Abstract
A total of 60 molecules of androstane derivatives of steroids (1–60) have been undertaken to predict their pharmacological effects, specific mechanisms of action, known toxicities, drug likeness, etc., by using the statistics of multilevel neighbourhoods of atoms (MNA) descriptors for active and inactive fragments. The biological activity spectra for substances have been correlated on SAR base (Structure–activity relationships data and knowledge base) which provides the different Pa (probability of activity) and Pi (probability of inactivity). The Lipinski’s rule predicts that all the androstane derivatives have stronger preponderance for “cancer-like-drug” molecules and some of their related analogous have been entered in the ANCI (American National Cancer Institute) database. Some selected bond distances and bond angles of interest have been taken into account and deviation of bond distances/bond angles, vis-a-vis the substitutional group and X–H···A intra/intermolecular hydrogen bonds have been discussed in detail. X–H···A intra/intermolecular hydrogen bonds in the identified molecules have been described with the standard distance and angle cut-off criteria. D–θ and d–θ scatter plots for X–H···A intra-and intermolecular interactions are presented for better understanding of packing interactions existing among these derivatives. Comparison of contacts from H(C) to O and H(O) to O, vis-a-vis their crystal structure reveals that contacts from H(O) to O predominate over H(C) to O. Solvent–solute/solute–solvent interactions have also been investigated to understand more complicated processes that occur for biomolecules in aqueous solutions. Most of the molecules show high value of drug-likeness whereas molecule-3 (82.5%), 36 (87.2%), 41 (83.7%), 43 (86.5%) and 50 (85.9%) exhibit low value of drug-likeness, instead of observed range of 90.3–99.2%.
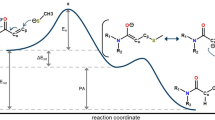
Graphical Abstract
Steroidal molecules are held in their defined 3-D structures by hydrogen bonds. The hydrogen bonding/solvent–solvent interactions for a steroidal molecule (androstane derivative) are plotted in Figure. The asymmetric unit cell in androstane derivative contains two crystallographically independent molecules and two acetic acid molecules. The two acetic acid molecules are connected to one another through solvent–solvent [O6(Acetic acid)–H6C(O6)···5′(Acetic acid); O6′(Acetic acid)–H6′C(O6′)···O5(Acetic acid)] interactions. The solvent-solvent interactions as observed in said steroidal derivative are rarely found in steroids and such investigations could be important to understand more complicated processes that occur for biomolecules in aqueous solutions.










Similar content being viewed by others
References
Makin HLJ (1975) Biochemistry of steroid hormones. Blackwell Scientific Publications, Oxford, p 4
Briggs MJ, Brothern J (1970) Steroid biochemistry and pharmacology. Academic Press, London/New York, p 121
Makin HLJ (1975) Biochemistry of steroid hormones. Blackwell Scientific Publications, Oxford, p 70
Gower DB, Bicknell DC (1972) Acta Endocr 70:567
Makin HLJ (1975) Biochemistry of steroid hormones. Blackwell Scientific Publications, Oxford, p 77
Bhavnaish Chand, Malik MA, Singh A (2009) Indian J Biochem Biophys (Communicated)
Bhavnaish C, Malik MA, Singh A (2009) Crystallogr Rep (Communicated)
Precioux PG, Busetta B, Courseille C, Hospital M (1975) Acta Crystallogr B31:1527
Weeks CM, Rohrer DC, Duax WL, Osawa Y (1975) Acta Crystallogr B31:2525
Rohrer DC, Duax WL, Osawa Y (1976) Acta Crystallogr B32:2410
Precioux PG, Busetta B, Hospital M (1977) Acta Crystallogr B33:563
