Abstract
The title compound C18H17NOS was synthesized by condensation of thiochroman-4-one with 4-dimethylaminobenzaldehyde. The possible existence of the 3-(4-(Dimethylamino)benzylidene)thiochroman-4-one (E)- and (Z)-geometrical isomers was investigated using B3LYP/6-31G**. Crystallographic and vibrational data are compared with the results of Density Functional Theory (DFT) B3LYP/6-31G** level. Bond orders were estimated using the NBO calculation method correlating with changes in the bond length. To explain deviation of an ideal molecular geometry, the concept of non-equivalent hybrid orbitals was invoked. The studies of X-ray diffraction revealed strong intermolecular interactions between dimers of the compound connected to each other through dipole–dipole interactions S–C, N–C y O–C in the absence of hydrogen bonding.
Graphical Abstract
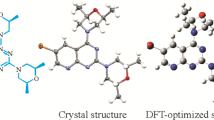
In this report, a study of the structural characterization of (Z)-3-(4-(dimethylamino)benzyliden)thiochroman-4-one is carried out by spectroscopic methods, single-crystal X-ray diffraction and functional density calculations.












Similar content being viewed by others
References
Hung TM, Thu CV, Dat NT, Ryoo S-W, Lee JH, Kim JC, Na M, Jung H-J, Bae K, Min BS (2010) Homoisoflavonoid derivatives from the roots of Ophiopogon japonicus and their in vitro anti-inflammation activity. Bioorg Med Chem Lett 20:2412–2416. https://doi.org/10.1016/j.bmcl.2010.03.043
Al Nakib T, Bezjak V, Meegan MJ, Chandy R (1990) Synthesis and antifungal activity of some 3-benzylidenechroman-4-ones, 3-benzylidenethiochroman-4-ones and 2-benzylidene-1-tetralones. Eur J Med Chem 25:455–462. https://doi.org/10.1016/0223-5234(90)90010-Z
Li Y-F, Liu Z-Q, Luo X-Y (2010) Properties of synthetic homoisoflavonoids to reduce oxidants and to protect linoleic acid and DNA against oxidation. J Agric Food Chem 58:4126–4131. https://doi.org/10.1021/jf904089q
Nguyen A-T, Fontaine J, Malonne H, Duez P (2006) Homoisoflavanones from Disporopsis aspera. Phytochemistry 67:2159–2163. https://doi.org/10.1016/j.phytochem.2006.06.021
Bennani B, Kerbal A, Daoudi M, Baba Filali B, Al Houari G, Jalbout AF, Mimouni M, Benazza M, Demailly G, Akkurt M, Öztürk S, Ben Hadda T (2007) Combined drug design of potential Mycobacterium tuberculosis and HIV-1 inhibitors: 3′,4′-di-substituted-4′H-spiro[isothiochromene-3,5′-isoxazol]-4(1H)-one. Arkivoc 2007:19–40. https://doi.org/10.3998/ark.5550190.0008.g03
Desideri N, Bolasco A, Fioravanti R, Proietti Monaco L, Orallo F, Yánez M, Ortuso F, Alcaro S (2011) Homoisoflavonoids: natural scaffolds with potent and selective monoamine oxidase-B inhibition properties. J Med Chem 54:2155–2164. https://doi.org/10.1021/jm1013709
Brien KA, Bandi RK, Kumar Behera A, Kumar Mishra B, Majumdar P, Satam V, Savagian M, Tzou S, Lee M, Zeller M, Robles AJ, Mooberry S, Pati H, Lee M (2012) Design, synthesis and cytotoxicity of novel chalcone analogs derived from 1-cyclohexylpyrrolidin-2-one and 2,3-dihydrobenzo[f]chromen-1-one. Arch Pharm Chem Life Sci 345:341–348. https://doi.org/10.1002/ardp.201100265
Wah Yee S, Jarno L, Sayed Gomaa M, Elford C, Ooi L-L, Coogan MP, McClelland R, Ian Nicholson R, Evans BAJ, Brancale A, Simons C (2005) Novel tetralone-derived retinoic acid metabolism blocking agents: synthesis and in vitro evaluation with liver microsomal and MCF-7 CYP26A1 cell assays. J Med Chem 48:7123–7131. https://doi.org/10.1021/jm0501681
