Abstract
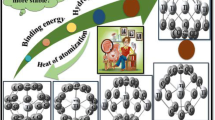
Structural, electronic and adsorption characteristics of TM@C70 (TM = V, Cr, Mn, Fe, Co, Ni) endohedral fullerenes are addressed by first principles calculations, through density functional theory. A thorough analysis is conducted to explore them employing formation mechanism, adsorption energy and frontier orbitals. The essential role of each TM atom on the C70 fullerene molecule is elucidated by means of stability, charge distribution, frontier orbitals, reactivity definers, energy and induced magnetic moment. It is found that TM@C70 complexes are stable molecules. The analysis on the charge population demonstrates that the direction of charge transfer is toward the C70 cage. HOMO–LUMO energy gap modified by the TM atom is correlated to hardness, softness, electronegativity and electrophilicity index. The catalytic activity and adsorption properties of TM@C70 structures are examined through 4-nitro thiophenol. They exhibit high chemical reactivity and favorable adsorption, indicating that the TM@C70 molecules are plausible in catalytic studies.





Similar content being viewed by others
References
H. W. Kroto, J. R. Heath, S. C. O’Brien, R. F. Curl, and R. E. Smalley (1985). Nature 318, 162.
W. Krätschmer, L. D. Lamb, K. Fostiropoulos, and D. R. Huffman (1990). Nature 347, 354.
B. Paulus (2003). Phys. Chem. Chem. Phys. 5, 3364–3367.
L. Echegoyen and L. E. Echegoyen (1998). Account. Chem. Res. 31, 593–601.
C. Roland, B. Larade, J. Taylor, and H. Guo (2001). Phys. Rev. B 65, 041401(R).
S. Caliskan (2018). Phys E 99, 43–50.
M. Kaur, R. S. Sawhney, and D. Engles (2016). J. Mater. Res. 31, 2025–2034.
H. Prinzbach, A. Weiler, P. Landenberger, F. Wahl, J. Wörth, L. T. Scott, M. Gelmont, D. Olevano, and B. V. Issendorff (2000). Nature 407, 60.
X.-J. Zhang, M.-Q. Long, K.-Q. Chen, Z. Shuai, Q. Wan, B. S. Zou, and Y. Zhang (2009). Appl. Phys. Lett. 94, 073503.
T. Pradeep, V. Vijayakrishnan, A. K. Santra, and C. N. R. Rao (1991). J. Phys. Chem. 95, 10564–10565.
X. Zhong, R. Pandey, A. R. Rocha, and S. P. Karna (2010). J. Phys. Chem. Lett. 1, 1584–1589.
Y.-P. An, C.-L. Yang, M.-S. Wang, X.-G. Ma, and D.-H. Wang (2009). J. Chem. Phys. 131, 024311.
K. S. Vinit and C. N. Sujith (2016). Ramachandran. J. Phys. Chem. A. 120, 6990–6997.
A. Equbal, S. Srinivasan, C. N. Ramachandran, and N. Sathyamurthy (2014). Chem. Phys. Lett. 610–611, 251–255.
L. Jing-Nan, G. Bing-Lin, and H. Ru-Shan (1992). Solid State Commun. 84, 807–810.
H. Sharma, I. Garg, K. Dharamvir, and V. K. Jindal (2009). J. Phys. Chem. A 113, 9002–9013.
D. S. Bethune, R. D. Johnson, J. R. Salem, M. S. de Vries, and C. S. Yannoni (1993). Nature 366, 123.
E. Dietel, A. Hirsch, B. Pietzak, M. Waiblinger, K. Lips, A. Weidinger, A. Gruss, and K. P. Dinse (1999). J. Am. Chem. Soc. 121, 2432–2437.
A. A. Popov, S. F. Yang, and L. Dunsch (2013). Chem. Rev. 113, 5989–6113.
J. U. Ahamed, S. Miyanaga, T. Kaneko, and R. Hatakeyama (2009). Trans. Mater. Res. Soc. Japan 34, 773–776.
J. Cioslowski and E. D. Fleischmann (1991). J. Chem. Phys. 94, 3730–3734.
A. Weidinger, M. Waiblinger, B. Pietzak, and T. Almeida Murphy (1998). Appl. Phys. A 66, 287–292.
Y.-P. An, C.-L. Yang, M.-S. Wang, X.-G. Ma, and D.-H. Wang (2011). J. Clust. Sci. 22, 31–39.
