Abstract
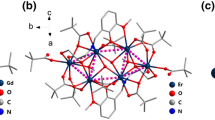
Employing tripodal Schiff base ligand of tris[4-(2-hydroxy-3-methoxyphenyl)-3-aza-3-butenyl]amine (H3L1) and auxiliary ligand of 2-hydroxy-5-chlorobenzaldehyde, a 3d–4f cluster of \(\left[ {{\text{Dy}}^{{{\text{III}}}} {{{\text{Mn}}}}_{ 2}^{{{\text{II}}}} \left( {{{\text{L}}}^{ 2} } \right)_{ 2} } \right]\left[ {{{\text{NO}}}_{ 3} } \right] \cdot 5 {{{\text{CH}}}}_{ 3} {{{\text{OH}}}}\) (1) was synthesized. Intriguingly, the H3L1 ligand has transformed into another Schiff base ligand of {N-[4-(2-hydroxy-5-chlorophenyl)-3-aza-3-butenyl]-N′,N′′-bis[4-(2-hydroxy-3-methoxyphenyl)-3-aza-3-butenyl]}amine (H3L2) in situ during the coordination process, that one of terminal groups of H3L1 ligand was substituted. A pair of such (L2)3− ligands chelate a DyIII and two MnII ions to form a quasi-liner {MnIIDyIIIMnII} skeleton of 1, in which the adjacent MnII and DyIII ions were bridged by three Ophenol. To the best of our knowledge, this in situ substitution of the terminal group of Schiff base ligand has not been reported in previous literature. DC magnetic susceptibility measurements were conducted on 1, revealing the weak antiferromagnetic interactions between the DyIII and MnII centers.
Graphic Abstract
A quasi-liner a {MnIIDyIIIMnII} cluster has been constructed from tripodal Schiff base ligand of H3L1. The H3L1 ligands underwent an in situ substitution in the coordination, which has been not reported in previous literature so far as we known. Magnetic studies reveal the weak antiferromagnetic interactions between DyIII and MnII ions.








Similar content being viewed by others
References
Y. S. Meng, S. D. Jiang, B. W. Wang, and S. Gao (2016). Acc. Chem. Res. 49, 2381.
D. N. Woodruff, R. E. P. Winpenny, and R. A. Layfield (2013). Chem. Rev. 113, 5110.
Z. H. Zhu, M. Guo, X. L. Li, and J. K. Tang (2018). Coordin. Chem. Rev. 378, 350.
J. L. Liu, Y. C. Chen, and M. L. Tong (2018). Chem. Soc. Rev. 47, 2431.
C. A. P. Goodwin, F. Ortu, D. Reta, N. F. Chilton, and D. P. Mills (2017). Nature 548, 439.
F. S. Guo, B. M. Day, Y. C. Chen, M. L. Tong, A. Mansikkamäki, and R. A. Layfield (2017). Angew. Chem. Int. Ed. 56, 11445.
F. S. Guo, B. M. Day, Y. C. Chen, M. L. Tong, A. Mansikkamäki, and R. A. Layfield (2018). Science 362, 1400.
M. Gregson, N. F. Chilton, A. M. Ariciu, F. Tuna, I. F. Crowe, W. Lewis, A. J. Blake, D. Collison, E. J. L. McInnes, R. E. P. Winpenny, and S. T. Liddle (2016). Chem. Sci. 7, 155.
F. Matilde, C. V. Julio, M. G. D. Ana, S. M. Jesús, M. H. Juan, and C. Enrique (2017). Inorg. Chem. 56, 5646.
Q. Q. Yang and J. K. Tang (2019). Dalton Trans. 48, 769.
M. Andruh, J. P. Costes, C. Diaz, and S. Gao (2009). Inorg. Chem. 48, 3342.
S. Goswami, K. M. Amit, and K. Sanjit (2015). Inorg. Chem. Front. 2, 687.
J. Li, R. M. Wei, T. C. Pu, F. Cao, L. Yang, Y. Han, Y. Q. Zhang, J. L. Zuo, and Y. Song (2017). Inorg. Chem. Front. 4, 114.
