Abstract
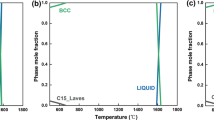
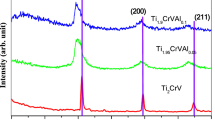
Aiming to unravel the effect of alloying on the hydrogen adsorption mechanism, we perform a comparative analysis of the sequential hydrogen loading over Ti11, binary Ti10V, and Ti6VAl4 ternary alloy clusters. For each cluster, the variation of adsorption energy as a function of hydrogen atom coverage was calculated up to 20 hydrogen molecules. The results show that the adsorption of H2 on pure and alloy clusters occurs in a similar fashion including three phases; (i) three-fold and subsequent two-fold dissociative adsorption, (ii) non-dissociative hydrogen adsorption through the Kubas interaction, (iii) non-dissociative physisorption. Our calculations reveal that Ti11, binary Ti10V, and Ti6VAl4 can adsorb at least 20 H2 molecules with the adsorption energies in the range of chemisorption and physisorption. The gravimetric density of H2 adsorbed on these clusters exceeds the ultimate 7.5 wt% limits, recommended for practical applications. However, the magnitude of adsorption energies for Ti6VAl4 are much smaller than those of pure Ti11 binary Ti10V clusters that favor its operating as hydrogen storage media around ambient temperature and pressure.






Similar content being viewed by others
References
K. Mazloomi and C. Gomes (2012). Renew. Sustain. Energy Rev. 16, 3024.
Y. Li and R. T. Yang (2016). J. Am. Chem. Soc. 28, 8136.
M. Kaur and K. Pal (2019). J. Energy Storage 23, 234.
L. Schlapbach and A. Zuttel (2001). Nature 414, 353.
P. Chen and M. Zhu (2008). Mater. Today 11, 36.
S. S. Kaye and J. R. Long (2005). J. Am. Chem. Soc. 127, 6506.
A. M. Seayad and D. M. Antonelli (2004). Adv. Mater. 16, 765.
Y. Li and R. T. Yang (2006). J. Am. Chem. Soc. 128, 8136.
C. Tang, S. Chen, W. Zhu, J. Kang, X. He, and Z. Zhang (2015). Int. J. Hydrogen Energy 40, 16271.
S. E. Kulkova, S. V. Eremeev, V. E. Egorushkin, J. S. Kim, and S. Y. Oh (2003). Solid State Commun. 126, 405.
H. Zhu, Y. Liu, Y. Chen, and Z. Wen (2010). Appl. Phys. Lett. 96, 054101.
P. Dibandjo, C. Zlotea, R. Gadiou, M. G. Camelia, C. Fermin, L. Michel, E. Leroy, and C. V. Guter (2013). Int. J. Hydrogen Energy 38, 952.
Q. H. Weng, X. B. Wang, C. Y. Zhi, Y. S. Bando, and D. Golberg (2013). ACS Nano 7, 1558.
Office of energy efficiency & renewable energy, fuel cell technologies office, materials-based hydrogen storage (2018). https://www.energy.gov/eere/fuelcells/materials-based-hydrogen-storage.
S. K. Bhatia and A. L. Myers (2006). Langmuir 22, 688.
H. Wu, X. Fan, and J. L. Kuo (2012). Int. J. Hydrogen Energy 37, 14336.
A. Mohajeri and A. Shahsavar (2018). Physica E 101, 167.
C. Li, J. Li, F. Wu, S. S. Li, J. B. Xia, and L. W. Wang (2011). J. Phys. Chem. C 115, 23221.
A. Omidvar and A. Mohajeri (2017). Int. J. Hydrogen Energy 42, 12327.
M. Yoon, S. Y. Yang, C. Hicke, E. G. Wang, D. Geohegan, and Z. Y. Zhang (2018). Phys. Rev. Lett. 100, 206806.
A. Züttel (2003). Mater. Today 6, 24.
S. V. Alapati, J. K. Johnson, and D. S. Sholl (2006). J. Phys. Chem. B 110, 8769.
Y. Song (2013). Phys. Chem. Chem. Phys. 15, 14524.
C. D. Gelatt Jr., H. Ehrenreich, and J. A. Weiss (1987). Phys. Rev. B 17, 1940.
T. Rapps, R. Ahlrichs, E. Waldt, M. M. Kappes, and D. Schooss (2013). Angew. Chem. Int. Ed. 52, 6102.
J. Vanbuel, E. M. Fernández, P. Ferrari, S. Gewinner, W. Schöllkopf, L. C. Balbás, A. Fielicke, and E. Janssens (2017). Chem. Eur. J. 23, 15638.
