Abstract
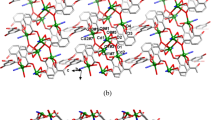
The organic ligand 1,10-phenanthroline-2,9-dicarboxylic acid (H2L) was used to react with ZnSO4·7H2O at 140 °C for two days to obtain the complex [Zn6(L)6(H2SO4)]·3CH3CN·Et3N (1). The structure of complex is confirmed by X-ray crystallography, TG and PXRD. The structure suggested that six ligands chelated with six Zn2+ ions. Every five-coordinated Zn(II) ion is surrounded by two N atoms and three O atoms (one sulfate radical or three ligands) with the N2O3 coordination environment. Each Zn(II) ion in the structure of complex 1 binds only one L2− ligand. The metal center Zn2 and Zn3 are bridged by SO42−. The photoluminescence of complex 1 is obvious. Moreover, in the presence of Cd2+ ions, the complex exhibits an efficient recognition ability, and it realizes the recognition of toxic metal ions.





Similar content being viewed by others

References
H. N. Kim, W. X. Ren, J. S. Kim, and J. Yoon (2012). Chem. Soc. Rev.41, 3210.
W. de Vries, P. F. Römkens, and G. Schütze (2007). Rev. Environ. Contam. Toxicol.191, 91.
C. N. McFarland, L. I. Bendell-Young, C. Guglielmo, and T. D. Williams (2002). J. Environ. Monit.4, 791.
Y. Shi, R. Wang, Y. Lu, S. Song, A. C. Johnson, A. Sweetman, and K. Jones (2016). Environ. Int.94, 283.
Y. Liu, T. Xiao, R. B. Perkins, J. Zhu, Z. Zhu, Y. Xiong, and Z. Ning (2017). J. Geochem. Explor.176, 42.
D. Sarkar, A. Pramanik, and S. Jana (2015). Sens. Actuators B Chem.209, 138.
Y. Ma, F. Wang, and S. Kambam (2013). Sens. Actuators B Chem.188, 1116.
X. Zhou, P. Li, Z. Shi, X. Tang, C. Chen, and W. Liu (2012). Inorg. Chem.51, 9226.
L. Xue, C. Liu, and H. Jiang (2009). Org. Lett.11, 1655.
L. Jarup and A. Akesson (2009). Toxicol Appl. Pharmacol.238, 201.
G. F. Jiang, L. Xu, S. Z. Song, C. C. Zhu, Q. Wu, and L. Zhang (2008). Toxicology244, 49.
A. Sil, A. Maity, D. Giri, and S. K. Patra (2016). Sens. Actuators B Chem.226, 403.
S. Park and H. J. Kim (2012). Sens. Actuators B Chem.168, 376.
S. Ellairaja, R. Manikandan, and M. T. Vijayan (2015). RSC Adv.5, 63287.
P. Wang, J. Wu, and L. Liu (2015). Dalton Trans.44, 18057.
J. Han, X. Tang, Y. Wang, R. Liu, L. Wang, and L. Ni (2018). Spectrochim. Acta A Mol. Biomol. Spectrosc.205, 597.
W.-M. Liao, M.-J. Wei, J.-T. Mo, P.-Y. Fu, Y.-N. Fan, M. Pan, and C.-Y. Su (2019). Dalton Trans.48, 4489.
J.-M. Li, R. Li, and X. Li (2018). CrystEngComm20, 4962.
W.-Q. Tong, T.-T. Liu, G.-P. Li, J.-Y. Liang, L. Hou, and Y.-Y. Wang (2018). New J. Chem.42, 9221.
H. Li, Y. Han, Z. Shao, N. Li, C. Huang, and H. Hou (2017). Dalton Trans.46, 12201.
G. Z. Edwin, C. Constable, C. E. Housecroft, and J. A. Zampese (2012). Polyhedron44, 150.
K. C. Gupta and A. K. Sutar (2008). Coordin. Chem. Rev.252, 1420.
P. Jiang and Z. Guo (2004). Coordin. Chem. Rev.248, 205.
Z.-N. Lu, L. Wang, X. Zhang, and Z.-J. Zhu (2019). Spectrochim. Acta A Mol. Biomol. Spectrosc.213, 57.
A. Kumar Shaily and N. Ahmed (2017). New J. Chem.41, 14746.
H. Singh, R. Sharma, G. Bhargava, S. Kumar, and P. Singh (2018). ChemistrySelect3, 7840.
Q. Wang, Y. Tan, N. Wang, Z. Lu, and W. Wang (2018). Spectrochim. Acta A Mol. Biomol. Spectrosc.201, 216.
Y. Zhou, Y. Xiao, and X. Qian (2008). Tetrahedron Lett.49, 3380.
J.-N. Hao and B. Yan (2015). Chem. Commun.51, 7737.
S. Pandey, P. Kumar, and R. Gupta (2018). Dalton Trans.47, 14686.
W.-M. Liao, M.-J. Wei, J.-T. Mo, P.-Y. Fu, Y.-N. Fan, M. Pan, and C.-Y. Su (2019). Dalton Trans.48, 4489.
Y. D. Farahani and V. Safarifard (2019). J. Solid. State. Chem.275, 131.
G. M. Sheldrick (2015). Acta Crystallogr. Sect. C Struct. Chem.71, 3–8.
Acknowledgements
This work was supported by the Foundation of Key Laboratory of Functional Metal-Organic Compounds of Hunan Province (M018K05).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflicts of interest
There are no conflicts to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Sheng, LB., Liu, MQ., Li, SX. et al. Highly Selective Sensing of Cd(II) Ion by Hexanuclear Functionalized Zn(II) Cluster. J Clust Sci 31, 429–434 (2020). https://doi.org/10.1007/s10876-019-01656-y
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10876-019-01656-y