Abstract
Introduction
Cycloxygenase-2 (COX-2) is an enzyme involved in prostaglandin E2 (PGE2) synthesis associated with higher renal cell carcinoma stage. COX-2 inhibition enhances interferon (IFN-α) anti-tumor immune effects in pre-clinical models. A phase II trial of celecoxib and IFN-α in a targeted population of metastatic renal cell carcinoma patients with maximal COX-2 expression was conducted.
Methods
Cytokine-naive metastatic renal cell carcinoma patients with tumors expressing ≥10% maximal COX-2 staining by immunohistochemistry received IFN-α 5 million units daily and celecoxib 400 mg orally twice daily in an open-label, single-arm phase II trial.
Results
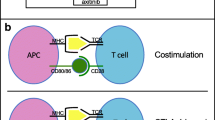
There were 3 partial responses among 17 patients (objective response rate 18%; 95% confidence interval, 4–43%). Time to progression was 5.6 months. Increased tumor staining 3+ for COX-2 was associated with increased baseline peripheral blood PGE2 levels, and these patients demonstrated less PGE2 decrease with therapy. Patients with more 3+ COX-2 staining had significantly more CD3+ (p = 0.004) and CD4+ (p = 0.002) IFN-γ T cells at baseline and a significantly greater decrease in these cells with therapy.
Discussion
Celecoxib plus IFN-α in renal cell carcinoma (RCC) patients with maximally staining COX-2 tumors does not significantly enhance overall response rates over IFN monotherapy.
Conclusion
COX-2-expressing RCC demonstrates inherent immunosuppression. COX-2 inhibition with IFN results in minimal immunomodulation and no augmented clinical activity in RCC.




Similar content being viewed by others
References
Onishi T et al. An assessment of the immunological environment based on intratumoral cytokine production in renal cell carcinoma. BJU Int. 1999;83(4):488–92.
Griffiths RW et al. Frequency of regulatory T cells in renal cell carcinoma patients and investigation of correlation with survival. Cancer Immunol Immunother. 2007;56(11):1743–53.
Wolf AM et al. Increase of regulatory T cells in the peripheral blood of cancer patients. Clin Cancer Res. 2003;9(2):606–12.
Li JF et al. The prognostic value of peritumoral regulatory T cells and its correlation with intratumoral cyclooxygenase-2 expression in clear cell renal cell carcinoma. BJU Int. 2009;103(3):399–405.
Troy AJ et al. Minimal recruitment and activation of dendritic cells within renal cell carcinoma. Clin Cancer Res. 1998;4(3):585–93.
Kiertscher SM et al. Tumors promote altered maturation and early apoptosis of monocyte-derived dendritic cells. J Immunol. 2000;164(3):1269–76.
Gigante M et al. Interferon-alpha (IFN-alpha)-conditioned DC preferentially stimulate type-1 and limit Treg-type in vitro T-cell responses from RCC patients. J Immunother. 2008;31(3):254–62.
Steitz J et al. Depletion of CD25(+) CD4(+) T cells and treatment with tyrosinase-related protein 2-transduced dendritic cells enhance the interferon alpha-induced, CD8(+) T-cell-dependent immune defense of B16 melanoma. Cancer Res. 2001;61(24):8643–6.
Buszello H. Antiproliferative effects of four different cytokines on renal carcinoma cell lines. Anticancer Res. 1995;15(3):735–8.
Uefuji K, Ichikura T, Mochizuki H. Cyclooxygenase-2 expression is related to prostaglandin biosynthesis and angiogenesis in human gastric cancer. Clin Cancer Res. 2000;6(1):135–8.
Masferrer JL et al. Antiangiogenic and antitumor activities of cyclooxygenase-2 inhibitors. Cancer Res. 2000;60(5):1306–11.
Waldmann TA. The interleukin-2 receptor. J Biol Chem. 1991;266(5):2681–4.
Akasaki Y et al. Induction of a CD4+ T regulatory type 1 response by cyclooxygenase-2-overexpressing glioma. J Immunol. 2004;173(7):4352–9.
Harizi H et al. Cyclooxygenase-2-issued prostaglandin e(2) enhances the production of endogenous IL-10, which down-regulates dendritic cell functions. J Immunol. 2002;168(5):2255–63.
Sharma S et al. Tumor cyclooxygenase 2-dependent suppression of dendritic cell function. Clin Cancer Res. 2003;9(3):961–8.
Sharma S et al. Tumor cyclooxygenase-2/prostaglandin E2-dependent promotion of FOXP3 expression and CD4+ CD25+ T regulatory cell activities in lung cancer. Cancer Res. 2005;65(12):5211–20.
Rini BI et al. Maximal COX-2 immunostaining and clinical response to celecoxib and interferon alpha therapy in metastatic renal cell carcinoma. Cancer. 2006;106(3):566–75.
Therasse P et al. New guidelines to evaluate the response to treatment in solid tumors. European Organization for Research and Treatment of Cancer, National Cancer Institute of the United States, National Cancer Institute of Canada. J Natl Cancer Inst. 2000;92(3):205–16.
Motzer RJ et al. Interferon-alfa as a comparative treatment for clinical trials of new therapies against advanced renal cell carcinoma. J Clin Oncol. 2002;20(1):289–96.
Miyata Y et al. Expression of cyclooxygenase-2 in renal cell carcinoma: correlation with tumor cell proliferation, apoptosis, angiogenesis, expression of matrix metalloproteinase-2, and survival. Clin Cancer Res. 2003;9(5):1741–9.
Kankuri-Tammilehto MK et al. Prognostic evaluation of COX-2 expression in renal cell carcinoma. Anticancer Res. 2010;30(7):3023–30.
Negrier S et al. Medroxyprogesterone, interferon alfa-2a, interleukin 2, or combination of both cytokines in patients with metastatic renal carcinoma of intermediate prognosis: results of a randomized controlled trial. Cancer. 2007;110(11):2468–77.
Tatsugami K, Eto M, Naito S. Influence of immunotherapy with interferon-alpha on regulatory T cells in renal cell carcinoma patients. J Interferon Cytokine Res. 2010;30(1):43–8.
Acknowledgment
This study was supported by NIH funding (R21CA123854 and R21CA123954). Funding for this study was provided by Merck & Co. Inc, Whitehouse Station, NJ (formerly Schering Plough Corp) and Pfizer Oncology (New York, NY).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Schwandt, A., Garcia, J.A., Elson, P. et al. Clinical and Immunomodulatory Effects of Celecoxib Plus Interferon-Alpha in Metastatic Renal Cell Carcinoma Patients with COX-2 Tumor Immunostaining. J Clin Immunol 31, 690–698 (2011). https://doi.org/10.1007/s10875-011-9530-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10875-011-9530-x