R. Trivedi and D. Bandyopadhyay (2016). Int. J. Hydrogen Energy 41, 20113.
S. H. Wei, Z. Zeng, J. Q. You, X. H. Yan, and X. G. Gong (2000). J. Chem. Phys. 113, 11127.
A. Mohajeri (2018). J. Alloys Compds. 735, 1962.
L. Guo and Y. Yang (2013). Int. J. Hydrogen Energy 38, 3640.
P. McNelles and F. Y. Naumkin (2009). Phys. Chem. Chem. Phys. 11, 2858.
I. Yarovsky and A. Goldberg (2005). Mol. Simul. 31, 475.
X. Liu, D. Tian, and C. Meng (2015). J. Mol. Struct. 1080, 105.
R. W. P. Wagemans, J. H. van Lenthe, P. E. de Jongh, A. Jos van Dillen, and K. P. de Jong (2005). J. Am. Chem. Soc. 127, 16675.
X. Liu, D. Tian, and C. Meng (2013). Chem. Phys. 415, 179.
E. Durgun, S. Ciraci, W. Zhou, and T. Yildirim (2006). Phys. Rev. Lett. 97.
B. Bogdanovic´, M. Felderhoff, S. Kaskel, A. Pommerin, K. Schlichte and F Schüth, (2003). Adv. Mater. 15, 1012.
A. K. Kandalam, B. Kiran, and P. Jena (2008). J. Phys. Chem. C 112, 6181.
E. Durgun, Y. R. Jang, and S. Ciraci (2007). Phys. Rev. B 76, 073413.
T. Yildirim, J. Iniguez, and S. Ciraci (2005). Phys. Rev. B 72, 153403.
R. R. Boyer (2010). JOM 62, 21.
H. J. Rack and J. I. Qazi (2006). Mater. Sci. Eng. C 26, 1269.
Q. M. Hu, S. J. Li, Y. L. Hao, R. Yang, B. Johansson, and L. Vitos (2008). Appl. Phys. Lett. 93, 121902.
R. Boyer, G. Welsch and E. W. Collings, Materials Properties Handbook: Titanium Alloy, ASM International, Materials Park, OH (1994).
S. W. Cho, C. S. Han, C. N. Park, and E. Akiba (1999). J. Alloys Comp. 288, 294.
M. Bououdina, D. Fruchart, S. Jacquet, L. Pontonnier, and J. L. Soubeyroux (1999). Int. J. Hydrogen Energy 24, 885.
A. Izanlou and M. K. Aydinol (2010). Int. J. Hydrogen Energy 35, 1681.
Y. V. Milman, D. B. Miracle, S. I. Chugunova, I. V. Voskoboinik, N. P. Korzhova, T. N. Legkaya, and Y. N. Podrezov (2001). Intermetallics 9, 839.
Y. Umakoshi, M. Yamaguchi, T. Sakagami, and T. Yamane (1989). J. Mater. Sci. 24, 1599.
W. Chang and B. C. Muddle (2003). Metall. Mater. Trans. A 34, 491.
Ö. Dursun, T. Tansel, E. Hatice, and Ç. İbrahim (2015). Mater. Res. Bull. 18, 813.
Y. Umakoshi, M. Yamaguchi, T. Yamane, and T. Hirano (1998). Philos. Mag. 58, 651.
M. Yamaguchi, Y. Umakoshi, and T. Yamane (1987). Philos. Mag. 55, 301.
W. Chang and B. C. Muddle (1995). Mater. Sci. Eng. A 192, 233.
A. Neville and B. A. B. McDougall (2001). Wear 250, 726.
A. R. Zaganelli, G. E. Pessanha Henriques, I. Ferreira and J. M. D. Almeida Rollo (2008). J. Prosthetic Dent.84, 274.
R. Ibarra, Y. Yamada and A. Goto (1983). Corrosion Fatigue: Mechanism, Metallurgy, Electrochemistry and Engineering, ed. T. W. Crocker, B. N. Leis. West Conshohocken, PA: ASTM International (1983) pp. 135–146.