Precioux PG, Busetta B, Hospital M (1977) Acta Crystallogr B33:566
Neubert LA, Carmack M, Huffman JC (1977) Acta Crystallogr B33:962
Rohrer DC, Strong PD, Duax WL, Segaloff A (1978) Acta Crystallogr B34:2913
Duax WL, Rohrer DC, Segaloff A (1982) Acta Crystallogr B38:531
De Cowe HJ, Cox PJ, Sim GA (1982) Acta Crystallogr B38:662
Cox PJ, Sim GA (1982) Acta Crystallogr B38:1360
Weeks CM, Strong PD, Duax WL, Vickery LE (1983) Acta Crystallogr C39:1698
Solans X, Piniella JF, Brainso JL, Miravitlles C (1987) Acta Crystallogr C43:2372
Danaci S, Kendi E, Mores FG, Behm H, Beurskens PT (1988) Acta Crystallogr C44:1677
Michel AG, Reul R, Dewez NM (1989) Acta Crystallogr C45:1760
Galdecki Z, Grochulski P, Wawrzak Z (1989) J Cryst Spect Res 19(3):577–587
Cox PJ, Mac Manus SM, Gibb BC, Nowell IW, Howie RA (1990) Acta Crystallogr C46:334
Eggleston DS, Lan-Hargest HY (1990) Acta Crystallogr C46:1686
Roszak AW, Codding PW (1990) Acta Crystallogr C46:1700
Drouin M, Ruel R, Michel AG (1991) Acta Crystallogr C47:1689
Meetsma A, Van Leusen D, Van Leusen AM (1993) Acta Crystallogr C49:351
Michel AG, Droun Marc (1993) Acta Crystallogr C49:1683
Brock CP, Song J (1995) Acta Crystallogr C51:2437
Steiner T (1996) Cryst Rev 6:1
Ramos Silva M, Paixao JA, De Almedia MJM, Tavares Da Silva EJ, Melo ML, Campos Neves AS (1996) Acta Crystallogr C52:2892
Paixao JA, Ramos Silva M, De Almedia MJM, Tavares Da Silva EJ, Melo ML, Campos Neves AS (1997) Acta Crystallogr C53:347
Brunskill APJ, Lalancette RA, Thompson HW (1997) Acta Crystallogr C53:903
Andradre LCR, Paixao JA, De Almedia MJM, Tavares Da Silva EJ, Melo ML, Campos Neves AS (1997) Acta Crystallogr C53:938
Anthony A, Jaskolski M, Nangia A, Desiraju GR (1998) Acta Crystallogr C54:1894
Anthony A, Jaskolski M, Nangia A, Desiraju GR (1998) Acta Crystallogr C54:1898
Lazar D, Stankovic S, Sakac M, Penov-Gasic K, Kovacevic R, Medic-Mijacevic L, Pilate T (1998) Acta Crystallogr C54:1965
Andrade LCR, Paixao JA, De Almedia MJM, Tavares Da Silva EJ, Melo ML, Campos Neves AS (1999) Acta Crystallogr C55:637
Anthony A, Jaskolski M, Nangia A (1999) Acta Crystallogr C55:787
Andrade LCR, Paixao JA, De Almedia MJM, Tavares Da Silva EJ, Melo ML, Campos Neves AS (1999) Acta Crystallogr C55:1186
Thompson HW, Lalancette RA, Brunskill APJ (1999) Acta Crystallogr C55:1680
Andrade LC R, Paixao JA, De Almedia MJM, Tavares Da Silva EJ, Melo ML, Campos Neves AS (1999) Acta Crystallogr C55:2149
Vasuki G, Parthasarthi V, Ramamurthi K, Jindal DP, Dubey S (2001) Acta Crystallogr C57:1062
Newman JM, Lalancette RA, Thompson HW (2002) Acta Crystallogr C58:o402
Hema R, Parthasarthi V, Thamotharan S, Dubey S, Jindal DP (2002) Acta Crystallogr C59:o421
Thamotharan S, Parthasarthi V, Gupta R, Jindal DP, Linden A (2002) Acta Crystallogr C58:o727
Hema R, Parthasarthi V, Thamotharan S, Dubey S, Jindal DP (2003) Acta Crystallogr C59:o213
Lalancette RA, Thompson HW (2003) Acta Crystallogr C59:o274
Thamotharan S, Parthasarthi V, Gupta R, Jindal DP, Linden A (2003) Acta Crystallogr C59:o724
Paixao JIF, Salvador JAR, Paixao JA, Beja AM, Ramos Silva M, Rocha Gonsalves AM (2004) Acta Crystallogr C60:o72
Thamotharan S, Parthasarthi V, Gupta R, Guleria S, Jindal DP, Linden A (2004) Acta Crystallogr C60:o75
Thamotharan S, Parthasarthi V, Dubey S, Jindal DP, Linden A (2004) Acta Crystallogr C60:o110
Thamotharan S, Parthasarthi V, Gupta R, Jindal DP, Linden A (2004) Acta Crystallogr C60:o158
Thamotharan S, Parthasarthi V, Gupta R, Jindal DP, Linden A (2004) Acta Crystallogr C60:o161
Paixao JIF, Salvador JAR, Paixao JA, Beja AM, Ramos Silva M, Rocha Gonsalves AM (2004) Acta Crystallogr C60:o630
