org/10.1213/ane.0000000000001029.
Ellerkmann RK, Grass A, Hoeft A, Soehle M. The response of the composite variability index to a standardized noxious stimulus during propofol-remifentanil anesthesia. Anesth Analg. 2013;116:580–8. https://doi.org/10.1213/ANE.0b013e31827ced18.
Bonhomme V, Uutela K, Hans G, Maquoi I, Born JD, Brichant JF, Lamy M, Hans P. Comparison of the surgical Pleth Index with haemodynamic variables to assess nociception-anti-nociception balance during general anaesthesia. Br J Anaesth. 2011;106:101–11. https://doi.org/10.1093/bja/aeq291.
Gruenewald M, Herz J, Schoenherr T, Thee C, Steinfath M, Bein B. Measurement of the nociceptive balance by analgesia nociception index and surgical pleth index during sevoflurane-remifentanil anesthesia. Minerva Anestesiol. 2015;81:480–9.
Bapteste L, Szostek AS, Chassard D, Desgranges FP, Bouvet L. Can intraoperative Surgical Pleth Index values be predictive of acute postoperative pain? Anaesth Crit Care Pain Med. 2018. https://doi.org/10.1016/j.accpm.2018.05.004.
Ilies C, Ludwigs J, Gruenewald M, Thee C, Hanf J, Hanss R, Steinfath M, Bein B. The effect of posture and anaesthetic technique on the surgical pleth index. Anaesthesia. 2012;67:508–13. https://doi.org/10.1111/j.1365-2044.2011.07051.x.
Hans P, Verscheure S, Uutela K, Hans G, Bonhomme V. Effect of a fluid challenge on the Surgical Pleth Index during stable propofol-remifentanil anaesthesia. Acta Anaesthesiol Scand. 2012;56:787–96. https://doi.org/10.1111/j.1399-6576.2011.02639.x.
Ledowski T, Ang B, Schmarbeck T, Rhodes J. Monitoring of sympathetic tone to assess postoperative pain: skin conductance vs surgical stress index. Anaesthesia. 2009;64:727–31. https://doi.org/10.1111/j.1365-2044.2008.05834.x.
Thee C, Ilies C, Gruenewald M, Kleinschmidt A, Steinfath M, Bein B. Reliability of the surgical Pleth index for assessment of postoperative pain: a pilot study. Eur J Anaesthesiol. 2015;32:44–8. https://doi.org/10.1097/eja.0000000000000095.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee (Samsung Medical Center Institutional Review Board (SMC 2018-04-129) and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Park, M., Kim, B.J. & Kim, G.S. Prediction of postoperative pain and analgesic requirements using surgical pleth index: a observational study. J Clin Monit Comput 34, 583–587 (2020). https://doi.org/10.1007/s10877-019-00338-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10877-019-00338-4