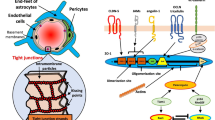
Abstract
We present a case study of a young male with a history of 22q11.2 deletion syndrome (22qDS), diagnosed with systemic capillary leak syndrome (SCLS) who presented with acute onset of diffuse anasarca and sub-comatose obtundation. We hypothesized that his co-presentation of neurological sequelae might be due to blood-brain barrier (BBB) susceptibility conferred by the 22q11.2 deletion, a phenotype that we have previously identified in 22qDS. Using pre- and post-intravenous immunoglobulins (IVIG) patient serum, we studied circulating biomarkers of inflammation and assessed the potential susceptibility of the 22qDS BBB. We employed in vitro cultures of differentiated BBB-like endothelial cells derived from a 22qDS patient and a healthy control. We found evidence of peripheral inflammation and increased serum lipopolysaccharide (LPS) alongside endothelial cells in circulation. We report that the patient’s serum significantly impairs barrier function of the 22qDS BBB compared to control. Only two other cases of pediatric SCLS with neurologic symptoms have been reported, and genetic risk factors have been suggested in both instances. As the third case to be reported, our findings are consistent with the hypothesis that genetic susceptibility of the BBB conferred by genes such as claudin-5 deleted in the 22q11.2 region promoted neurologic involvement during SCLS in this patient.


Similar content being viewed by others
Data Availability
The raw data supporting the conclusions of this study will be made available by the authors, without undue reservation, to any qualified researcher.
References
Gur RE, Bassett AS, McDonald-McGinn DM, Bearden CE, Chow E, Emanuel BS, et al. A neurogenetic model for the study of schizophrenia spectrum disorders: the International 22q11.2 deletion syndrome brain Behavior Consortium. Mol Psychiatry. 2017;22(12):1664–72.
Sullivan KE. The clinical, immunological, and molecular spectrum of chromosome 22q11.2 deletion syndrome and DiGeorge syndrome. Curr Opin Allergy Clin Immunol. 2004;4(6):505–12.
Tang SX, Yi JJ, Calkins ME, Whinna DA, Kohler CG, Souders MC, et al. Psychiatric disorders in 22q11.2 deletion syndrome are prevalent but undertreated. Psychol Med. 2014;44(6):1267–77.
McDonald-McGinn DM, Sullivan KE, Marino B, Philip N, Swillen A, Vorstman JA, et al. 22q11.2 deletion syndrome. Nat Rev Dis Primers. 2015;1:15071.
Arinami T. Analyses of the associations between the genes of 22q11 deletion syndrome and schizophrenia. J Hum Genet. 2006;51(12):1037–45.
Cioffi S, Martucciello S, Fulcoli FG, Bilio M, Ferrentino R, Nusco E, et al. Tbx1 regulates brain vascularization. Hum Mol Genet. 2014;23(1):78–89.
Karayiorgou M, Gogos JA. The molecular genetics of the 22q11-associated schizophrenia. Brain Res Mol Brain Res. 2004;132(2):95–104.
Greene C, Hanley N, Campbell M. Claudin-5: gatekeeper of neurological function. Fluids Barriers CNS. 2019;16(1):3.
Abbott NJ. Inflammatory mediators and modulation of blood-brain barrier permeability. Cell Mol Neurobiol. 2000;20(2):131–47.
Cheslow L, Alvarez JI. Glial-endothelial crosstalk regulates blood-brain barrier function. Curr Opin Pharmacol. 2016;26:39–46.
Crockett AM, Ryan SK, Vásquez AH, Canning C, Kanyuch N, Kebir H et al. Disruption of the blood-brain barrier in 22q11.2 deletion syndrome. Brain. 2021.
Guo Y, Singh LN, Zhu Y, Gur RE, Resnick A, Anderson SA et al. Association of a functional Claudin-5 variant with schizophrenia in female patients with the 22q11.2 deletion syndrome. Schizophr Res. 2019.
Li Y, Xia Y, Zhu H, Luu E, Huang G, Sun Y et al. Investigation of neurodevelopmental deficits of 22 q11.2 deletion syndrome with a Patient-iPSC-Derived blood-brain barrier model. Cells. 2021;10(10).
Alvarez JI, Saint-Laurent O, Godschalk A, Terouz S, Briels C, Larouche S, et al. Focal disturbances in the blood-brain barrier are associated with formation of neuroinflammatory lesions. Neurobiol Dis. 2015;74:14–24.
Greene C, Kealy J, Humphries MM, Gong Y, Hou J, Hudson N, et al. Dose-dependent expression of claudin-5 is a modifying factor in schizophrenia. Mol Psychiatry. 2018;23(11):2156–66.
Druey KM, Parikh SM. Idiopathic systemic capillary leak syndrome (Clarkson disease). J Allergy Clin Immunol. 2017;140(3):663–70.