Lazar D, Klisuric O, Stankovic S, Penov-Gasic K, Djurendic E, Kovacevic R (2004) Acta Crystallogr C60:o671
Rajnikant V, Dinesh J, Sawhney A, Mousmi B, Gupta BD (2004) J Chem Crystallogr 34(8):523
Andrade LCR, Paixao JA, De Almedia MJM, Fernandes Roleria FM, Travares Da Silva EJ (2005) Acta Crystallogr C61:o131
Rajnikant, Dinesh, Mousmi (2005) J Chem Cryst 36(5):283
Filimonov DA, Poroikov VV, Borodina Yu, Gloriozova T (1999) J Chem Inf Comput Sci 39:666
Poroikov VV, Filimonov DA (2001) Computer-assisted predictions of biological activity in search for and optimization of new drugs. Iridium Press, Moscow, p 149
Poroikov VV, Akimov DA, Shabelnikova E, Filimonov DA (2001) SAR and QSAR in Environ Res 12(4):327
Anzali S, Barnickel G, Cezanne B, Krug M (2001) J Med Chem 44:2432
Poroikov VV, Filimonov DA, Ihlenfeldt WD, Gloriozova TA, Lagunin AA, Borodina YV, Stepanchikova AV, Nicklaus MC (2003) J Chem Inf Comput Sci 43:228
Lipinski CA, Lombardo F, Dominy BW, Feeney PJ (1997) Adv Drug Deliv Rev 23:3
Sutton LE (1965) Tables of interatomic distances and configuration in molecules and ions. Special Publication No. 18. Chemical Society, London
Allen FH, Kennard O, Watson DG, Bramer L, Orpen AG, Taylor R (1987) J Chem Soc Perkin Trans 2:S1–S19
Bartell LS, Bonham RA (1960) J Chem Phys 32(3):824
Palenik GJ (1965) Acta Cryst 19:47
Sudralingam M (1966) Acta Crystallogr B21:495
Pletcher J, Sax M (1972) J Am Chem Soc 94:3998
Taylor R, Kennard O (1982) J Am Chem Soc 104:5063
Steiner T, Saenger W (1992) Acta Crystallogr B48:819
Steiner T, Saenger W (1992) J Am Chem Soc 114:10146
Steiner T (1996) Cryst Rev 6:7
Jefferey GA (1997) An introduction to hydrogen bonding. Oxford University Press, New York, p 400
Steiner T (1998) Acta Crystallogr B54:456
Desiraju GR, Steiner T (1999) The weak hydrogen bond in structural chemistry and biology. Oxford University Press Inc, New York, p 66
Steiner T (2002) Angew Chem Int Ed Eng 41:48
Olovsson I, Jonsson PG (1976) The hydrogen bond. Recent developments in theory & experiment, vol 2. North Holland, Amsterdam, p 393
Desiraju GR (1991) Acc Chem Res 24:270
Bernstein J (1994) In: Burgi HB, Dunitz JD (eds) Structure correlation, vol 2. VCH, Weinheim, p 431
Jeffery GA (1999) J Mol Struct 485:293
Desiraju GR, Steiner T (1999) The weak hydrogen bond in structural chemistry and biology. Oxford University Press Inc, New York, p 116
Steiner T (2001) Acta Crystallogr C57:775
Desiraju GR, Steiner T (1999) The weak hydrogen bond in structural chemistry and biology. Oxford University Press Inc, New York, p 13
Rivelino R, Canuto S, Coutinho K (2004) Braz J Phys 34(1):84
Scheiner S (1997) Hydrogen bonding: a theoretical perspective. Oxford University Press Inc, New York
Cramer CJ, Truhlar DG (1999) Chem Rev 99:2161
Kollman P (1993) Chem Rev 93:2395
Baldridge KK, Jonas V, Bain AD (2000) J Chem Phys 113(17):7519
Allen MP, Tildesley DJ (1987) Computer simulation of liquids. Oxford University Press, New York
Coutinho K, Canuto S, Zerner MC (2000) J Chem Phys 112:9874
Canuto S, Coutinho K, Trzesniak D (2002) Adv Quantum Chem 41:161
Jedlovszky P, Turi L (1997) J Phys Chem B101:5429
Tezuka T, Nakagawa M, Yokoi K, Nagawa Y, Yamagaki YT, Nakanishi H (1997) Tetrahedron Lett 38(24):4223
Davidson MG, Lamb S (1997) Polyhedron 16:4393
Klebe G, Mietzner T, Weber F (1999) J Comput-Aided Mol Des 13:35
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chand, B., Malik, M.A. Biological-Activity Predictions, Crystallographic Comparison and Role of Packing Interactions in Androstane Derivatives of Steroids. J Chem Crystallogr 41, 255–275 (2011). https://doi.org/10.1007/s10870-010-9979-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10870-010-9979-0