Abstract
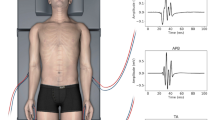
This report presents a method for tracking Motor Evoked Potential (MEP) amplitudes over the course of a case using a moving least squares linear regression (LSMAs). During a case, newly obtained MEP amplitudes are compared to those predicted by a just previous linear regression (least squares moving average or LSMA). When detected by this comparison, a set criterion amplitude loss will then trigger linear regression of ensuing MEP amplitudes on an expanding step function which tracks the persistence of the amplitude loss for the remainder of the case. Three cases are presented. One in which the patient woke up with a newly acquired weakness in the left tibialis anterior and another in which MEP amplitudes were suddenly lost from the right foot, but after intervention, they were restored again. In a third case the patient again woke up with a new post-operative deficit, but MEP trial sampling had been more limited and variable than in the first two cases. When the linear trending method was applied to the affected myotome in the first case, the expanding step function regression was triggered after the moment of MEP loss and remained at a high level until the end of case. In the second case, the expanding step function regression was also triggered in the relevant myotome at the time of the reported MEP change, but diminished by end of case. In the third case the tracking method again successfully triggered a predictive R-Square despite the limited number of pre-event trials. The R-Square value of the expanding step function regression appears to have discriminative capability with regard to new post-op deficit. Given the importance of the intra-operative MEP for monitoring motor functioning and the high degree of variability that can affect it, the development of new quantitative, statistical methods to detect real from apparent MEP change will be necessary.







Similar content being viewed by others
References
Balvin MJ, Song KM, Slimp JC. Effects of anesthetic regimens and other confounding factors affecting the interpretation of motor evoked potentials during pediatric spine surgery. Am J Electroneurodiagn Technol. 2010;50(3):219–44.
Chong CT, Manninen P, Sivanaser V, Subramanyam R, Lu N, Venkatraghavan L. Direct comparison of the effect of desflurane and sevoflurane on intraoperative motor-evoked potentials monitoring. J Neurosurg Anesthesiol. 2014;26(4):306–12.
Fishback AS, Shields CB, Linden RD, Zhang YP, Burke D. The effects of propofol on rat transcranial magnetic motor evoked potentials. Neurosurgery 1995;37(5):969–74.
Kleinbaum DG, Kupper LL, Muller KE. Applied regression analysis and other multivariable methods. 2nd ed. Boston: PWS-Kent Publishing Co.; 1988.
Kobayashi S, Matsuyama Y, Shinomiya K, Kawabata S, Ando M, Kanchiku T, Saito T, Takahashi M, Ito Z, Muramoto A, Fujiwara Y, Kida K, Yamada K, Wada K, Yamamoto N, Satomi K, Tani T. A new alarm point of transcranial electrical stimulation motor evoked potentials for intraoperative spinal cord monitoring: a prospective multicenter study from the Spinal Cord Monitoring Working Group of the Japanese Society for Spine Surgery and Related Research. J Neurosurg Spine. 2014;20(1):102–7.
Lieberman JA, Lyon R, Feiner J, Diab M, Gregory GA. The effect of age on motor evoked potentials in children under propofol/isoflurane anesthesia. Anesth Analg. 2006;103(2):316–21.
Lieberman JA, Feiner J, Lyon R, Rollins MD. Effect of hemorrhage and hypotension on transcranial motor-evoked potentials in swine. Anesthesiology. 2013;119(5):1109–19.
Lieberman JA, Feiner J, Rollins M, Lyon R, Jasiukaitis P. Changes in transcranial motor evoked potentials during hemorrhage are associated with increased serum propofol concentrations. J Clin Monit Comput. 2017. https://doi.org/10.1007/s10877-017-0057-4.
Lotto ML, Banoub M, Schubert A. Effects of anesthetic agents and physiologic changes on intraoperative motor evoked potentials. J Neurosurg Anesthesiol. 2004;16(1):32–42.
Lyon R, Feiner J, Lieberman JA. Progressive suppression of motor evoked potentials during general anesthesia: the phenomenon of “anesthetic fade”. J Neurosurg Anesthesiol. 2005;17(1):13–9.
Pinder JP. Introduction to business analytics using simulation. London: Academic Press; 2017.
Sloan TB, Heyer EJ. Anesthesia for intraoperative neurophysiologic monitoring of the spinal cord. J Clin Neurophysiol. 2002;19(5):430–43.
Tamkus A, Rice KS, Hoffman G. Transcranial motor evoked potential alarm criteria to predict foot drop injury during lumbosacral surgery. Spine 2017;43(4):E227–33.
Thirumala PD, Huang J, Brahme IS, Thiagarajan K, Cheng H, Crammond DJ, Balzer J. Alarm criteria for motor evoked potentials. Neurol India. 2017;65(4):708–15.
Wang AC, Than KD, Etame AB, La Marca F, Park P. Impact of anesthesia on transcranial electric motor evoked potential monitoring during spine surgery: a review of the literature. Neurosurg Focus. 2009;27(4):E7.
Zouridakis G, Papanicolaou AC. A concise guide to intraoperative monitoring. Boca Raton: CRC Press; 2001.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflict of interest with regards to this study. The data reported are retrospective data. Patient treatment was not altered or affected in any way by gathering this data. No patient or medical provider identities are revealed in the submitted article.
Rights and permissions
About this article
Cite this article
Jasiukaitis, P., Lyon, R. A motor evoked potential trending system may discriminate outcome: retrospective application with three cases. J Clin Monit Comput 33, 481–491 (2019). https://doi.org/10.1007/s10877-018-0181-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10877-018-0181-9