Abstract
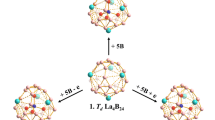
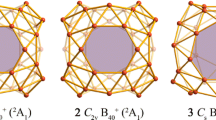
The newly discovered cage-like borospherene D2d B40 with two η6-B6 hexagons and four η7-B7 heptagons on the surface may serve as an effective multi-dentate ligand to coordinate transition metals. Based upon extensive density functional theory calculations, we present herein the possibility of the exohedral complex series (C6H6Cr)n&B40 (n = 1–6) with n Cr centres sandwiched between the central η6/7-B40 unit and n planar η6-C6H6 ligands. C6H6Cr fragments in (C6H6Cr)n&B40 occupy the η6-B6 hexagonal coordination site atop the B40 cage first, the four neighboring η7-B7 heptagons on the waist next, and finally the η6-B6 hexagon at the bottom, forming a multinuclear complex series effectively stabilized by n C6H6Cr fragments. The two η6-B6 hexagons and four η7-B7 heptagons on the spherical surface of B40 can be practically viewed as six independent coordination sites to coordinate Cr centers with almost the same coordination energies. Detailed bonding analyses indicate that the eclipsed C2v C6H6Cr&B40 (1-1) possesses a coordination bonding pattern similar to that of dibenzenechromium, with the B40 ligand inheriting the σ + π double delocalization bonding pattern of the parent borospherene. The IR, Raman, and UV–vis absorption spectra of C2v C6H6Cr&B40 (1-1) are theoretically simulated to facilitate its future experimental characterization.




Similar content being viewed by others
References
J. W. Buchanan, G. A. Grieves, J. E. Reddic, and M. A. Duncan (1999). Int. J. Mass. Spectrom. 182, 323.
E. D. Jemmis, M. Manoharan, and P. K. Sharma (2000). Organometallics. 19, 1879.
E. D. Jemmis and P. K. Sharma (2001). J. Mol. Graph. Moedl. 19, 256.
E. G. Galpern, A. R. Sabirov, and I. V. Stankevich (2007). Phys. Solid. State. 49, 2330.
R. Salcedo (2009). Polyhedron. 28, 431.
J. I. Martínez, J. M. García-Lastra, M. J. López, and J. A. Alonso (2010). J. Chem. Phys. 132, 044314.
L. Y. Zhu, T. T. Zhang, M. X. Yi, and J. L. Wang (2010). J. Phys. Chem. A. 114, 9398.
Z. Yang, B. Zhang, X. Liu, Y. Yang, X. Li, S. Xiong, and B. Xu (2013). J. Phys. B: At. Mol. Opt. Phys. 46, 035101.
M. Sawamura, Y. Kuninobu, M. Toganoh, Y. Matsuo, M. Yamanaka, and E. Nakamura (2002). J. Am. Chem. Soc. 124, 9354.
Y. Matsuo, K. Tahara, and E. Nakamura (2006). J. Am. Chem. Soc. 128, 7154.
H. J. Zhai, Y. F. Zhao, W. L. Li, Q. Chen, H. Bai, H. S. Hu, Z. A. Piazza, W. J. Tian, H. G. Lu, Y. B. Wu, Y. W. Mu, G. F. Wei, Z. P. Liu, J. Li, S. D. Li, and L. S. Wang (2014). Nat. Chem. 6, 727.
Q. Chen, H. R. Li, W. J. Tian, H. G. Lu, H. J. Zhai, and S. D. Li (2016). Phys. Chem. Chem. Phys. 18, 14186.
W. J. Tian, Q. Chen, H. R. Li, M. Yan, Y. W. Mu, H. G. Lu, H. J. Zhai, and S. D. Li (2016). Phys. Chem. Chem. Phys. 18, 9922.
Q. Chen, H. R. Li, C. Q. Miao, Y. J. Wang, H. G. Lu, Y. W. Mu, G. M. Ren, H. J. Zhai, and S. D. Li (2016). Phys. Chem. Chem. Phys. 18, 11610.
Q. Chen, S. Y. Zhang, H. Bai, W. J. Tian, T. Gao, H. R. Li, C. Q. Miao, Y. W. Mu, H. G. Lu, H. J. Zhai, and S. D. Li (2015). Angew. Chem. Int. Edit. 54, 8160.