T. J. D. Kumar, P. Tarakeshwar, and N. Balakrishnan (2008). J. Chem. Phys. 128.
T. J. D. Kumar, P. F. Weck, and N. Balakrishnan (2007). J. Phys. Chem. C 111, 7494.
M.J. Frisch, G.W. Trucks, H.B. Schlegel, G.E. Scuseria, M.A. Robb, J.R. Cheeseman, G. Scalmani, V. Barone, B. Mennucci, G.A. Petersson, H. Nakatsuji, M. Caricato, X. Li, H.P. Hratchian, A.F. Izmaylov, J. Bloino, G. Zheng, J.L. Sonnenberg, M. Hada, M. Ehara, K. Toyota, R. Fukuda, J. Hasegawa, M. Ishida, T. Nakajima, Y. Honda, O. Kitao, H. Nakai, T. Vreven, J.A. Montgomery Jr., J.E. Peralta, F. Ogliaro, M. Bearpark, J.J. Heyd, E. Brothers, K.N. Kudin, V.N. Staroverov, T. Keith, R. Kobayashi, J. Normand, K. Raghavachari, A. Rendell, J.C. Burant, S.S. Iyengar, J. Tomasi, M. Cossi, N. Rega, J.M. Millam, M. Klene, J.E. Knox, J.B. Cross, V. Bakken, C. Adamo, J. Jaramillo, R. Gomperts, R.E. Stratmann, O. Yazyev, A.J. Austin, R. Cammi, C. Pomelli, J.W. Ochterski, R.L. Martin, K. Morokuma, V.G. Zakrzewski, G.A. Voth, P. Salvador, J.J. Dannenberg, S. Dapprich, A.D. Daniels, O. Farkas, J.B. Foresman, J.V. Ortiz, J. Cioslowski and D.J. Fox, Gaussian 09, revision D.01, Gaussian. Inc., Wallingford CT (2013).
M. Ernzerhof and G. E. Scuseria (1999). J. Chem. Phys. 110, 5029.
S. Grimme (2006). Semiempirical J. Chem. Phys. 27, 1787.
M. Castro, S. R. Liu, H. J. Zhai, and L. S. Wang (2003). J. Chem. Phys. 118, 2116.
B. Delley (1990). J. Chem. Phys. 92, 508.
B. Delley (1996). J. Phys. Chem. 100, 6107.
B. Delley (2000). J. Chem. Phys. 113, 7756.
Y. Inada, H. Orita, and K. Accelrys (2007). J. Comput. Chem. 29, 225.
N. S. Venkataramanan, R. Sahara, H. Mizuseki, and Y. Kawazoe (2010). J. Phys. Chem. A 114, 5049.
J. Du, X. Sun, J. Chen, and G. Jiang (2010). J. Phys. B 43, 205103.
P. L. Rodríguez-Kessler and A. R. Rodríguez-Domínguez (2016). J. Phys. Chem. A 120, 2401.
M. Doverstal, L. Karlsson, B. Lindgren, and U. Sassenberg (1997). Chem. Phys. Lett. 270, 273.
M. Salazar-Villanueva, P. H. Hernández Tejeda, U. Pal, J. F. Rivas-Silva, J. I. Rodríguez Mora, and J. A. Ascencio (2006). J. Phys. Chem. A 110, 10274.
S. R. Liu, H. J. Zhai, M. Castro, and L. S. Wang (2013). J. Chem. Phys. 118, 2108.
J. Medina, R. de Coss, A. Tapia, and G. Canto (2010). Eur. Phys. J. B 76, 427.
P. Tarakeshwar, T. J. D. Kumar, and N. Balakrishnan (2008). J. Phys. Chem. A 112, 2846.
L. Wang and D. D. Johnson (2007). J. Am. Chem. Soc. 129, 3658.
G. J. Kubas (2005). Catalysis letters104, 79.
R. Y. Sathe, S. Kumar, and T. J. D. Kumar (2018). Int. J. Hydrogen Energy 43, 5680.
R. Y. Sathe, H. Bae, H. Lee, and T. J. D. Kumar (2020). Int. J. Hydrogen Energy 45, 9936.
Acknowledgments
The authors would like to acknowledge Shiraz University for financial support. We are also immensely grateful to Prof. Mehdi Neek-Amal (Shahid Rajaee Teacher Training University) for helpful comments and Shahid Rajaee Teacher Training University for providing the computing resources.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
No potential conflict of interest was reported by the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Nafissi, S., Hassani, N. & Mohajeri, A. Hydrogen Adsorption on Ti–V Binary and Ti–V–Al Ternary Alloys of Ti11 Cluster. J Clust Sci 34, 1337–1346 (2023). https://doi.org/10.1007/s10876-022-02311-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10876-022-02311-9