Abstract
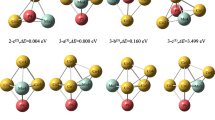
In this work, the results of DFT-based calculations on the structures, stabilities, vibrational, electronic, and hydrogen storage behavior of (CaO) n rings are presented and discussed systematically. The equilibrium ring structures of Ca–O clusters for n = 2–5 are found to be stable. Vibrational frequencies and IR intensities further support the enhanced stability with an increase in the size of Ca–O clusters. The HOMO–LUMO surfaces and their derived parameters are used to explain the electronic properties of the titled systems. For efficient hydrogen storage, metals especially, the transition metals with large cohesive energy (CE) suffer from the problem of cohesion as it is expected that the adsorption energies of metal decorated absorbents should be larger than the CEs of metal. In order to avoid this, hydrogen adsorbed directly on the absorbents is preferred. Due to relatively smaller CEs of the s-block metals, hydrogen adsorbs directly on the cluster which indeed solves the problem of cohesion. The hydrogen storage capacity of (CaO) n clusters, considering hydrogen adsorption on (CaO)4 and (CaO)5 rings is studied. The outcomes appear to give meaningful and satisfactory results. Thus the present work is expected to lead further the applications of small clusters for easy, efficient, and eco-friendly hydrogen storage.





Similar content being viewed by others
References
A. Jain, V. Kumar, M. Sluiter, and Y. Kawazoe (2006). Comput. Mater. Sci. 36, 171–175.
P. N. Kapoor, A. K. Bhagi, R. S. Mulukutla, and K. J. Klabunde Dekker Encyclopedia of Nanoscience and Technology (Marcel Dekker, New York, 2004).
