Abstract
Purpose
Common variable immunodeficiency (CVID) is characterized by hypogammaglobulinemia and clinical manifestations such as infections, autoimmunity, and malignancy. We sought to determine if responsiveness to interleukin-21 (IL-21), a key cytokine for B cell differentiation, correlates with distinct clinical phenotypes in CVID.
Methods
CVID subjects were recruited through the Canadian Primary Immunodeficiency Evaluative Survey registry. Peripheral blood mononuclear cells were cultured with anti-CD40 ± interferon-gamma, interleukin-4 (IL-4), IL-21, and/or IL-4+IL-21. B cell subpopulations and IgG production were determined at baseline and day 7 by flow cytometry and ELISA. Clinical complications were compared using contingency tables.
Results
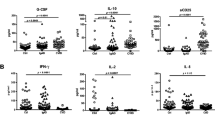
CVID subjects exhibited decreased CD27+ B cells and IgG production after 7 days of stimulation with anti-CD40+IL-21 (p < 0.05). In a subset of subjects [CVID responders (R)], the addition of IL-4 led to significant increases in CD27+ B cells and IgG (p < 0.05). In CVID non-responders (NR), CD27+ B cells and IgG remained lower despite the addition of IL-4. CVID NR experienced significantly more non-infectious clinical complications of CVID than R [OR 8.8, 95% confidence interval (CI) 1.6 to 48.13]. Previous studies reported that CVID subjects with ≤ 2% class-switched memory B cells were more at risk of these complications, but no significant association was found among this cohort of subjects [OR 3.5, CI 0.9 to 13.3]. In fact, 34.6% of CVID NR had > 2% class-switched memory B cells at baseline.
Conclusions
The IL-4 and IL-21 in vitro assays distinguish two groups of CVID subjects and can be used with baseline B cell subpopulation phenotyping to better identify patients experiencing more vs. fewer clinical non-infectious complications and potentially to modulate therapy.





Similar content being viewed by others
References
Gathmann B, Grimbacher B, Beauté J, Dudoit Y, Mahlaoui N, Fischer A, et al. The European internet-based patient and research database for primary immunodeficiencies: result 2006-2008. Clin Exp Immunol. 2009;157(Suppl 1):3–11.
Castigli E, Geha R. Molecular basis of common variable immunodeficiency. J Allergy Clin Immunol. 2006;117(4):740–6.
Kainulainen L, Vuorinen T, Rantakokko-Jalava K, Osterback R, Ruuskanen O. Reccurent and persistent respiratory tract viral infections in patients with primary hypogammaglobulinemia. J Allergy Clin Immunol. 2010;126:120–6.
Cunningham-Rundles C, Bodian C. Common variable immunodeficiency: clinical and immunological features of 248 patients. Clin Immunol. 1999;92(1):24–48.
Glocker E, Ehl S, Brimbacher B. Common variable immunodeficiency in children. Curr Opin Pediatr. 2007;19(6):685–92.
Park M, Li J, Hagan J, Maddox D, Abraham R. Common variable immunodeficiency: a new look to an old disease. Lancet. 2008;372(9637):489–502.
Ahn S, Cunningham-Rundles C. Role of B cells in common variable immune deficiency. Expert Rev Clin Immunol. 2009;5:557–64.
Maffucci P, Filion CA, Boisson B, Itan Y, Shang L, Casanova J-L, et al. Genetic diagnosis using whole exome sequencing in common variable immunodeficiency. Front Immunol. 2016;7:220.
Wehr C, Kivioja T, Schmitt C, Ferry B, Witte T, Eren E, et al. The EUROclass trial: defining subgroups in common variable immunodeficiency. Blood. 2008;111(1):77–85.
Warnatz K, Denz A, Drager R, Braun M, Groth C, Wolff-Vorbeck G, et al. Severe deficiency of switched memory B cells (CD27(+)IgM(−)IgD(−)) in subgroups of patients with common variable immunodeficiency: a new approach to classify a heterogeneous disease. Blood. 2002;99(5):1544–51.
Piqueras B, Lavenu-Bombled C, Galicier L, Bergeron-van der Cruyssen F, Mouthon L, Chevret S, et al. Common variable immunodeficiency patient classification based on impaired B cell memory differentiation correlates with clinical aspects. J Clin Immunol. 2003;23(5):385–400.
