Abstract
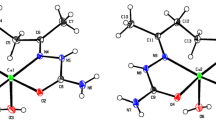
Previously unknown crystal structure of a copper(II) tetrafluoroborate hexahydrate salt was determined using single crystal X-ray diffraction. The unit cell parameters were determined at different temperatures (90, 150 and 270 K). The structure is isotypical with copper(II) perchlorate hexahydrate. The Raman spectrum was also recorded and discussed.
Graphical Abstract
The K.P.I. coefficient (78.0) and the FUV index (256.21 Å3) indicating very effective packing of the ions in the discussed structure, whereas the β angle is very close to the 90° and, in this way, the crystal could undergo a monoclinic \(\to \) orthorhombic phase transition at some lower temperatures.



Similar content being viewed by others
Data Availability
Crystallographic data of the compound copper(II) tetrafluoroborate hexahydrate Cu(BF4)2·6H2O or [CuII(H2O)6](BF4)2 (CSD 2310386) was deposited at the Cambridge Crystallographic Data Centre and could be obtained free of charge upon application to CCDC, 12 Union Road, Cambridge CB21EZ, UK [fax: (+ 44) 1223-336-033; email: deposit@ccdc.cam.ac.uk].
References
Pinaevskaya EN, Morgunova EM, Antoshkina NL, Sushkova SG (1964) J Appl Chem USSR 37(6):1176–1182
Kirgincev AN, Trushnikova LN, Lavrenteva VG (1972) Solubility of inorganic compounds in water. Chemistry 1972:216–217
Dewan JC, Thompson LK (1982) Can J Chem 60:121–132
Degtyarik MM, Lyakhov AS, Ivashkevich LS, Matulis VE, Matulis VE, Gruschinski S, Voitekhovich SV, Kersting B, Ivashkevich OA (2015) Dalton Trans 44(42):18518–18526
Gaughan AP, Dori Z, Ibers JA (1974) Inorg Chem 13(7):1657–1667
Brunton G (1968) Acta Crystallogr B Struct Crystallogr Cryst Chem 24(12):1703–1704
Brunton G (1969) Acta Crystallogr B Struct Crystallogr Cryst Chem 25(10):2161–2162
Jordan TH, Dickens B, Schroeder LW, Brown WE (1975) Acta Crystallogr B Struct Crystallogr Cryst Chem 31(3):669–672
Goreshnik E, Vakulka A, Žemva B (2010) Acta Crystallogr C 66:e9
Brunot B (1974) Chem Phys Lett 29(3):371–375
Asch L, Dézsi I, Lohner T, Molnár B (1976) Chem Phys Lett 39(1):177–179
Moss KC, Russell DR, Sharp DWA (1961) Acta Crystallogr 14(3):330–330
Tutton AE (1896) J Chem Soc Trans 69:344–495
Jahn HA, Teller E (1937) Proc R Soc A 161:220–235
Aramburu JA, Bhowmik A, García-Lastra JM, García-Fernández P, Moreno M (2019) J Phys Chem C 123(5):3088–3101
Robinson MWC, Pillinger KS, Graham AE (2006) Tetrahedron Lett 47(33):5919–5921
Garg SK, Kumar R, Chakraborti AK (2005) Tetrahedron Lett 46(10):1721–1724
Zagorac D, Müller H, Ruehl S, Zagorac J, Rehme S (2019) J Appl Crystallogr 52:918–925
CrysAlisPro, Agilent Technologies, Version 1.171.37.31 (release 14-01-2014 CrysAlis171 .NET).
Sheldrick GM (2015) Acta Crystallogr A 71:3–8
Sheldrick GM (2015) Acta Crystallogr C 71:3–8
Dolomanov OV, Bourhis LJ, Gildea RJ, Howard JAK, Puschmann H (2009) OLEX2: a complete structure solution, refinement and analysis program. J Appl Crystallogr 42:339–341
Crystal Impact GbR, Bonn, Germany (2004–2005) DIAMOND v3.1.
Fortes AD, Suard E, Lemée-Cailleau MH, Pickard CJ, Needs RJ (2009) J Am Chem Soc 131:13508–13515
Boeré RT (2023) Crystals 13(2):293
Ballirano P, Belardi G, Bosi F (2007) Acta Crystallogr E 63(7):i164–i165
Bosi F, Belardi G, Ballirano P (2009) Am Mineral 94(1):74–82
Janes R, Moore EA (2004) Metal-ligand bonding. The Royal Society of Chemistry, London, p 130
Figgis BN, Sobolev AN, Simmons CJ, Hitchman MA, Stratemeier H, Riley MJ (2000) Acta Crystallogr B 56(3):438–443
Zibaseresht R, Hartshorn RM (2006) Acta Crystallogr E 62:i19–i22
Zibaseresht R, Hartshorn RM (2012) Acta Crystallogr E 69(1):e1–e1
Spek AL (2003) J Appl Crystallogr 36:7–11
West CD (1935) Z Kristallogr 91:480–493
Rajakumar V, Krishnamurthy N, Ramakrishnant V, Kumar J (1992) J Raman Spectrosc 23:75–79
Bates JB, Quist AS, Boyd GE (1971) J Chem Phys 54(1):124–126
Barashkov MV, Zazhogin AA, Komyak AI, Shashkov SN (2000) J Appl Spectrosc 67(4):605–611
Gallucci JC, Gerkin RE (1989) Acta Cryst C45:1279–1284
Acknowledgements
The authors gratefully acknowledge the Slovenian Research Agency (ARRS) for financial support of the present study within the research program P1-0045 (Inorganic Chemistry and Technology).
Funding
Funding was provided by Slovenian Research Agency (ARRS) (Grant No. P1-0045).
Author information
Authors and Affiliations
Contributions
AV wrote the main manuscript, prepared figures, graph. abstract etc. AV prepared the crystalline specimen. EG refined cryst. structure and recorded the Raman spectrum. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
No potential conflict of interest was reported by the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Vakulka, A., Goreshnik, E. Copper(II) Tetrafluoroborate Hexahydrate: Preparation, Structure and Raman Spectrum. J Chem Crystallogr 54, 157–162 (2024). https://doi.org/10.1007/s10870-024-01008-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10870-024-01008-3