M. Bezi Javan, N. Tajabor, M. Behdani, and M. Rezaee Rokn-Abadi (2010). Phys. B 405, 4937–4942.
M. B. Javan, N. Tajabor, M. R. Roknabadi, and M. Behdani (2011). Phys. E 43, 1351–1359.
G. Li, R. F. Sabirianov, J. Lu, X. C. Zeng, and W. N. Mei (2008). J Chem. Phys. 128, 074304.
I. Zutic, J. Fabian, and S. Das Sarma (2004). Rev. Mod. Phys. 76, 323–410.
A. R. Rocha, V. M. Garcia-Suarez, S. W. Bailey, C. J. Lambert, J. Ferrer, and S. Sanvito (2005). Nat. Mater. 4, 335–339.
D. Waldron, P. Haney, B. Larade, A. MacDonald, and H. Guo (2006). Phys. Rev. Lett. 96, 166804.
S. Caliskan and A. Laref (2014). Sci. Rep. 4, 7363.
S. Caliskan and A. Laref (2014). Phys. Chem. Chem. Phys. 16, 13191–13208.
M. T. Baei, A. Soltani, P. Torabi, and F. Hosseini (2014). Monatshefte für Chemie Chem. Mon. 145, 1401–1405.
A. S. Rad and K. Ayub (2018). Mater. Res. Bull. 97, 399–404.
D. R. McKenzie, C. A. Davis, D. J. H. Cockayne, D. A. Muller, and A. M. Vassallo (1992). Nature 355, 622.
Y. Morinaka, R. Zhang, S. Sato, H. Nikawa, T. Kato, K. Furukawa, M. Yamada, Y. Maeda, M. Murata, A. Wakamiya, S. Nagase, T. Akasaka, and Y. Murata (2017). Angew Chem. Int. Ed. 129, 6588–6591.
S. Caliskan (2019). Physica E 108, 83–89.
S. Saito and A. Oshiyama (1991). Phys Rev B 44, 11532–11535.
P. Wurz, K. R. Lykke, M. J. Pellin, and D. M. Gruen (1991). J Appl. Phys. 70, 6647–6652.
D. Changgeng, Y. Jinlong, H. Rongsheng, and W. Kelin (2001). Phys. Rev. A 64, 043201.
M. Brandbyge, J. L. Mozos, P. Ordejon, J. Taylor, and K. Stokbro (2002). Phys. Rev. B 65, 165401.
Synopsys QuantumATK. https://www.synopsys.com/silicon/quantumatk.html.
J. P. Perdew, K. Burke, and M. Ernzerhof (1996). Phys. Rev. Lett. 77, 3865–3868.
Z. Rostami, A. Hosseinian, and A. Monfared (2018). J. Mol. Graph. Model. 81, 60–67.
X. Zhang, Y. Liu, X. Ma, and B. Abulimiti (2019). J. Clust. Sci. 30, 319–328.
Q. Xiang, Y. Liu, X. Zhang, Y. Duan, A. Bumaliya, M. Xiang (2019). J. Clust. Sci. https://doi.org/10.1007/s10876-019-01700-x.
S. M. Lee, R. J. Nicholls, D. Nguyen-Manh, D. G. Pettifor, G. A. D. Briggs, S. Lazar, D. A. Pankhurst, and D. J. H. Cockayne (2005). Chemi. Phys. Lett. 404, 206–211.
N. Troullier and J. L. Martins (1991). Phys. Rev. B 43, 1993–2006.
H. J. Monkhorst and J. D. Pack (1976). Phys Rev B 13, 5188–5192.
J. M. Soler, E. Artacho, J. D. Gale, A. Garcia, J. Junquera, P. Ordejon, and D. Sanchez-Portal (2002). J. Phys-Condens Mat. 14, 2745–2779.
T. Koopmans (1934). Physica 1, 104–113.
R. G. Pearson (1985). J Am Chem Soc 107, 6801–6806.
R. G. Parr, R. A. Donnelly, M. Levy, and W. E. Palke (1978). J. Chem. Phys. 68, 3801–3807.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Caliskan, S. Structural, Electronic and Adsorption Characteristics of Transition Metal doped TM@C70 Endohedral Fullerenes. J Clust Sci 32, 77–84 (2021). https://doi.org/10.1007/s10876-020-01762-2
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10876-020-01762-2