Avery DT, Deenick EK, Ma CS, Suryani S, Simpson N, Chew GY, et al. B cell-intrinsic signaling through IL-21 receptor and STAT3 is required for establishing long-lived antibody responses in humans. J Exp Med. 2010;207(1):155–71.
Zotos D, Coquet JM, Zhang Y, Light A, D'Costa K, Kallies A, et al. IL-21 regulates germinal center B cell differentiation and proliferation through a B cell-intrinsic mechanism. J Exp Med. 2010;207(2):365–78.
Desjardins M, Mazer B. B-cell memory and primary immune defiencies: interleukin-21 related defects. Curr Opin Allergy Clin Immunol. 2013;13(6):639–45.
O’Keefe A, Halbrich M. Primary immunodeficiency for the primary care provider. Paediatr Child Health. 2016;21(2):e10–e4.
McCusker C, Warrington R. Primary immunodeficiency. Allergy Asthma Clin Immunol. 2011;7(Suppl 1):S11.
Conley M, Notarangelo L, Etzioni A. Diagnostic criteria for primary immunodeficiencies. Representing PAGID (pan-American group for immunodeficiency) and ESID (European society for immunodeficiencies). Clin Immunol. 1999;93(3):190–7.
Al Rushood M, McCusker C, Mazer B, Alizadehfar R, Grimbacher B, Depner M, et al. Autosomal dominant cases of chronic mucocutaneous candidiasis segregates with mutations of signal transducer and activator of transcription 1, but not of Toll-like receptor 3. J Pediatr. 2013;163(1):277–9.
Halbrich M, Ben-Shoshan M, McCusker C. Autoimmune hemolytic anemia in a teenager: a wolf in sheep’s clothing. Eur J Haematol. 2013;91(3):262–4.
Gathmann B, Mahlaoui N, Gérard L, Oksenhendler E, Warnatz K, Schulze I, et al. Clinical picture and treatment of 2212 patients with common variable immunodeficiency. J Allergy Clin Immunol. 2014;134:116–26.
Resnick E, Moshier E, Godbold J, Cunningham-Rundles C. Morbidity and mortality in common variable immune deficiency over 4 decades. Blood. 2012;119:1650–7.
Srinivasa B, Alizadehfar R, Desrosiers M, Shuster J, Pant Pai N, Tsoukas C. Adult primary immune deficiency: what are we missing? Am J Med. 2012;125(8):779–86.
Hajoui O, Zheng H, Guay J, Letuve S, Fawaz L, Mazer B. Regulation of IL-13 receptor alpha 1 expression and signaling on human tonsillar B-lymphocyte subsets. J Allergy Clin Immunol. 2007;120(6):1425–32.
Hajoui O, Janani R, Tulic M, Joubert P, Ronis T, Hamid Q, et al. Synthesis of IL-13 by human B lymphocytes: regulation and role in IgE production. J Allergy Clin Immunol. 2004;114(3):657–63.
Parrish-Novak J, Dillan S, Nelson A, Hammond A, Sprecher C, Gross J, et al. Interleukin-21 and its receptor are involved in NK cells expansion and regulation of lymphocyte function. Nature. 2000;408:57–63.
Ozaki K, Kikly K, Michalovich D, Young P, Leonard W. Cloning of a type I cytokine receptor most related to the IL-2 receptor beta chain. Proc Natl Acad Sci U S A. 2000;97(21):11439–44.
Clemente A, Pons J, Matamoros N, Iglesias J, Ferrer JB. Cells from common variable immunodeficiency patients fail to differentiate to antibody secreting cells in response to TLR9 ligand (CpG-ODN) or anti-CD40 + IL-21. Cell Immunol. 2011;268:9–15.
Borte S, Pan-Hammarstrom Q, Liu C, Sack U, Borte M, Wagner U, et al. Interleukin-21 restores immunoglobulin production ex vivo in patients with common variable immunodeficiency and selective IgA deficiency. Blood. 2009;114:4089–98.
Avery D, Bryant V, Ma C, Malefyt R, Tangye S. IL-21-induced isotype switching to IgG and IgA by human naive B cells is differentially regulated by IL-4. J Immunol. 2008;181:1767–79.
Yu J, Zhang L, Radigan L, Sanchez-Ramon S, Cunningham-Rundles C. TLR-mediated B cell defects and INF-alpha in common variable immunodeficiency. J Clin Immunol. 2012;32(1):50–60.
