Abstract
Four new isostructural salts, [M(phen)3](I2Br)2·3CH2Cl2 where, M=Co (1), Ni (2), Cu (3), or Zn (4) have been synthesized by reacting the metal nitrates with 1,10–phenanthroline (phen) in presence of potassium bromide and iodine in a two phase procedure. The [Cu(phen)3]2+ coordination corresponds to tetragonally elongated octahedron while the other three are much more symmetrical. The pure Cu2+ complex as well as other salts doped with Cu2+ give hyperfine resolved EPR spectra in a wide temperature range showing dynamic Jahn–Teller distortion. The trihalide ion is nearly linear with bond distances in the range, I–I (2.786–2.833 Å) and I–Br (2.759–2.794 Å). Geometry optimization (DFT and MP2 methods) predicts a much more unsymmetrical “gas phase” structure for I2Br− ion. Calculated Raman frequencies are in reasonable agreement with observed values. Electronic spectral results show that the integrity of the trihalide ion is retained in solution.
Graphical Abstract
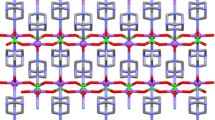
Three new isostructural compounds having the general formula [M(phen)3](I2Br)2 having nearly symmetrical I2Br− ion are synthesized and characterized.









Similar content being viewed by others
References
Svenson PH, Kloo L (2003) Chem Rev 103:1649
Blake AJ, Devillanova FA, Gould RO, Sheung Li W, Lippolis V, Parsons S, Radek C, Schroder M (1998) Chem Soc Rev 27:195
Haller H, Riedel S (2014) Anorg Z Allg Chem 640:1281
Babu R, Bhargavi G, Rajasekharan MV (2015) Eur J Inorg Chem 28:4689
Easton ME, Ward AJ, Hudson T, Turner P, Masters AF, Maschmeyer T (2015) Chem Eur J 21:2961
Buist AR, Kennedy AR (2014) Cryst Growth Des 14:6508
Rujas JM, Meazza L, Lim GK, Terraneo G, Pilati T, Harris KDM, Metrangolo P, Resnati G (2013) Angew Chem Int Ed 52:13444
Chernovyants MS, Burykin IV, Kostrub VV, Tsupak EB, Starikova ZA, Kirsanova JA (2012) J Mol Struct 1010:98
Chernovyants MS, Burykin IV, Berezov PN, Starikova ZA (2010) Mendeleev Commun 20:182
Chekhlov AN (2007) J Struct Chem 48:137
Kazheva ON, Aleksandrov GG, Dyachenko OA, Chernovyants MS, Simonyan SS, Lykova EO (2003) Russ Coord Chem 29:819
Babu R, Bhargavi G, Rajasekharan MV (2017) Eur J Inorg Chem 2017:2155
Ferraro JR, Beno MA, Thorn RJ, Wang HH, Webb KS, Williams JM (1986) J Phys Chem Solids 47:301
Torrent MM, Ribera E, Tkacheva V, Mata I, Molins E, Gancedo JV, Khasanov S, Zorina L, Shibaeva R, Wojciechowski R, Ulanski J, Wurst K, Veciana J, Laukhin V, Canadell E, Laukhina E, Rovira C (2002) Chem Mater 14:3295
Laukhina EE, Narymbetov BZ, Zorina LV, Khasanov SS, Rozenberg LP, Shibaeva RP, Buravov LI, Yagubskii EB, Avramenko NV, Van K (1997) Synth Met 90:101
Tateno A, Udagawa T, NaitoT, Kobayashi H, Kobayashib A, Nogami T (1994) J Mater Chem 4:1559
Emge VH, Wang H, Beno MA, Leung PCW, Firestone MA, Jenkins MC, Cook JD, Carlson KD, Williams JM (1985) Inorg Chem 24:1738
Dautatel OJ, Fourmigue M, Canadell E (2001) Chem Eur J 7:2635
Kobayashi H, Kato R, Kobayashi A, Saito G, Tokumoto M, Anzai H, Ishiguro T (1985) Chem Lett 14:1293
Endres H, Hiller M, Keller HJ, Bender K, Gogu E, Heinen I, Schweitzer D (1985) Z Naturforsch 40B:164
Schultz AJ, Emge TJ, Leung PCW, Beno MA, Wang HH, Williams JM (1986) Physica B C 143:351
Emge TJ, Leung PCW, Beno MA, Wang HH, Firestone MA, Webb KS, Carlson KD, Williams JM (1986) Mol Cryst Liq Cryst 132:363
Daoben Z, Ping W, Meixiang W, Zhaolou Y, Nailue Z, Gartner S, Schweitzer D (1986) Physica B and C 143:281
Carpenter GB (1966) Acta Cryst 20:330
Andrews L, Prochaska ES, Loewenschuss A (1980) Inorg Chem 19:463