Lévai A, Schág JB (1979) Synthesis of 3-benzylidenechroman-4-ones and -1-thiochroman-4-ones. Pharmazie 34:749
Böhler P, Tamm Ch (1967) The homo-isoflavones, a new class of natural product. Isolation and structure of Eucomin and Eucomol. Tetrahedron Lett 8:3479–3483. https://doi.org/10.1016/S0040-4039(01)89826-9
Tóth G, Halász J, Lévai A, Nemes C, Patonay T (1996) UV-induced isomerisation and ring transformation of (E)-3-arylidene-1-thiochromanones and -1-thioflavanones. Perkin Trans 2(4):547–550. https://doi.org/10.1039/P29960000547
Cheng X-M, Huang Z-T, Zheng Q-Y (2011) Topochemical photodimerization of (E)-3-benzylidene-4-chromanone derivatives from & #x03B2;-type structures directed by halogen groups. Tetrahedron 67:9093–9098. https://doi.org/10.1016/j.tet.2011.09.087
Modzelewska A, Pettit C, Achanta G, Davidson NE, Huang P, Khan SR (2006) Anticancer activities of novel chalcone and bis-chalcone derivatives. Bioorg Med Chem 14:3491–3495. https://doi.org/10.1016/j.bmc.2006.01.003
Sheldrick GM (2008) A short history of SHELX. Acta Cryst A 64:112–122. https://doi.org/10.1107/S0108767307043930
Putz H, Brandenburg K (2004) Diamond-crystal and molecular structure visualization. Bonn, Germany. http://www.crystalimpact.com/diamond
Frisch MJ, et al (2009) GAUSSIAN09. Revision D01. Gaussian Inc., Pittsburgh, Pennsylvania. http://gaussian.com
Valkonen A, Laihia K, Kolehmainen E, Kauppinen R, Perjési P (2012) Structural studies of seven homoisoflavonoids, six thiohomoisoflavonoids, and four sctruturally related compounds. Struct Chem 23:209–217. https://doi.org/10.1007/s11224-011-9860-6
Wiberg KB (1968) Application of the pople-santry-segal CNDO mathod to the cyclopropylcarbinyl and cyclobutyl catión and to bicyclobutane. Tetrahedron 24:1083–1096. https://doi.org/10.1016/0040-4020(68)88057-3
Katrusiak A, Ratajczak-Sitarz M, Kaluski Z, Orlov VD (1987) 3-Benzylidene-4-chromanone. Acta Cryst C 43:103–105. https://doi.org/10.1107/S0108270187096847
Marx A, Manivannan V, Suresh R, Kanagam CC, Sridhar B (2008) 8-Ethoxy-3-(4-isopropylbenzylidene)-6-methylchroman-4-one. Acta Cryst E 64:o328. https://doi.org/10.1107/S1600536807066925
Foster JP, Weinhold F (1980) Natural hybrid orbitals. J Am Chem Soc 102:7211–7218. https://doi.org/10.1021/ja00544a007
Andersson K, Sadlej AJ (1992) Electric dipole polarizabilities of atomic valence states. Phys Rev A 46:2356–2362. https://doi.org/10.1103/PhysRevA.46.2356
Acknowledgements
We would like to thank the Centro de Investigaciones Tecnológico y Humanístico de la Universidad Politécnica Territorial del Oeste de Sucre “Clodosbaldo Russian”. We thank FONACIT (Grant No. LAB-97000821) for financial support, Consejo de Investigación de la Universidad de Oriente (Project No. CI 02-010201-1898-14), Proyecto Estratégico Misión Ciencia FONACIT (Grant No. 2007001522). We want to thank the researcher Marielsys Moya for contacting the crystallographers.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Urdaneta, N., Núñez, J., Liendo, G. et al. (Z)-3-(4-(Dimethylamino)benzylidene)thiochroman-4-one: Synthesis, Crystal Structure and Molecular Modelling Using Density Functional Theory. J Chem Crystallogr 50, 176–186 (2020). https://doi.org/10.1007/s10870-019-00779-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10870-019-00779-4