Abstract
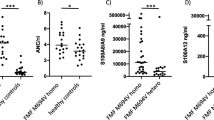
Syndrome of undifferentiated recurrent fever (SURF) is characterized by recurrent fevers, a lack of confirmed molecular diagnosis, and a complete or partial response to colchicine. Despite the clinical similarities to familial Mediterranean fever (FMF), the underlying inflammatory mechanisms of SURF are not yet understood. We here analyzed the in vitro activation of the pyrin inflammasome in a cohort of SURF patients compared to FMF and PFAPA patients. Peripheral blood mononuclear cells (PBMC) were collected from SURF (both colchicine-treated and untreated), FMF, PFAPA patients, and healthy donors. PBMC were stimulated ex vivo with Clostridium difficile toxin A (TcdA) and a PKC inhibitor (UCN-01), in the presence or absence of colchicine. The assembly of the pyrin inflammasome was evaluated by measuring the presence of apoptosis-associated Speck-like protein containing caspase recruitment domain (ASC) specks in monocytes using flow cytometry. IL-1β secretion was quantified using an ELISA assay. No differences in TcdA-induced activation of pyrin inflammasome were observed among FMF, PFAPA, and healthy donors. Untreated SURF patients showed a reduced response to TcdA, which was normalized after colchicine treatment. In contrast to FMF, SURF patients, similar to PFAPA patients and healthy donors, did not exhibit pyrin inflammasome activation in response to UCN-01-mediated pyrin dephosphorylation. These data demonstrate that in vitro functional analysis of pyrin inflammasome activation can differentiate SURF from FMF and PFAPA patients, suggesting the involvement of the pyrin inflammasome in the pathophysiology of SURF.




Similar content being viewed by others
Data Availability
Not applicable.
References
Russo RAG, Brogan PA. Monogenic autoinflammatory diseases. Rheumatology. 2014;53:1927–39. https://doi.org/10.1093/rheumatology/keu170.
Gattorno M, Hofer M, Federici S, Vanoni F, Bovis F, Aksentijevich I, et al. Classification criteria for autoinflammatory recurrent fevers. Ann Rheum Dis. 2019;78:1025–32. https://doi.org/10.1136/annrheumdis-2019-215048.
Papa R, Penco F, Volpi S, Sutera D, Caorsi R, Gattorno M. Syndrome of undifferentiated recurrent fever (SURF): an emerging group of autoinflammatory recurrent fevers. J Clin Med. 2021;10:1963. https://doi.org/10.3390/jcm10091963.
Luu I, Nation J, Page N, Carvalho D, Magit A, Jiang W, et al. Undifferentiated recurrent fevers in pediatrics are clinically distinct from PFAPA syndrome but retain an IL-1 signature. Clin Immunol. 2021;226:108697. https://doi.org/10.1016/j.clim.2021.108697.
Sutera D, Bustaffa M, Papa R, Matucci-Cerinic C, Matarese S, D’Orsi C, et al. Clinical characterization, long-term follow-up, and response to treatment of patients with syndrome of undifferentiated recurrent fever (SURF). Semin Arthritis Rheum. 2022;55:152024. https://doi.org/10.1016/j.semarthrit.2022.152024.
Batu ED, Basaran O, Bilginer Y, Ozen S. Familial Mediterranean fever: how to interpret genetic results? How to treat? A quarter of a century after the association with the Mefv gene. Curr Rheumatol Rep. 2022;24:206–12. https://doi.org/10.1007/s11926-022-01073-7.
Schnappauf O, Chae JJ, Kastner DL, Aksentijevich I. The pyrin inflammasome in health and disease. Front Immunol. 2019;10:1745. https://doi.org/10.3389/fimmu.2019.01745.
Jamilloux Y, Magnotti F, Belot A, Henry T. The pyrin inflammasome: from sensing RhoA GTPases-inhibiting toxins to triggering autoinflammatory syndromes. Pathog Dis. 2018;76. https://doi.org/10.1093/femspd/fty020.
Park YH, Wood G, Kastner DL, Chae JJ. Pyrin inflammasome activation and RhoA signaling in the autoinflammatory diseases FMF and HIDS. Nat Immunol. 2016;17:914–21. https://doi.org/10.1038/ni.3457.
Van Gorp H, Saavedra PHV, De Vasconcelos NM, Van Opdenbosch N, Vande Walle L, Matusiak M, et al. Familial Mediterranean fever mutations lift the obligatory requirement for microtubules in Pyrin inflammasome activation. Proc Natl Acad Sci. 2016;113:14384–9. https://doi.org/10.1038/ni.3457.
Magnotti F, Malsot T, Georgin-lavialle S, Abbas F, Martin A, Belot A, et al. Fast diagnostic test for familial Mediterranean fever based on a kinase inhibitor. Ann Rheum Dis. 2021;80:128–32. https://doi.org/10.1136/annrheumdis-2020-218366.
Magnotti F, Lefeuvre L, Benezech S, Malsot T, Waeckel L, Martin A, et al. Pyrin dephosphorylation is sufficient to trigger inflammasome activation in familial Mediterranean fever patients. EMBO Mol Med. 2019;11:e10547. https://doi.org/10.15252/emmm.201910547.
Moradian MM, Sarkisian T, Ajrapetyan H, Avanesian N. Genotype–phenotype studies in a large cohort of Armenian patients with familial Mediterranean fever suggest clinical disease with heterozygous MEFV mutations. J Hum Genet. 2010;55:389–93. https://doi.org/10.1038/jhg.2010.52.