Maki AG, Forneris R (1967) Spectrochimi Acta 23A:867
Assouma CD, Crochet A, Cheremond Y, Giese B, Fromm KM (2013) Angew Chem Int Ed 52:4682
Johnson MT, Dzolic Z, Cetina M, Wendt OF, Ohrstrom L, Rissanen K (2012) Cryst Growth Des 12:362
Chaviara AT, Tsipis AC, Cox PJ, Bolos CA (2005) Eur J Inorg Chem 2005:3491
Baker PK, Drew MGB, Meehan MM (2000) Inorg Chem Commun 3:393
Chernovyants MS, Burykin IV, Starikova ZA, Rassoshenko AA (2016) Russ J. Inorg Chem 61:217
Giese M, Albrecht M, Ivanova G, Valkonen A, Rissanen K (2012) Supramole Chem 24 48
Aragoni MC, Arca M, Devillanova FA, Hursthouse MB, Huth SL, Isaia F, Lippolis V, Mancini A, Verani G (2008) Eur J Inorg Chem 2008:3921
Parlow A, Hartl H, (1985) Z Naturforsch 40b:45
Minkwitz R, Berkei M, Ludwig R (2001) Inorg Chem 40:25
Mann L, Vobnacker P, Muller C, Riedel S (2016) Chem Eur J 23:244
Greatzel M (2006) Prog Photovolt 14:429
Fabjan C, Drobits J, Besenhard JO (eds) (1999) In Handbook of BatteryMaterials. Wiley-VCH, Weinheim
Khan MD, Majibur R, Gotoh Y, Morikawa H, Miura M (2009) Textile Res J 79:1305
Ramalakshmi D (1999) Shodhganga: a reservoir of Indian theses @INFLIBNET:Ph. D Thesis. University of Hyderabad, Hyderabad. http://hdl.handle.net/10603/1912
Ramalakshmi D, Reddy KR, Padmavathi D, Rajasekharan MV, Arulsamy N, Hodgson DJ (1999) Inorg Chim Acta 284:158
Menon S, Rajasekharan MV (1997) Inorg Chem 36:4983
Petermann L, Staehle R, David Pilz T, Sorsche D, Gorls H, Rau S (2015) Eur J Inorg Chem 2015:750
Joy S, Kramer T, Paul ND, Banerjee P, McGrady JE, Goswami S (2011) Inorg Chem 50:9993
Ha K, Kristallogr Z (2011) NCS 226:365
Faoro E, Oliveira GMD, Lang ES (2009) Polyhedron 28:63
Du J-M, Zhang Z-J, Lin H-M, Lia W, Guob G-C (2007) Acta Cryst E63:m3206
Tershansy MA, Goforth AM, Smith MD, Peterson LJ, Loyea H-CZ (2005) Acta Cryst E61:m1680
Blanchard S, Neese F, Bothe E, Bill E, Weyhermuller T, Wieghardt K (2005) Inorg Chem 44:3636
Horn C, Berben L, Chow H, Scudder M, Dance I (2002) CrystEngComm 4:7
Horn C, Scudder M, Dance I (2001) CrystEngComm 2:9
Horn C, Scudder M, Dance I (2001) CrystEngComm 2:1
Wieczorrek C (2000) Acta Cryst C56:1079
Horn C, Scudder M, Dance I (2000) CrystEngComm 2:196
Horn C, Scudder M, Dance I (2000) CrystEngComm 2:53
Kavoosian AG, Nafepour S, Nagel K, Tabbe K-F (1998) Z Naturforsch 53B:641
Freckmann B, Tabbe K-F (1981) Acta Cryst A37:c228
SAINTPLUS (2003) Bruker AXS Inc., Madison, WI
Sheldrick GM, SADABS (1996) Program for Empirical Absorption Correction. University of Gottingen, Gottingen
Sheldrick GM (2015) Acta Cryst C71:3
Neese F (2012) ORCA–an ab initio, density functional and semiempirical program package, Version 3.0.3. Max Planck Institute for Chemical Energy Conversion, Mulheim
Hitchman MA, Simmons CJ, Stratemeier H (2000) Appl Magn Reson 19:121
Allen HC Jr, Kokoszka GF, Inskeep RG (1964) J Am Chem Soc 86:1023
Mizuno M, Tanaka J, Harada I (1981) J Phys Chem 85:l1789
Acknowledgements
R. B. thanks the UGC, New Delhi for a Senior Research Fellowship. G. B. acknowledges support from the UGC in the form of a Women Post Doctoral Fellowship. This work was supported by the UPE-II (UGC), CAS(UGC) and PURSE-II (DST) programs.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Babu, R., Bhargavi, G. & Rajasekharan, M.V. Synthesis, Crystal Structures, Raman and EPR Studies of Mixed Trihalides in Association with Transition Metal (CoII–ZnII) Tris-chelates. J Chem Crystallogr 47, 86–94 (2017). https://doi.org/10.1007/s10870-017-0683-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10870-017-0683-1