Abstract
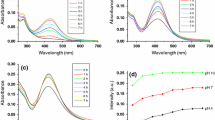
We report a facile, cost effective, and environmentally friendly green chemistry method for preparing silver nanoparticles (AgNPs) using Rubus crataegifolius bge (RCB) fruit extract. The amount of the fruit extract used was found to be important parameters in the growth of AgNPs. In this study, the effect of RCB fruit extract on the synthesis of AgNPs was studied using UV–Vis spectroscopy, transmission electron microscopy, scanning electron microscopy, energy-dispersive X-ray spectroscopy, X-ray diffraction (XRD), and dynamic light scattering analyses were performed to characterize the RCB fruit extract-stabilized AgNPs. The formation of the AgNPs was confirmed by the color change of the reaction medium and the absorbance peak observed at 420 nm. The XRD analysis confirmed the face centered cubic structure of the AgNPs. The catalytic property of the as-synthesized AgNPs was analyzed for the reduction of 4-nitrophenol to 4-aminophenol.






Similar content being viewed by others
References
A. Dawson and P. V. Kamat (2001). J. Phys. Chem. B 105, 960–966.
P. K. Jain, X. Huang, I. H. El-Sayed, T. Balakrishna, and R. Ramanibai (2008). Acc. Chem. Res. 41, 1578–1586.
G. Markovich, C. Patrick Collier, S. E. Henrichs, F. Remacle, R. D. Levine, and J. R. Heath (1999). Acc. Chem. Res 32, 415–423.
X. Du, J. He, J. Zhu, L. Sun, and S. An (2012). Appl. Surf. Sci. 258, 2717–2723.
P. Lui and M. Zhao (2009). Appl. Surf. Sci. 7, (255), 3989–3993.
R. R. Arvizo, S. Bhattacharyya, R. A. Kudgus, K. Giri, R. Bhattacharyya, and P. Mukherjee (2012). Chem. Soc. Rev. 41, 2943–2970.
C. N. R. Rao, H. S. S. R. Matte, R. Voggu, and A. Govindaraj (2012). Dalton Trans. 41, 5089–5120.
M. Rycenga, C. M. Cobley, J. Zeng, W. Li, C. H. Morgan, Q. Zhang, D. Qin, and Y. Xia (2011). Chem. Rev. 111, 3669–3721.
R. Jin, Y. Cao, C. Mirkin, K. Kelly, G. Schatz, and J. Zheng (2001). Science 294, 1901–1903.
R. Jin, Y. Cao, K. Kelly, G. Schatz, J. Zheng, and C. Mirkin (2003). Nature 425, 487–490.
Y. Sun (2007). Chem. Mater. 19, 5845–5847.
L. Jiang, S. Xu, J. Zhu, J. Zhang, J. Zhu, and H. Chen (2004). Inorg. Chem. 43, 5877–5883.
M. H. Kim, S. K. Kwak, S. H. Im, J. B. Lee, K. Y. Choi, and D. J. Byun (2014). J. Mater. Chem. C 2, 6165–6170.
R. S. Verma (2012). Curr. Opin. Chem. Eng. 1, 123–128.
A. A. Hebeish, M. H. El-Rafie, F. A. Abdel-Mohdy, E. S. Abdel-Halim, and H. E. Emam (2010). Carbohydr. Polym 82, 933–941.
B. Liu, X. Li, C. Zheng, X. Wang, and R. Sun (2013). Nanotechnology 24, 235601–235609.
A. R. Silva and G. Unali (2011). Nanotechnology 22, 315605–315610.
P. Raveendran, J. Fu, and S. L. Wallen (2003). J. Am. Chem. Soc. 125, 13940–13941.
V. Kathiravan, S. Ravi, and S. Ashokkumar (2014). Spectrochim Acta Part A 130, 116–121.
Y. Gao, Q. Huang, Q. Su, and R. Liu (2014). Spectrosc. Lett. 47, 790–795.
M. Zargar, K. Shameli, G. R. Najafi, and F. Farahani (2014). J. Ind. Eng. Chem. 20, 4169–4175.
A. Saxena, R. M. Tripathi, F. Zafar, and P. Singh (2012). Mater. Lett 67, 91–94.
M. S. Abdel-Aziz, M. S. Shaheen, A. A. El-Nekeety, and M. A. Abdel-wahhab (2014). J. Saudi Chem. Soc. 18, 356–363.
M. P. Patil, A. A. Rokade, D. Ngabire, and G. D. Kim (2016). J. Clust. Sci. 27, 1737–1750.
E. Murugan and J. N. Jebaranjitham (2012). J. Mol. Catal. A 365, 128–135.
Chinese Academy of Science China Flora Editorial Board Traditional Chinese Medicine Flora (Science Press, Beijing, 1981), p. 117.
K. M. Moon, J. E. Kim, H. Y. Kim, J. S. Lee, G. A. Son, S. W. Nam, B. W. Kim, and J. H. Lee (2011). J. Life Sci. 9, 1214.
J. Huang, S. Vongehr, S. Tang, H. Lu, and X. Meng (2010). J. Phys. Chem. C 114, 15005–150010.
H. Zhang, X. Li, and G. Chen (2009). J. Mater. Chem. 19, 8223–8231.
Y. Du, H. Chen, and N. Xu (2004). Appl. Catal. A. 277, 259–264.
Y. Chen, J. Qiu, X. Wang, and J. Siu (2006). J. Catal 242, 227–230.
Acknowledgements
This work was supported by the Technological R&D Program of SMBA (C0405639) and R&D Program of Ministry of Trade, Industry and Energy (10049521).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rokade, A.A., Kim, J.H., Lim, S.R. et al. A Novel Green Synthesis of Silver Nanoparticles Using Rubus crataegifolius Bge Fruit Extract. J Clust Sci 28, 2017–2026 (2017). https://doi.org/10.1007/s10876-017-1196-y
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10876-017-1196-y