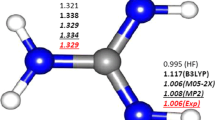
Abstract
We investigated the interactions of uracil with LiF and water using density functional calculations and determined the structures of (Ura)−LiF(H2O)n (n = 0–6) complexes. In all clusters from n = 0 to 6, Li+ of LiF unit binds directly to the O4 site of uracil via electrostatic interaction. (Ura)−LiF connects one water molecule via F− to form a sandwich structure. When the water number reaches 2, the interaction strength between uracil and LiF decreased significantly. Li+ and F− always stay together that exist as contact ion pairs. Excess electrons were found on C6, C4, and both O sites of uracil in NPA charge studies.








Similar content being viewed by others
Data Availability
All data generated or analyzed during this study are included in this published article and in the Supplementary Material.
References
A. Savelyev and A. D. MacKerell (2015). J. Phys. Chem. Lett. 6, 212.
S. Sardar, E. Jabeen, A. Mumtaz, M. Yasinzai, and J.-M. Leveque (2020). Synth. J. Mol. Liq. 300, 112–255.
C. Kalaivanan, M. Sankarganesh, M. Y. Suvaikin, G. B. Karthi, S. Gurusamy, R. Subramanian, and R. N. Asha (2020). J. Mol. Liq. 320, 114423.
X. Hu, H. Li, L. Zhang, and S. Han (2007). J. Phys. Chem. B. 111, 9347.
I. Morfin, F. Horkay, P. J. Basser, F. Bley, A.-M. Hecht, C. Rochas, and E. Geissler (2004). Biophys. J. 87, 2897.
V. Andrushchenko and P. Bouř (2009). J. Phys. Chem. B. 113, 283.
J. Burda, J. Šponer, and J. Leszczynski (Computational Approach, 2012), pp. 1277–1308.
M. Kabeláč and P. Hobza (2006). J. Phys. Chem. B. 110, 14515.
C. Trujillo, A. M. Lamsabhi, O. Mó, M. Yáñez, and J.- Y. Salpin (2011). Int. J. Mass. Spectrom. 306, 27.
N. Russo, M. Toscano, and A. Grand (2001). J. Am. Chem. Soc. 123, 10272.
B. A. Cerda and C. Wesdemiotis (1996). J. Am. Chem. Soc. 118, 11884.
R. Ghiasi, M. Monajjem, F. Asadian, and H. Passdar (2004). J. Chem. Res. 2004, 445.
W. Zhu, X. Luo, C. M. Puah, X. Tan, J. Shen, J. Gu, K. Chen, and H. Jiang (2004). J. Phys. Chem. A. 108, 4008.
A. A. Zinchenko and K. Yoshikawa (2005). Biophys. J. 88, 4118.
D. E. Draper, D. Grilley, and A. M. Soto (2005). Annu. Rev. Bioph. Biom. 34, 221.
K. J. Koch, T. Aggerholm, S. C. Nanita, and R. G. Cooks (2002). J. Mass. Spectrom. 37, 676.
S. Rochut, C. Pepe, J.-P. Paumard, and J.-C. Tabet (2004). Rapid Commun. Mass. Sp. 18, 1686.
K. Rajabi, E. A. L. Gillis, and T. D. Fridgen (2010). J. Phys. Chem. A. 114, 3449.
M. T. Rodgers and P. B. Armentrout (2000). J. Am. Chem. Soc. 122, 8548.
Z. Yang and M. T. Rodgers (2004). J. Am. Chem. Soc. 126, 16217.
Z. Yang and M. T. Rodgers (2006). J. Phys. Chem. A. 110, 1455.
Z. Yang and M. T. Rodgers (2012). Phys. Chem. Chem. Phys. 14, 4517.
E. A. L. Gillis, M. Demireva, K. Nanda, G. Beran, E. R. Williams, and T. D. Fridgen (2012). Phys. Chem. Chem. Phys. 14, 3304.