A. Khaleel, P. N. Kapoor, and K. J. Klabunde (1999). NanoStruct. Mat. 11, 459–468.
R. C. Whited, C. J. Flaten, and W. C. Walker (1973). Solid State Commun. 13, 1903–1905.
I. S. Elfimov, S. Yunoki, and G. A. Sawatzky (2002). Phys. Rev. Lett. 89, 216403–216407.
B. K. Olga, L. Isabelle, and V. Alexander (1997). Chem. Mater. 9, 2468–2480.
O. Koper, X. L. Yong, and J. K. Kenneth (1993). Chem. Mater. 5, 500–505.
W. A. Saunders (1988). Phys. Rev. B 37, 6583–6586.
T. P. Martin and T. Bergmann (1989). J. Chem. Phys. 90, 6664–6667.
P. J. Ziemann and A. W. Castleman (1992). J. Phys. Chem. 96, 4271–4276.
F. Bawa and I. Panas (2002). Phys. Chem. Chem. Phys. 4, 103–108.
A. Vibok and G. J. Halasz (2001). Phys. Chem. Chem. Phys. 3, 3042–3047.
J. L. C. Rowsell and O. M. Yaghi (2006). J. Am. Chem. Soc. 128, 1304–1315.
A. M. Seayad and D. M. Antonelli (2004). Adv. Mater. 16, 765–777.
S. K. Bhatia and A. L. Myers (2006). Langmuir 22, 1688–1700.
Q. Sun, Q. Wang, P. Jena, and Y. Kawazoe (2005). J. Am. Chem. Soc. 127, 14582–14583.
Q. Sun, P. Jena, Q. Wang, and M. Marquez (2006). J. Am. Chem. Soc. 128, 9741–9745.
J. Zhou, Q. Wang, Q. Sun, and P. Jena (2011). J. Phys. Chem. C 115, 6136–6140.
T. Hussain, B. Pathak, T. A. Maark, C. M. Araujo, R. H. Scheicher, and R. Ahuja (2011). Europhys. Lett. 96, 27013–27016.
H. Dodziuk and G. Dolgonos (2002). Chem. Phys. Lett. 356, 79–83.
M. Shiraishi, T. Takenobu, and M. Ata (2003). Chem. Phys. Lett. 367, 633–636.
H. Kajiura, S. Tsutsui, K. Kadono, M. Kakuta, M. Ata, and Y. Murakami (2003). Appl. Phys. Lett. 82, 1105–1107.
C. Li, J. Li, F. Wu, S. S. Li, J. B. Xia, and L. W. Wang (2011). J. Phys. Chem. C 115, 23221–23225.
J. Li, Z. Hu, and G. Yang (2012). Chem. Phys. 392, 16–20.
H. Y. Wu, X. Fan, J. L. Kuo, and W. Q. Deng (2011). J. Phys. Chem. C 115, 9241–9249.
Y. Wang, X. Li, F. Wang, B. Xu, J. Zhang, Q. Sun, and Y. Jia (2013). Chem. Phys. 415, 26–30.
Y. S. Wang, Y. Ji, M. Li, P. F. Yuan, Q. Sun, Y. Jia (2011). J. Appl. Phys. 110, 094311/1-6.
A. K. Srivastava and N. Misra (2015). Mol. Simu. 42, 208–214.
A. D. Becke (1993). J. Chem. Phys. 98, 5648–5652.
A. Lee, W. Yang, and R. G. Parr (1988). Phys. Rev. B 37, 785–789.
Y. Zhao and D. G. Truhlar (2006). J. Chem. Phys. 125, 194101.
Y. Zhao and D. G. Truhlar (2008). Theor. Chem. Acc. 120, 215.
S. H. Mehdi, R. M. Ghalib, S. Awasthi, S. F. Alshahateet, R. Hashim, O. Sulaiman, and S. K. Pandey (2017). ChemistrySelect 2, 1–13.
S. K. Pandey, M. F. Khan, S. Awasthi, R. Sangwan, and S. Jain (2017). Aus. J. Chem. 70, 328–337.
F. Bawa and I. Pavas (2001). Phys. Chem. Chem. Phys. 3, 3042–3047.
J. Frisch, G. W. Trucks, H. B. Schlegel, G. E. Scuseria, M. A. Robb, J. R. Cheeseman, G. Scalmani, V. Barone, B. Mennucci, G. A. Petersson, H. Nakatsuji, M. Caricato, X. Li, H. P. Hratchian, A. F. Izmaylov, J. Bloino, G. Zheng, J. L. Sonnenberg, M. Hada, M. Ehara, K. Toyota, R. Fukuda, J. Hasegawa, M. Ishida, T. Nakajima, Y. Honda, O. Kitao, H. Nakai, T. Vreven, J. A. Montgomery Jr., J. E. Peralta, F. Ogliaro, M. Bearpark, J. J. Heyd, E. Brothers, K. N. Kudin, V. N. Staroverov, R. Kobayashi, J. Normand, K. Raghavachari, A. Rendell, J. C. Burant, S. S. Iyengar, J. Tomasi, M. Cossi, N. Rega, J. M. Millam, M. Klene, J. E. Knox, J. B. Cross, V. Bakken, C. Adamo, J. Jaramillo, R. Gomperts, R. E. Stratmann, O. Yazyev, A. J. Austin, R. Cammi, C. Pomelli, J. W. Ochterski, R. L. Martin, K. Morokuma, V. G. Zakrzewski, G. A. Voth, P. Salvador, J. J. Dannenberg, S. Dapprich, A. D. Daniels, Ö. Farkas, J. B. Foresman, J. V. Ortiz, J. Cioslowski, and D. J. Fox Gaussian 09, Rev B.01 (Gaussian Inc, Wallingford, 2010).
K. P. Huber and G. Herzberg Molecular Spectra and Molecular Structure, IV. Constants of Diatomic Molecules (van Nostrand Reinhold, New York, 1979).
A. K. Srivastava and N. Misra (1047). Comput. Theor. Chem. 2014, 1–5.
E. Kadossov and U. Burghaus (2008). J. Phys. Chem. C 112, 7390–7400.
M. Samadizadeh, A. A. Peyghan, and S. F. Rastegar (2016). Main Group Chem. 15, 107–116.
M. Nayebzadeh, A. A. Peyghan, and H. Soleymanabadi (2014). Physica E 62, 48–54.
J. Kakemam and A. A. Peyghan (2013). Comput. Mat. Sci. 79, 352–355.
R. Shinde and M. Tayade (2014). J. Phys. Chem. C 118, 17200–17204.
Acknowledgements
A. K. Srivastava acknowledges Council of Scientific and Industrial Research (CSIR), New Delhi, India for a research fellowship [Grant No. 09/107(0359)/2012-EMR-I]. The authors acknowledge the Department of Science and Technology, Government of India, New Delhi, for providing the Computational Facilities in the Department of Chemistry, Indian Institute of Technology Kanpur, India. We thank to the reviewers for their valuable comments and suggestions for improving the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Srivastava, A.K., Misra, N. & Pandey, S.K. DFT Study on Planar (CaO) n Rings (n = 1–5) and Their Hydrogen Storage Behavior: Ca–O Versus Mg–O Clusters. J Clust Sci 29, 57–65 (2018). https://doi.org/10.1007/s10876-017-1306-x
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10876-017-1306-x