Hoss F, Rolfes V, Davanso MR, Braga TT, Franklin BS. Detection of ASC speck formation by flow cytometry and chemical cross-linking. Methods Mol Biol. 2018;1714:149–65. https://doi.org/10.1007/978-1-4939-7519-8_10.
Papa R, Rusmini M, Volpi S, Caorsi R, Picco P, Grossi A, et al. Next generation sequencing panel in undifferentiated autoinflammatory diseases identifies patients with colchicine-responder recurrent fevers. Rheumatology (Oxford). 2020;59(2):344–60. https://doi.org/10.1093/rheumatology/kez270.
Van Gorp H, Huang L, Saavedra P, Vuylsteke M, Asaoka T, Prencipe G, et al. Blood-based test for diagnosis and functional subtyping of familial Mediterranean fever. Ann Rheum Dis. 2020;79:960–8. https://doi.org/10.1136/annrheumdis-2019-216701.
Manthiram K, Zhou Q, Aksentijevich I, Kastner DL. The monogenic autoinflammatory diseases define new pathways in human innate immunity and inflammation. Nat Immunol. 2017;18:832–42. https://doi.org/10.1038/ni.3777.
Ben-Zvi I, Herskovizh C, Kukuy O, Kassel Y, Grossman C, Livneh A. Familial Mediterranean fever without MEFV mutations: a case–control study. Orphanet J Rare Dis. 2015;10:34. https://doi.org/10.1186/s13023-015-0252-7.
Ozturk K, Cakan M. The analysis of genotype-phenotype correlation in familial Mediterranean fever. Pediatr Int. 2022;64:e15017. https://doi.org/10.1111/ped.15017.
Federici S, Calcagno G, Finetti M, Gallizzi R, Meini A, Vitale A, et al. Clinical impact of MEFV mutations in children with periodic fever in a prevalent western European Caucasian population. Ann Rheum Dis. 2012;71:1961–5. https://doi.org/10.1136/annrheumdis-2011-200977.
Ben-Chetrit E, Ozdogan H. Can we make a diagnosis of autoinflammatory diseases based upon clinical features only? Clin Exp Rheumatol. 2017;35(Suppl 108):16–8.
Dufies O, Boyer L. RhoGTPases and inflammasomes: guardians of effector-triggered immunity. PLoS Pathog. 2021;17:e1009504. https://doi.org/10.1371/journal.ppat.1009504.
Molad Y. Update on colchicine and its mechanism of action. Curr Rheumatol Rep. 2002;4:252–6. https://doi.org/10.1007/s11926-002-0073-2.
Gao W, Yang J, Liu W, Wang Y, Shao F. Site-specific phosphorylation and microtubule dynamics control Pyrin inflammasome activation. Proc Natl Acad Sci. 2016;113:E4857–66. https://doi.org/10.1073/pnas.1601700113.
Heilig R, Broz P. Function and mechanism of the pyrin inflammasome. Eur J Immunol. 2018;48:230–8. https://doi.org/10.1002/eji.201746947.
Gattorno M, Piccini A, Lasigliè D, Tassi S, Brisca G, Carta S, et al. The pattern of response to anti–interleukin-1 treatment distinguishes two subsets of patients with systemic-onset juvenile idiopathic arthritis. Arthritis Rheum. 2008;58:1505–15. https://doi.org/10.1002/art.23437.
Batu ED. Periodic fever, aphthous stomatitis, pharyngitis, and cervical adenitis (PFAPA) syndrome: main features and an algorithm for clinical practice. Rheumatol Int. 2019;39:957–70. https://doi.org/10.1007/s00296-019-04257-0.
Soriano A, Soriano M, Espinosa G, Manna R, Emmi G, Cantarini L, et al. Current therapeutic options for the main monogenic autoinflammatory diseases and PFAPA syndrome: evidence-based approach and proposal of a practical guide. Front Immunol. 2020;11:865. https://doi.org/10.3389/fimmu.2020.0086.
Funding
The study has been supported by Ricerca Corrente of the Italian Ministry of Health.
Author information
Authors and Affiliations
Contributions
IP, MG, and SP conceived the study. IP and MG were responsible for study supervision. SP and IP coordinated the study and acquired data. FP, AB, and AC performed cytokine assays. PB and CS participated in sample collection. MB, CM-C, RP, RC, ED, and SV participated in patient evaluation and selection. All authors have contributed in a substantial and intellectual manner to this manuscript; SP, IP, AR, and MG wrote the essential sections of the manuscript, which was critically revised by all authors.
Corresponding author
Ethics declarations
Ethical Approval
The study protocol was approved by the Ethical board of the G. Gaslini Institute in Genova (Italy).
Consent to Participate
Written informed consent was obtained from all participants included in the study. In case of minor patients, written informed consent was obtained from the parents.
Consent for Publication
Not applicable.
Competing Interests
MG has received consultant and speaker honoraria from Novartis and SOBI. All other authors declare they have no financial interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Palmeri, S., Penco, F., Bertoni, A. et al. Pyrin Inflammasome Activation Defines Colchicine-Responsive SURF Patients from FMF and Other Recurrent Fevers. J Clin Immunol 44, 49 (2024). https://doi.org/10.1007/s10875-023-01649-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10875-023-01649-7