J. E. D. Bene (1984). J. Phys. Chem. 88, 5927.
M. T. Rodgers and P. B. Armentrout (2007). Int. J. Mass. Spectrom. 267, 167.
N. Russo, M. Toscano, and A. Grand (2001). J. Phys. Chem. B 105, 4735.
G.- Y. Lee (2002). B. Korean. Chem. Soc. 23, 1023.
H. Farrokhpour and S. Khoshkhou (2021). Spectrochim. Acta. A. 244, 118862.
E. A. L. Gillis, K. Rajabi, and T. D. Fridgen (2009). J. Phys. Chem. A. 113, 824.
M. Gebala, G. M. Giambaşu, J. Lipfert, N. Bisaria, S. Bonilla, G. Li, D. M. York, and D. Herschlag (2015). J. Am. Chem. Soc. 137, 14705.
A. Savelyev and G. A. Papoian (2006). J. Am. Chem. Soc. 128, 14506.
M. J. Frisch, G. W. Trucks, H. B. Schlegel, G. E. Scuseria, and M. A. Robb (2009). Gaussian 09.
S. Grimmean, J. Antony, S. Ehrlicdh, and H. Krieg (2010). J. Chem. Phys. 132, 154104.
E. S. Kryachko, M. T. Nguyen, and T. Zeegers-Huyskens (2001). J. Phys. Chem. A. 105, 1288.
R. D. Brown, P. D. Godfrey, D. McNaughton, and A. P. Pierlot (1988). J. Am. Chem. Soc. 110, 2329.
O. A. Vydrov, J. Heyd, A. V. Krukau, and G. E. Scuseria (2006). J. Chem. Phys. 125, 074106.
O. A. Vydrov and G. E. Scuseria (2006). J. Chem. Phys. 125, 234109.
O. A. Vydrov, G. E. Scuseria, and J. P. Perdew (2007). J. Chem. Phys. 126, 154109.
J.-D. Chai and M. Head-Gordon (2008). Phys. Chem. Chem. Phys. 10, 6615.
E. R. Johnson, S. Keinan, P. Mori-Sánchez, J. Contreras-García, A. J. Cohen, and W. Yang (2010). J. Am. Chem. Soc. 132, 6498.
R. F. W. Bader (1991). Chem. Rev. 91, 893.
M. Z. Hopffgarten and G. Frenking (2012). Wires. Comput. Mol. Sci. 2, 43.
T. Lu and F. Chen (2012). J. Comput. Chem. 33, 580.
R.-Z. Li, C.- W. Liu, Y. Q. Gao, H. Jiang, H.- G. Xu, and W.- J. Zheng (2013). J. Am. Chem. Soc. 135, 5190.
J. Akola, M. Manninen, H. Häkkinen, U. Landman, X. Li, and L. S. Wang (2000). Phys. Rev. B 62, 13216.
J. A. Sordo (2001). J. Mol. Struct. (Theochem) 537, 245.
K. Vanommeslaeghe, P. Mignon, S. Loverix, D. Tourwé, and P. Geerlings (2006). J. Chem. Theory. Comput. 2, 1444.
R.-Z. Li, Y.-Y. Liu, and M. Yang (2017). Comput Theor. Chem. 1115, 119.
Acknowledgements
This work was supported by the Natural Science Foundation of Shaanxi, China (Grant No. 2019JM-292). Part of the theoretical calculations was conducted on the ScGrid and DeepComp 7000 of the Supercomputing Center, Computer Network Information Center of the Chinese Academy of Sciences.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
There are no conflicts of interest to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Xu, XY., Li, RZ., Lu, L. et al. Interaction of Uracil with LiF and Water Studied by Density Functional Theory Study on Anionic Complexes. J Clust Sci 34, 1249–1258 (2023). https://doi.org/10.1007/s10876-022-02296-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10876-022-02296-5