Abstract
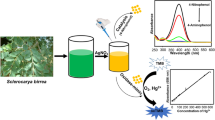
In the present study, we have synthesized glutathione modified silver nanoparticles (GSH-AgNPs) in aqueous medium and are characterized by absorption, high resolution transmission electron microscope (HR-TEM), selective area electron diffraction (SAED) pattern, dynamic light scattering (DLS), Zeta potential and Fourier transform infrared (FT-IR) spectroscopic measurements. Catalytic activity of GSH-AgNPs has been evaluated for the reduction reactions of 4-nitrophenol (4-NP), methylene blue (MB dye) and eosin Y (EY dye) in presence of sodium borohydride including the effect of catalyst concentration on the catalytic activity. Furthermore, the rate constants of reduction reactions are determined, which are linearly enhanced upon increasing the concentrations of GSH-AgNPs. It is explored that reduction reactions such as 4-NP, organic dyes by NaBH4 in the presence of catalyst follow a pseudo first order kinetics. The catalytic reduction of 4-NP and organic dyes proceed with a faster rate even in the presence of nanomolar concentration of catalyst.










Similar content being viewed by others
References
T. A. Wertime (1964). Science 146, 1257.
T. A. Wertime (1973). Science 182, 875.
L. M. Liz-Marzan (2006). Langmuir 22, 32.
D. D. Evanoff and G. Chumanov (2005). Chem. Phys. Chem. 6, 1221.
N. Okada, Y. Hamanaka, A. Nakamura, I. Pastoriza-Santos, and L. M. Liz-Marzan (2004). J. Phys. Chem. B. 108, 8751.
J. C. Riboh, A. J. Haes, A. D. McFarland, C. R. Yonzon, and R. P. VanDuyne (2003). J. Phys. Chem. B. 107, 1772.
Y. Shiraishi and N. Toshima (1999). J. Mol. Catal. A. 141, 187.
A. Dobosz and A. Sobczynski (2003). Water Res. 37, 1489.
L. Bhavani Devi and A. B. Manda (2013). RSC Adv. 3, 5238.
R. Fenger, E. Fertitta, H. Kirmse, A. F. Theunemann, and K. Rademann (2012). Phys. Chem. Chem. Phys. 14, 9343.
P. Zhang, C. Shao, Z. Zhang, M. Zhang, J. Mu, Z. Guo, and Y. Liu (2011). Nanoscale 3, 3357.
Z. Wang, S. Zhai, B. Zhai, Z. Xiao, F. Zhang, and Q. An (2014). New J. Chem. 38, 3999.
K. Esumi, R. Isono, and T. Yoshimura (2004). Langmuir 20, 237.
Y. Liu, H. Tsunoyama, H. Akita, S. Xie, and T. Tsukuda (2011). ACS Catal. 2, 1.
J. Cerda, G. Nuñez, H. Gómez, and L. Lópeza (2014). Mater. Sci. Eng C. 43, 21.
P. K. Khanna and V. V. V. S. Subbarao (2003). Mater. Lett 57, 2242.
Q. Xia, X. Chen, K. Zhao, and J. Liu (2008). Mater. Chem. Phys. 111, 8.
H. C. Yang, W. H. Wang, K. S. Huang, and M. H. Hon (2010). Carbohydr. Polym. 79, 176.
Z. Zaheer, M. A. Malik, F. M. Al-Nowaiser, and Z. Khan (2010). Coll. Surf. B. 81, (2), 587.
A. Rafey, K. B. L. Shrivastavaa, S. A. Iqbal, and Z. Khan (2011). J. Coll. Interface Sci. 354, 190.
Z. Khan, S. A. AL-Thabaiti, A. Y. Obaid, Z. A. Khan, A. O. Abdulrahman, and J. A. Youbi (2012). Colloid. Interface Sci. 367, 101.
I. S. Lim, D. Mott, W. Ip, P. N. Njoki, Y. Pan, S. Q. Zhou, and C. J. Zhong (2008). Langmuir 24, 8857.
H. Li, Z. Cui, and C. Han (2009). Sens. Actuators B. 143, 87.
B. Marco and B. Thomas (2005). Langmuir 21, 1354.
B. Marco and B. Thomas (2005). J. Phys. Chem. B. 109, 10243.
B. Marco and B. Thomas (2005). J. Phys. Chem. B. 109, 22476.
X. S. Kou, S. Z. Zhang, Z. Yang, C. K. Tsung, G. D. Studky, L. D. Sun, J. F. Wang, and C. H. Yan (2007). J. Am. Chem. Soc. 129, 6402.
