Abstract
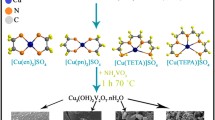
Applying of the most toxic halogenated and aromatic flame retardants is limited with respect to the environmental requirements. Nontoxic Al(OH)3 nanoparticles were synthesized via a simple surfactant-free precipitation reaction at room temperature. The effect of various precipitation-agents on the morphology of the products was investigated. Al(OH)3 nanoparticles were added to the polysulfone and poly styrene (PS) matrices. Electron microscope images show excellent dispersion of aluminium hydroxide in PS matrix. Nanoparticles appropriately enhanced both thermal stability and flame retardant property of the polymeric matrices. The enhancement of flame retardancy is due to endothermic decomposition of Al(OH)3 that absorbs heat and simultaneously releases of water (makes combustible gases diluted and cold). Dispersed nanoparticles play the role of a barrier layer against flame, oxygen and polymer volatilization. Al(OH)3 was converted to Al2O3 and its photo-catalyst property in degradation three different organic dyes as pollutants was investigated.













Similar content being viewed by others
References
F. Laoutid, L. Bonnaud, M. Alexandre, J. Lopez-Cuesta, and Ph. Dubois (2009). Mater. Sci. Eng. R 63, 100.
D. Ghanbari, M. Salavati-Niasari, and M. Sabet (2013). Compos. B 45, 550.
A. B. Morgan and C. A. Wilkie Flame Retardant Polymer Nanocomposite, Chapter 1 (Wiley, New Jersey, 2007).
B. Y. Geng, J. Z. Ma, and J. H. You (2008). Cryst. Growth Des. 8, 1443.
D. Ghanbari, M. Salavati-Niasari, and M. Sabet (2012). J. Clust. Sci. 23, 1081.
B. Jia and L. Gao (2008). J. Phys. Chem. C 112, 666.
D. Ghanbari, M. Salavati-Niasari, and M. Ghasemi-Kooch (2014). J. Ind. Eng. Chem. 20, 3970.
X. Sun, C. Zheng, F. Zhang, Y. Yang, G. Wu, A. Yu, and N. Guan (2009). J. Phys. Chem. C 113, 16002.
S. Liu, J. Ying, X. Zhou, and X. Xie (2009). Mater. Lett. 63, 911.
F. Gholamian, M. Salavati-Niasari, D. Ghanbari, and M. Sabet (2013). J. Clust. Sci. 24, 73.
H. Wang, P. Fang, Z. Chen, and S. Wang (2007). Appl. Surf. Sci. 253, 8495.
P. Jamshidi, M. Salavati-Niasari, D. Ghanbari, and H. R. Shams (2013). J. Clust. Sci. 24, 1151.
F. Gholamian, G. Nabiyouni, D. Ghanbari, R. Jalajerdi, and A. Aminifazl (2013). High Temp. Mater. Proc. 32, 125.
W. Q. Cai, J. G. Yu, and M. Jaroniec (2010). J. Mater. Chem. 20, 4587.
N. K. Renuka, A. V. Shijina, and A. K. Praveen (2012). Mater. Lett. 82, 42.
Y. K. Park, E. H. Tadd, M. Zubris, and R. Tannenbaum (2005). Mater. Res. Bull. 40, 1506.
F. Yaripour, Z. Shariatinia, S. Shariatinia, and A. Irandoukht (2015). Fuel 139, 40.
A. A. Pechenkin, S. D. Badmaev, and V. D. Belyaev (2015). Appl. Catal. B 166, 535.
T. K. Ghosh and N. N. Nair (2015). Surf. Sci. 632, 20.
S. Y. Hosseini and M. R. K. Nikou (2014). J. Ind. Eng. Chem. 20, 4421.
P. Manivasakan, A. Karthik, and V. Rajendran (2013). Powder Technol. 234, 84.
X. Wu, B. Zhang, and Z. Hu (2013). Powder Technol. 239, 155.
P. Jamshidi, D. Ghanbari, and M. Salavati-Niasari (2014). J. Ind. Eng. Chem. 20, 350.
M. Yousefi, E. Noori, D. Ghanbari, M. Salavati-Niasari, and T. Gholami (2014). J. Clust. Sci. 25, 397.
Acknowledgments
Authors are grateful to council of University of Kashan for providing financial support to undertake this work by Grant No (159271/295).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Goudarzi, M., Ghanbari, D., Salavati-Niasari, M. et al. Synthesis and Characterization of Al(OH)3, Al2O3 Nanoparticles and Polymeric Nanocomposites. J Clust Sci 27, 25–38 (2016). https://doi.org/10.1007/s10876-015-0895-5
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10876-015-0895-5