Abstract
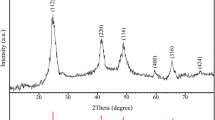
In the present study, ZnIn2S4 quantum dot was successfully synthesized via a simple hydrothermal method by employing Zn(OAc)2, InCl3, and thioglycolic acid (TGA) as the starting reagents. TGA was used as both the sulfur source and the capping agent. The obtained ZnIn2S4 quantum dot was characterized by XRD, SEM, TEM, EDS, Pl and DRS. The ZnIn2S4 quantum dot exhibited enhanced photocatalytic activity in the degradation of methylene blue under visible light irradiation. Furthermore, to examine the solar cell application of as-synthesized ZnIn2S4 quantum dot, FTO/TiO2/ZIS-QDs/N719/Pt-FTO structure was created by deposited ZnIn2S4 film on top of the TiO2 layer by Doctor’s blade method.







Similar content being viewed by others
References
C. B. Murray, D. J. Norris, and M. G. Bawendi (1993). J. Am. Chem. Soc. 115, 8706.
L. D. Trizio, M. Prato, A. Genovese, A. Casu, M. Povia, R. Simonutti, M. J. P. Alcocer, C. D’Andrea, F. Tassone, and L. Manna (2012). Chem. Mater. 24, 2400.
L. Q. Yin, Y. Bai, J. Zhou, J. Cao, X. W. Sun, and J. H. Zhang (2015). Opt. Mater. 42, 187.
W. Chung, K. Park, H. Yu, J. Kim, B. Chun, and S. Kim (2010). Opt. Mater. 32, 515.
M. G. Panthani, V. Akhavan, B. Goodfellow, J. P. Schmidtke, L. Dunn, A. Dodabalapur, P. F. Barbara, and B. A. Korgel (2008). J. Am. Chem. Soc. 49, 16770.
M. Nanu, J. Schoonman, and A. Goossens (2005). Nano Lett. 5, 1716.
R. C. Somers, M. G. Bawendi, and D. G. Nocera (2007). Chem. Soc. Rev. 36, 579.
I. L. Medintz, M. H. Stewart, S. A. Trammell, K. Susumu, J. B. Delehanty, B. C. Mei, J. S. Melinger, J. B. Blanco-Canosa, P. E. Dawson, and H. Mattoussi (2010). Nat. Mater. 9, 676.
X. Michalet, F. F. Pinaud, L. A. Bentolila, J. M. Tsay, S. Doose, J. J. Li, G. Sundaresan, A. M. Wu, S. S. Gambhir, and S. Weiss (2005). Science 307, 538.
L. Li, T. J. Daou, I. Texier, T. K. C. Tran, Q. L. Nguyen, and P. Reiss (2009). Chem. Mater. 21, 2422.
M. Mousavi-Kamazani, M. Salavati-Niasari, S. M. Hosseinpour-Mashkani, and M. Goudarzi (2015). Mater. Lett. 145, 99.
P. K. Santra and P. V. Kamat (2012). J. Am. Chem. Soc. 134, 2508.
Y. K. Lai, Z. Q. Lin, D. J. Zheng, L. F. Chi, R. G. Du, and C. J. Lin (2012). Electrochim. Acta 79, 175.
Y. Y. Yang, Q. X. Zhang, T. Z. Wang, L. F. Zhu, X. M. Huang, Y. D. Zhang, X. Hu, D. M. Li, Y. H. Luo, and Q. B. Meng (2012). Electrochim. Acta 88, 44.
N. Balis, V. Dracopoulos, K. Bourikas, and P. Lianos (2013). Electrochim. Acta 91, 246.
H. H. Yang, W. G. Fan, A. Vaneski, A. S. Susha, W. Y. Teoh, and A. L. Rogach (2012). Adv. Funct. Mater. 22, 2821.
M. Mousavi-Kamazani, M. Salavati-Niasari, and M. Sadeghinia (2015). Mater. Lett. 142, 145.
Z. Lei, W. You, M. Liu, G. Zhou, T. Takata, M. Hara, K. Domen, and C. Li (2003). Chem. Commun. 17, 2142.
Y. Yu, G. Chen, G. Wang, and Z. Lv (2013). Int. J. Hydrog. Energy 38, 1278.
T. X. Wang, S. H. Xu, and F. X. Yang (2012). Mater. Lett. 83, 46.
Z. Chen, D. Li, W. Zhang, C. Chen, W. Li, M. Sun, Y. He, and X. Fu (2008). J. Inorg. Chem. 47, 9766.
W. Cai, Y. Zhao, J. Hu, J. Zhong, and W. Xian (2011). J. Mater. Sci. Technol. 27, 559.
B. Xuefeng and L. Jinshu (2011). Mater. Res. Bull. 46, 1028.
M. Salavati-Niasari, M. Ranjbar, and M. Sabet (2013). J. Inorg. Organomet. Polym. 23, 452.
F. Fang, L. Chen, Y. Biao, and L. Mu (2010). J. Phys. Chem. C 114, 2393.
X. Gou, F. Cheng, Y. Shi, L. Zhang, S. Peng, J. Chen, and P. Shen (2007). J. Cryst. Growth Des. 7, 2444.
S. Shen, L. Zhao, and L. Guo (2009). J. Mater. Res. Bull. 44, 100.
X. Hu, J. Yu, J. Gong, and Q. Li (2007). J. Cryst. Growth Des. 12, 8.
P. Tang, H. Chen, and F. Cao (2012). Mater. Lett. 68, 171.
M. Panahi-Kalamuei, M. Mousavi-Kamazani, M. Salavati-Niasari, and S. M. Hosseinpour-Mashkani (2015). Ultrason. Sonochem. 23, 246.
F. Guo, J. He, J. Li, W. Wu, Y. Hang, and J. Hua (2013). J. Colloid Interface Sci. 408, 59.
W. H. Yuan, Z. L. Xia, and L. Li (2013). Chin. Chem. Lett. 24, 984.
Acknowledgments
This work was supported by the Chemistry Research Center at Islamic Azad University Birjand.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ebadi, M., Ramezani, M. & Zarghami, Z. A Facile Hydrothermal Route to the Synthesis of ZnIn2S4 Quantum Dots in the Presence of Thioglycolic Acid and Investigation Its Light Harvesting Application. J Clust Sci 27, 341–350 (2016). https://doi.org/10.1007/s10876-015-0933-3
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10876-015-0933-3