Abstract
Direct crystallization of sulfur-nitrogen containing heterocyclic supramolecule I from acetic acid (solvent free) or in presence of an inert solvent afforded the proton-transfer compound III. The presence of nitrogen and sulfur atoms enhanced the solid-state packing of III to adopt interesting supramolecular interactions such as; S···H, S···N, N···O, O···H, S···H. Compounds I and III inhibited the growth of the Gram positive bacteria, B. subtilis and M. luteus.
Graphical Abstract
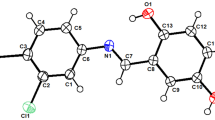
The supramolecularity of sulfur-nitrogen containing heterocyclic supramolecule I which crystallized from acetic acid in presence of an inert solvent has been investigated and afforded the proton-transfer compound III. In addition, the antimicrobial activity of compounds I and III has been examined and revealed that both compounds inhibited the growth of the Gram positive bacteria, B. subtilis and M. luteus. Part of compound III crystal structure showing only the cationic fragment. Heteroatoms were only labeled for clarity.







Similar content being viewed by others
References
Balasubramanian M, Keay JG (1996) In: Katritzky AR, Rees CW, Scriven EFV (eds) Comprehensive heterocyclic chemistry II, vol 5. Pergamon Press, Oxford, p 245
Bilker O, Lindo V, Panico M, Etiene AE, Paxton T, Dell A, Rogers M, Sinden RE, Morris HR (1998) Nature 392:289
Charris J, Martinez P, Dominguez J, Lopez S, Angel J, Espinoza G (2003) Heterocycl Commun 9:251
Chen YL, Fang KC, Sheu JY, Hsu SL, Tzeng CC (2001) J Med Chem 44:2374
Doube D, Bloun M, Brideau C, Chan C, Desmarais S, Eithier D, Falgueyeret JP, Friesen RW, Girad M, Girad Y, Guay J, Tagari P, Yong RN (1998) Bioorg Med Chem Lett 8:1255
Franck X, Fournet A, Prina E, Mahieux R, Hocquemiller R, Figadere B (2004) Bioorg Med Chem Lett 14:3635
Hu B, Collini M, Unwalla R (2006) J Med Chem 49:6151
Michael JP (1997) Nat Prod Rep 14:605
Musiol R, Jampilek J, Buchta V, Silva L, Niedbala H, Bodeszwa B, Palka A, Majerz-Maniecka K, Oleksyn B, Polanski J (2006) Bioorg Med Chem 14:3592
Roma G, Braccio MD, Grossi G, Chia M (2000) Eur J Med Chem 35:1021
Saeed A, Elhadi S (2011) Synth Commun 41:1435
Tempone AG, daSilva ACMP, Brandt CA, Martinez FS, Borborema SET, da Silveira MAB, de Andrade HF (2005) Antimicrob Agents Chemother 49:1076
Tsotinis A, Vlachou M, Zouroudis S, Jeney A, Timar F, Thurston DE, Roussakis C (2005) Lett Drug Des Discov 2:189
Vangapamdu S, Jain M, Jain R, Kaur S, Singh PP (2004) Bioorg Med Chem 12:2501
Geng X, Li S, Bian X, Xie Z, Wang C (2008) ARKIVOC xiv:50
Alshahateet SF (2010) Mol Cryst Liq Cryst 533:152
Alshahateet SF (2011) J Chem Crystallogr 41:276
Alshahateet SF, Bishop R, Craig DC, Kooli F, Scudder ML (2008) Cryst Eng Comm 10(3):297
Alshahateet SF, Bishop R, Craig DC, Scudder ML (2003) Cryst Eng Comm 5:417
Alshahateet SF, Bishop R, Scudder ML, Hu CY, Lau EHE, Kooli F, Judeh ZMA, Chow PS, Tan RBH (2005) Cryst Eng Comm 7:139
Alshahateet SF, Ong TT, Bishop R, Kooli F, Messali M (2006) Cryst Growth Des 6(7):1676
Kooli F, Hian PC, Weirong Q, Alshahateet SF, Chen F (2006) J Porous Mater 13(3):319
Kooli F, Mianhui L, Alshahateet SF, Chen F, Yinghuai Z (2006) J Phys Chem Solids 67(5):926
Zaghal MH, Qaseer HA, El-qisairi AK, Alshahateet SF, Shatnawi MY, Dawe LN (2009) J Chem Crystallogr 39(8):564
A preliminary account of the crystal structure of III (CCDC 691695) has been published: Alghezawi NM, Alshahateet SF, Bishop R (2010) JJC 5(3):211
Abu-Shanab B, Adwan G, Abu-Safiya D, Jarrar N, Adwan K (2004) Turk J Biol 28:99
Bishop R (1992) Org Synth 70:120
Bishosp R (1998) Org Synth IX:692
Cheng C–C, Yang C-J (1982) Org React 28:37
Alshahateet SF (2011) JJC 6(1):21
Alshahateet SF (2011) J Chem Crystallogr 41:570
Sekhon BS (2009) Ars Pharm 50(3):99
Whitesides GM, Wong AP (2006) MRS Bull 31:19
Corlette EM, Tieknik ERT (2009) J Chem Crystallogr 39:603
Smith G, Wermuth UD, Sagatys DS (2011) J Chem Crystallogr 41:17
Acknowledgments
The authors would like to thank Mutah University for support needed to conduct this work. Special thanks to Dr. Zaher Judeh (Nanyang Technological University, Singapore) for his technical help and valuable comments.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Alshahateet, S.F., Al-Zereini, W.A. & Alghezawi, N.M. Structural and Antimicrobial Activity Analyses of Different Forms of Sulfur-Nitrogen Containing Heterocyclic Supramolecule. J Chem Crystallogr 41, 1807–1811 (2011). https://doi.org/10.1007/s10870-011-0176-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10870-011-0176-6