Abstract
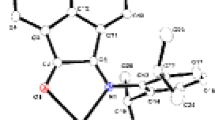
Two zinc binuclear complexes based on a hexaselenidobistannate bridging ligand, [{Zn(tren)}2(μ-Sn2Se6)] (1) and [{Zn(tepa)}2(μ-Sn2Se6)] (2) were prepared by solvothermal reactions of Zn, Sn, and Se in tris(2-aminoethyl)amine (tren) and tetraethylenepentamine (tepa) solvents, respectively. Compound 1 crystallizes in monoclinic space group C2/c, while 2 in tetragonal I4 1 /a. Acting as a bidentate bridging ligand, the bimeric [Sn2Se6]4− anion joins two [Zn(tren]2+ or two [Zn(tepa]2+ units via two trans terminal Se atoms to form the binuclear complexes 1 and 2, respectively. The Zn2+ ion is in a trigonal bipyramidal environment in 1 and in an octahedral environment in 2. Compounds 1 and 2 show well-defined absorption edges with band gaps of 2.38 and 2.31 eV at room temperature.
Graphical Abstract
New transition metal complexes base on hexaselenidobistannate ligand, [{Zn(tren)}2(μ-Sn2Se6)] and [{Zn(tepa)}2(μ-Sn2Se6)] were solvothermaly synthesized in tren and tepa (tren = tris(2-aminoethyl)amine, tepa = tetraethylenepentamine) solvents, respectively.







Similar content being viewed by others
References
Parise JB, Ko Y, Rijssenbeek J, Nellis DM, Tan K, Koch S (1994) Chem Commun 4:527
Zimmermann C, Anson CE, Weigend F, Clérac R, Dehnen S (2005) Inorg Chem 44:5686
Wu M, Su W, Jasutkar N, Huang X, Li J (2005) Mater Res Bull 40:21
Palchik O, Iyer RG, Liao JH, Kanatzidis MG (2003) Inorg Chem 42:5052
Manos MJ, Iyer RG, Quarez E, Liao JH, Kanatzidis MG (2005) Angew Chem Int Ed 44:3552
Bedard RL, Vail LD, Milson ST, Flanigen EM (1988) US Patent, 4880761
Bedard RL, Milson ST, Vaill LD, Bennett JM, Flanigen EM (1989) Zeolite: facts, figures, future, Proc 8th International Zeolite Conference. Elsevier, Amsterdam, p 375
Sheldrick WS, Wachhold M (1998) Coord Chem Rev 176:211
Sheldrick WS (2000) J Chem Soc Dalton Trans. doi: 10.1039/B003762L
Dehnen S, Melullis M (2007) Coord Chem Rev 251:1259
Zhou J, Dai J, Bian GQ, Li CY (2009) Coord Chem Rev 253:1221
Seidlhofer B, Pienack N, Bensch W (2010) Z Naturforsch 65:937
Zhou J (2016) Coord Chem Rev 315:112
Li J, Marler B, Kessler H, Soulard M, Kallus S (1997) Inorg Chem 36:4697
Jiang T, Lough A, Ozin GA (1998) Adv Mater 10:42
Jiang T, Lough A, Ozin GA, Bedard RL (1998) J Mater Chem 8:733
Fehlker A, Blacknik R (2001) Z Anorg Allg Chem 627:1128
Fehlker A, Blacknik R (2001) Z Anorg Allg Chem 627:411
Dehnen S, Zimmermann C (2002) Z Anorg Allg Chem 628:2463
Mitzi DB (2005) Inorg Chem 44:3755
Jia DX, Zhang Y, Dai J, Zhu QY, Gu XM (2004) Z Anorg Allg Chem 630:313
Chen Z, Wang RJ (1999) Acta Phys-Chim Sin 12:1070
Liang JJ, Chen JF, Zhao J, Pan YL, Zhang Y, Jia DX (2011) Z Anorg Allg Chem 637:445
Li J, Chen Z, Emge TJ, Yuen T, Proserpio DM (1998) Inorg Chim Acta 273:310
Shreeve-Keyer JL, Warren CJ, Dhingra SS, Haushalter RC (1997) Polyhedron 16:1193
Jia DX, Dai J, Zhu QY, Zhang Y, Gu XM (2004) Polyhedron 23:937
Gu XM, Dai J, Jia DX, Zhang Y, Zhu QY (2005) Cryst Growth Des 5:1845
Melullis M, Brandmayer MK, Dehnen S (2006) Z Anorg Allg Chem 632:64
Pienack N, Näther C, Bensch W (2009) Eur J Inorg Chem 2009:1575
Behrens M, Scherb S, Nather C, Bensch W (2003) Z Anorg Allg Chem 629:1367
Zhou J, Liu X, Chen GQ, Zhang F, Li LR (2010) Z Naturforsch 65b:1229
Kromm, A, Sheldrick WS (2006) Acta Crystallogr E62:m2767
Han JY, Liu Y, Lu JL, Tang CY, Shen YL, Zhang Y, Jia DX (2015) J Chem Crystallogr 45:355
Pienack N, Lehmann S, Lühmann H, El-Madani M, Näther C, Bensch W (2008) Z Anorg Allg Chem 634:2323
Zhou J, Zhang Y, Tang AB, Bian GQ, Jia DX, Dai J (2007) Inorg Chem Comm 10:348
Lu JL, Wang F, Shen YL, Tang CY, Zhang Y, Jia DX (2014) J Solid State Chem 216:65
Hilbert J, Näther C, Bensch W (2014) Z Anorg Allg Chem 640:2858
Liu GN, Guo GC, Zhang MJ, Guo JS, Zeng HY, Huang JS (2011) Inorg Chem 50:9660
Wendlandt WW, Hecht HG (1966) Reflectance spectroscopy. Interscience Publishers, New York
Sheldrick GM (1997) SHELXS-97, program for structure solution. University of Göttingen, Göttingen
Sheldrick GM (1997) SHELXL-97, program for structure refinement. University of Göttingen, Göttingen
Lu JL, Shen YL, Wang F, Tang CY, Zhang Y, Jia DX (2015) Z Anorg Allg Chem 641:561
Acknowledgements
This work was supported by the National Natural Science Foundation China (NSFC, no. 21171123), and the project funded by the Priority Academic Program Development (PAPD) of Jiangsu Higher Education Institutions.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Sun, Pp., Liu, Sz., Shen, Yl. et al. Solvothermal Syntheses, Crystal Structures and Optical Properties of New Members of Transition Metal Complexes Based on Hexaselenidobistannate Ligand. J Chem Crystallogr 47, 133–139 (2017). https://doi.org/10.1007/s10870-017-0689-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10870-017-0689-8