Crotty S. T follicular helper cell differentiation, function, and roles in disease. Immunity. 2014;41(4):529–42.
Saiki O, Ralph P, Cunningham-Rundles C, Good RA. Three distinct stages of B-cells defects in common varied immunodeficiency. Proc Natl Acad Sci U S A. 1982;79(19):6008–12.
Bryant A, Calver NC, Toubi E, Webster ADB, Farrant J. Classification of patients with common variable immunodeficiency by B cell secretion of IgM and IgG in response to anti-IgM and interleukin-2. Clin Immunol Immunopathol. 1990;56(2):239–48.
Lu X, Su B, Xia H, Zhang X, Liu Z, Ji Y, et al. Low double-negative CD3+CD4-CD8- T-cells are associated with incomplete restoration of CD4+ T-cells and higher immune activation in HIV-1 immunological non-responders. Front Immunol. 2016;7:579.
Rösel AL, Scheibenbogen C, Schliesser U, Sollwedel A, Hoffmeister B, Hanitsch L, et al. Classification of common variable immunodeficiencies using flow cytometry and a memory B-cell functionality assay. J Allergy Clin Immunol. 2015;135(1):198–208.
Kotlarz D, Zietara N, Uzel G, Weidemann T, Braun C, Diestelhorst J, et al. Loss-of-function mutations in the IL-21 receptor gene cause a primary immunodeficiency syndrome. J Exp Med. 2013;210:433–43.
Stepensky P, Keller B, Abuzaitoun O, Shaag A, Yaacov B, Unger S, et al. Extending the clinical and immunological phenotype of human interleukin-21 receptor deficiency. Haematologica. 2015;100:e72–e6.
Erman B, Bilic I, Hirschmugl T, Salzer E, Çagdas D, Esenboga S, et al. Combined immunodeficiency with CD4 lymphopenia and sclerosing cholangitis caused by a novel loss-of-function mutation affecting IL21R. Haematologica. 2015;100:e216–e9.
Salzer E, Kansu A, Sic H, Májek P, Ikincioğullari A, Dogu F, et al. Early-onset inflammatory bowel disease and common variable immunodeficiency-like disease caused by IL-21 deficiency. J Allergy Clin Immunol. 2014;133:1651–9.
Deenick E, Avery D, Chan A, Berglund L, Ives M, Moens L, et al. Naive and memory human B cells have distinct requirements for STAT3 activation to differentiate into antibody-secreting plasma cells. J Exp Med. 2013;2(12):2739–53.
Clemente A, Pons J, Lanio N, Matamoros N, Ferrer J. CD27+ B cells from a subgroup of common variable immunodeficiency patients are less sensitive to apoptosis rescue regardless of interleukin-21 signalling. Clin Exp Immunol. 2013;174:97–108.
Berrón-Ruiz L, López-Herrera G, Vargas-Hernández A, Santos-Argumedo L, López-Macías C, Isibasi A, et al. Impaired selective cytokine production by CD4(+) T cells in common variable immunodeficiency asssociated with the absence of memory B-cells. Clin Immunol. 2016;166-167:19–26.
Azizi G, Rezaei N, Kiaee F, Tavakolinia N, Yazdani R, Mirshafiey A, et al. T cell abnormalities in common variable immunodeficiency. J Investig Allergol Clin Immunol. 2016;26(4):233–43.
Acknowledgements
This project was supported by the Meakins-Christie Laboratories of the Research Institute of McGill University Health Centre, the Richard and Edith Strauss Canada Foundation, Immunodeficiency Canada, and AllerGen NCE. MD and MBS are recipients of Clinician Scientist Awards from the AllerGen NCE.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Ethical Approval
All procedures performed involving human participants were in accordance with the ethical standards of the institutional research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. Informed consent was obtained from all individual participants included in the study.
Electronic Supplementary Material
ESM 1
(DOCX 296 kb)
Rights and permissions
About this article
Cite this article
Desjardins, M., Béland, M., Dembele, M. et al. Modulation of the Interleukin-21 Pathway with Interleukin-4 Distinguishes Common Variable Immunodeficiency Patients with More Non-infectious Clinical Complications. J Clin Immunol 38, 45–55 (2018). https://doi.org/10.1007/s10875-017-0452-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10875-017-0452-0