J. M. Slocik and D. W. Wright (2003). Biomacromolecules 4, 1135.
C. B. Chory, C. Remenyi, H. Strohm, and G. Muller (2004). J. Phys. Chem. B. 108, 7637.
Y. G. Zheng, Z. C. Yang, and J. Y. Ying (2007). Adv. Mater. 19, 1475.
Y. T. Woo and D. Y. Lai Aromatic Amino And Nitro-Amino Compounds And Their Halogenated Derivatives Patty Toxicology (Wiley, New York, 2001).
S. C. Mitchell and R. H. Waring Ullmann’s Encyclopedia Of Industrial Chemistry (Wiley-VCH, Weinheim, 2002).
S. Panigrahi, S. Basu, S. Praharaj, S. Pande, S. Jana, A. Pal, S. K. Ghosh, and T. Pal (2007). J. Phys. Chem. C 111, 4596.
P. Zhao, X. Feng, D. Huang, G. Yang, and D. Astru (2015). Coord. Chem. Rev. 287, 114.
B. Bagchi, P. Thakur, A. Kool, S. Das, and P. Nandy (2014). RSC Adv. 4, 61114.
I. A. Alaton and I. A. Balcioglu (2001). J. Photochem. Photobiol. 141, 247.
F. Faisal, M. A. Tariq, and M. Muneer (2007). Dyes Pigm. 72, 233.
K. T. Chung and C. E. Cerniglia (1992). Mutat. Res. 277, 201.
S. Srivaji, R. Sinha, and D. Roy (2004). Aquat. Toxicol. 66, 319.
N. Vasimalai and S. A. John (2013). Talanta 115, 24.
J. S. Lee, M. S. Han, and C. A. Mirkin (2007). Angew. Chem. Int. Ed. 46, 4093.
A. S. Sharma and M. Ilanchelian (2014). Photochem. Photobiol. Sci. 13, 1741.
P. K. Sudeep, S. T. S. Joseph, and K. G. Thomas (2005). J. Am. Chem. Soc. 127, 6516.
A. Chompoosor, G. Han, and V. M. Rotello (2008). Bioconjugate Chem. 19, 1342.
Y. Luo, H. Miao, and X. Yank (2015). Talanta 144, 488.
R. T. Tom, V. Suryanarayanan, P. G. Reddy, S. Baskaran, and T. Pradeep (2004). Langmuir 20, 1909.
N. Nishida, E. S. Shibu, H. Yao, T. Oonishi, K. Kimura, and T. Pradeep (2008). Adv. Mater. 20, 4719.
N. Vasimalai and S. A. John (2013). J. Mater. Chem. A. 1, 4475.
J. Liu, G. Qin, P. Raveendran, and Y. Ikushima (2006). Chem. Eur. J. 12, 2131.
L. Ai, H. Yue, and J. Jiang (2012). J. Mater. Chem. 22, 23447.
A. Murugadoss and A. Chattopadhyay (2008). Nanotechnology 19, 1.
B. Baruah, G. J. Gabriel, M. J. Akbashev, and M. E. Booher (2013). Langmuir 29, 4225.
R. Kaur, C. Giordano, M. Gradzielski, and S. Mehta (2014). Chem. Asian J. 9, 189.
M. Zhu, C. Wang, D. Meng, and G. Diao (2013). J. Mater. Chem. A. 1, 2118.
S. Joseph and B. Mathew (2015). J. Mol. Liq. 204, 184.
Z. J. Jiang, C. Y. Liu, and L. W. Sun (2005). J. Phys. Chem. B. 109, 1730.
V. S. Suvith and D. Philip (2014). Spectrochimica Acta Part A. 118, 526.
V. K. Vidhu and D. Philip (2014). Micron 56, 54.
J. Santhanalakshmi and P. Venkatesan (2011). J. Nanopart. Res. 13, 479.
T. Pal, S. De, N. R. Jana, N. Pradhan, R. Mandal, A. Pal, A. E. Beezer, and J. C. Mitchel (1998). Langmuir 14, 4724.
S. Ghosh, T. Kundu, S. Mandal, and M. Pal (2002). Langmuir 18, 8756.
Acknowledgments
The authors R. R and M. I gratefully acknowledge the University Grants Commission (UGC-MRP, Project No. 41-309/2012 (SR)) and Department of Science and Technology (DST-SERB, Project No. SR/FT/CS-015/2009), New Delhi, India for the financial support. K. S acknowledges the DST-Inspire fellowship (SRF), Goverment of India for Financial support (IF110497).
Author information
Authors and Affiliations
Corresponding author
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Rajamanikandan, R., Shanmugaraj, K. & Ilanchelian, M. Concentration Dependent Catalytic Activity of Glutathione Coated Silver Nanoparticles for the Reduction of 4-Nitrophenol and Organic Dyes. J Clust Sci 28, 1009–1023 (2017). https://doi.org/10.1007/s10876-016-1095-7
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10876-016-1095-7