L. R. Piquer and E. C. Sañudo (2015). Dalton. Trans. 44, 8771.
C. Papatriantafyllopoulou, W. Wernsdorfer, K. A. Abboud, and G. Christou (2011). Inorg. Chem. 50, 421.
M. Holynska, D. Premuzic, I. R. Jeon, W. Wernsdorfer, R. Clerac, and S. Dehnen (2011). Chem. Eur. J. 17, 9605.
T. C. Stamatatos, S. J. Teat, W. Wernsdorfer, and G. Christou (2018). Angew. Chem. Int. Ed. 48, 521.
V. M. Mereacre, A. M. Ako, R. Clérac, W. Wernsdorfer, G. Filoti, J. Bartolome, C. E. Anson, and A. K. Powell (2007). J. Am. Chem. Soc. 129, 9248.
S. Langley, B. Moubaraki, and K. S. Murray (2010). Dalton Trans. 39, 5066.
M. M. Hänninen, A. J. Mota, R. Sillanpää, S. Dey, G. Velmurugan, G. Rajaraman, and E. Colacio (2018). Inorg. Chem. 57, 3683.
L. B. L. Escobar, G. P. Guedes, S. Soriano, R. A. A. Cassaro, J. Marbey, S. Hill, M. A. Novak, M. Andruh, and M. G. F. Vaz (2018). Inorg. Chem. 57, 326.
K. Griffiths and G. E. Kostakis (2018). Dalton Trans. 47, 12011.
X. X. Zhou, Y. P. Cai, S. Z. Zhu, Q. G. Zhan, M. S. Liu, Z. Y. Zhou, and L. Chen (2008). Cryst. Growth Des. 8, 2076.
Q. Wei, Z. P. Zheng, H. X. Feng, X. J. Hong, X. Huang, H. J. Peng, and Y. P. Cai (2016). RSC Adv. 6, 94687.
S. Hu, J. C. Chen, M. L. Tong, B. Wang, Y. X. Yan, and S. R. Batten (2015). Angew. Chem. Int. Ed. 44, 5471.
S. J. Bradberry, A. J. Savyasachi, M. M. Calvo, and T. G. Coord (2014). Chem. Rev. 226, 273.
K. Wang, Z. L. Chen, H. H. Zou, K. Hu, H. Y. Li, Z. Zhang, W. Y. Sun, and F. P. Liang (2016). Chem. Commun. 52, 8297.
K. Wang, Z. L. Chen, H. H. Zou, S. H. Zhang, Y. Li, X. Q. Zhang, W. Y. Sun, and F. P. Liang (2018). Dalton Trans. 47, 233.
J. P. Costes, F. Dahan, A. Dupuis, S. Lagrave, and J. P. Laurent (1998). Inorg. Chem. 37, 153.
G. M. Sheldrick SHELXL, program for crystal structure refinement (University of Go¨ttingen, Germany, 2014).
G. M. Sheldrick (2015). Acta Crystallogr. C 71, 3.
O. V. Dolomanov, L. J. Bourhis, R. J. Gildea, J. A. K. Howard, and H. Puschmann (2009). J. Appl. Crystallogr. 42, 339.
L. J. Bourhis, O. V. Dolomanov, R. J. Gildea, J. A. K. Howard, and H. Puschmann (2015). Acta Crystallogr. A 71, 59.
H. Zabrodsky, S. Peleg, and D. Avnir (1992). J. Am. Chem. Soc. 114, 7843.
M. Pinsky and D. Avnir (1998). Inorg. Chem. 37, 5575.
E. M. Pineda, N. F. Chilton, F. Tuna, R. E. P. Winpenny, and E. J. L. McInnes (2015). Inorg. Chem. 54, 5930.
Y. Peng, M. K. Singh, V. Mereacre, C. E. Anson, G. Rajaraman, and A. K. Powell (2019). Chem. Sci. 10, 5528.
Acknowledgements
This work was financially supported by the National Natural Science Foundation of China (Nos. 21961008 and 21771043) and the Guangxi Natural Science Foundation (No. 2018GXNSFAA138123).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Xu, F., Luo, XJ., Wu, CE. et al. A Quasi-Liner {MnIIDyIIIMnII} Cluster Featuring In Situ Schiff Base Ligand Transformation. J Clust Sci 32, 1411–1418 (2021). https://doi.org/10.1007/s10876-020-01901-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10876-020-01901-9