Bozzini MA, Milani GP, Bianchetti MG, Fossali EF, Lava SAG. Idiopathic systemic capillary leak syndrome (Clarkson syndrome) in childhood: systematic literature review. Eur J Pediatr. 2018;177(8):1149–54.
Hsu P, Xie Z, Frith K, Wong M, Kakakios A, Stone KD, et al. Idiopathic systemic capillary leak syndrome in children. Pediatrics. 2015;135(3):e730–5.
Günes AR, Berlit P, Weber R. Severe cerebral involvement due to idiopathic systemic capillary leak syndrome. Clin Case Rep. 2016;4(4):429–31.
Benallegue N, Kapoor R, Kebir H, Crockett AM, Li C, Cheslow L, et al. The hedgehog pathway suppresses neuropathogenesis in CD4 T cell-driven inflammation. Brain. 2021;144(6):1670–83.
Crockett AM, Kebir H, Benallegue N, Adelman P, Gur RE, Sullivan K, et al. Immune status of the murine 22q11.2 deletion syndrome model. Eur J Immunol. 2023;53(1):e2249840.
Neal EH, Marinelli NA, Shi Y, McClatchey PM, Balotin KM, Gullett DR, et al. A simplified, fully defined differentiation Scheme for producing blood-brain barrier endothelial cells from human iPSCs. Stem Cell Rep. 2019;12(6):1380–8.
Alvarez JI, Dodelet-Devillers A, Kebir H, Ifergan I, Fabre PJ, Terouz S, et al. The hedgehog pathway promotes blood-brain barrier integrity and CNS immune quiescence. Science. 2011;334(6063):1727–31.
Percik R, Nethanel A, Liel Y. Capillary-leak syndrome: an unrecognized early immune adverse effect of checkpoint-inhibitors treatment. Immunotherapy. 2021;13(8):653–9.
Nielsen MM, Witherden DA, Havran WL. γδ T cells in homeostasis and host defence of epithelial barrier tissues. Nat Rev Immunol. 2017;17(12):733–45.
Severance EG, Gressitt KL, Stallings CR, Origoni AE, Khushalani S, Leweke FM, et al. Discordant patterns of bacterial translocation markers and implications for innate immune imbalances in schizophrenia. Schizophr Res. 2013;148(1–3):130–7.
Simonin M, Corbeau P, Durand P, Rosenzwajg M, Filleron A, Tran TA. A possible role for IL-17 in Clarkson’s disease. Eur J Paediatr Neurol. 2016;20(6):953–6.
Sion-Sarid R, Lerman-Sagie T, Blumkin L, Ben-Ami D, Cohen I, Houri S. Neurologic involvement in a child with systemic capillary leak syndrome. Pediatrics. 2010;125(3):e687–92.
Profaci CP, Munji RN, Pulido RS, Daneman R. The blood-brain barrier in health and disease: important unanswered questions. J Exp Med. 2020;217(4).
Acknowledgements
We thank Dr. Amit Bar-Or’s lab for sharing access to their LSR Fortessa, as well as the University of Pennsylvania Cytomics and Cell Sorting Resource Laboratory for their maintenance of the machines. We appreciate the generosity of Dr. Mark Khan’s lab for sharing their ECIS measurement device. We thank the support of the Immune Dysregulation Program at CHOP and appreciate the doctors, nurses, and staff at CHOP who aided in caring for the patient described here and taking samples for study. Lastly, we want to thank the patient and his family, as well as the control donor.
Funding
The has supported this work by the following grants: Uytengsu-Hamilton 22q11 Neuropsychiatry Research Program of the Maternal and Child Health Research Institute (MCHRI) at Stanford University, grant UH22QEXTFY22-03 (JIA), NIH MH134797-01 (JIA), NIH MH110185-03 and MH066912 (SA), and Jeffrey Modell Foundation (NR). AMC held a scholarship from the Blavatnik Family Foundation. HK held a fellowship from the Multiple Sclerosis Society of Canada (MSSC).
Author information
Authors and Affiliations
Contributions
AMC and HK: Carried out the experiments. AMC and JIA: Drafted the initial manuscript. NR and SJ: Provided clinical care. SA: Provided reagents and supervision to iPSC assays. JIA: supervised the study. All authors: Revised the manuscript critically for important intellectual content.
Corresponding author
Ethics declarations
Ethics Approval and Consent to Participate
Consent from the next of kin was obtained for the case presented here under a human subject’s protocol approved by the Children’s Hospital of Philadelphia (CHOP).
Consent for Publication
The subject and his family were provided the opportunity to review the case report prior to submission and provided assent/consent for this work to be published.
Conflicts of Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Crockett, A.M., Kebir, H., Anderson, S.A. et al. 22q11.2 Deletion-Associated Blood-Brain Barrier Permeability Potentiates Systemic Capillary Leak Syndrome Neurologic Features. J Clin Immunol 44, 87 (2024). https://doi.org/10.1007/s10875-024-01686-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10875-024-01686-w