Q. Chen, W. L. Li, Y. F. Zhao, S. Y. Zhang, H. S. Hu, H. Bai, H. R. Li, W. J. Tian, H. G. Lu, H. J. Zhai, S. D. Li, J. Li, and L. S. Wang (2015). Acs Nano. 9, 754.
Z. Rostami and F. Firoznasab (2018). J. Mol. Model. 24, 194.
M. Moradi, Z. Bagheri, and A. Bodaghi (2017). Physica. E. 89, 148.
E. Shakerzadeh, Z. Biglari, and E. Tahmasebi (2016). Chem. Phys. Lett. 654, 76.
W. Fa, S. Chen, S. Pande, and X. C. Zeng (2015). J. Phys. Chem. A. 119, 11208.
H. Bai, B. Bai, L. Zhang, W. Huang, Y. W. Mu, H. J. Zhai, and S. D. Li (2016). Sci. Rep. 18, (6), 35518.
Y. An, M. Zhang, D. Wu, Z. Fu, T. Wang, and C. Xia (2016). Phys. Chem. Chem. Phys. 18, 12024.
H. Bai, Q. Chen, H. J. Zhai, and S. D. Li (2015). Angew. Chem. Int. Edit. 54, 941.
C. Z. Wang, T. Bo, J. H. Lan, Q. Y. Wu, Z. F. Chai, J. K. Gibson, and W. Q. Shi (2018). Chem. Commun. 54, 2248.
T. R. Yu, Y. Gao, D. Xu, and Z. Wang (2017). Nano. Res. 11, 354.
S. X. Li, Z. P. Zhang, Z. W. Long, and S. J. Qin (2017). RSC. Adv. 7, 38526.
W. Wang, Y. D. Guo, and X. H. Yan (2016). RSC Adv. 6, 40155.
P. Jin, Q. Hou, C. Tang, and Z. Chen (2015). Theor. Chem. Acc. 134, 13.
C. M. Tang and X. Zhang (2016). Int. J. Hydrogen Energ. 41, 16992.
H. R. Li, X. X. Tian, X. M. Luo, M. Yan, Y. W. Mu, H. G. Lu, and S. D. Li (2017). Sci. Rep. 7, 5701.
N. Karmodak and E. D. Jemmis (2016). Chem. Asian J. 11, 3350.
R. Krishnan, J. S. Binkley, R. Seeger, and J. A. Pople (1980). J. Chern. Phys. 72, 650.
P. V. R. Schleyer, C. Maerker, A. Dransfeld, H. Jiao, and N. J. R. van Hommes (1996). J. Am. Chem. Soc. 118, 6317.
Z. Chen, C. S. Wannere, C. Corminboeuf, R. Puchta, and P. V. Schleyer (2005). Chem. Rev. 105, (10), 3842.
E. D. Glendening, J. K. Badenhoop, A. E. Reed, J. E. Carpenter, J. A. Bohmann, C. M. Morales, C. R. Landis and F. Weinhold (2013). NBO 6.0. (http://nbo6.chem.wisc.edu).
D. Y. Zubarev and A. I. Boldyrev (2008). Phys. Chem. Chem. Phys. 10, 5207.
D. Y. Zubarev and A. I. Boldyrev (2008). J. Org. Chem. 73, 9251.
Riidiger Bauernschmitt and R. Ahlrichs (1996). Chem. Phys. Lett. 256, 454.
M. E. Casida, C. Jamorski, K. C. Casida, and D. R. Salahub (1998). J. Chem. Phys. 108, 4439.
M. J. Frisch, et al. (2016) Gaussian 16, Revision B.01, Gaussian Inc., Wallingford, CT.
B. Jiang, M. H. Tang, J. C. Li, Y. G. Xiao, Z. H. Tang, H. Q. Cai, X. S. Lv, and Y. C. Zhou (2012). J. Phys. D: Appl. Phys. 45, 025102.
Y. Yuan and L. J. Cheng (2013). J. Chem. Phys. 138, 024301.
C. Xu, L. J. Cheng, and J. L. Yang (2015). Int. J. Quant. Chem. 115, 1621.
Acknowledgements
The work was supported by the National Natural Science Foundation of China (21720102006 to S.-D. Li).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All the authors of this paper have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Pei, L., Li, DZ., Li, HR. et al. (C6H6Cr)n&B40 (n = 1–6): Exohedral Borospherene Complexes with Cage-like B40 as an Effective Ligand with Multiple Coordination Sites. J Clust Sci 31, 1363–1369 (2020). https://doi.org/10.1007/s10876-019-01747-w
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10876-019-01747-w