Abstract
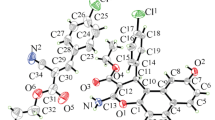
X-Ray analyses of 4-(naphthalen-1-ylamino)-3-nitro-chromen-2-one and 3-nitro-4-phenylamino-chromen-2-one showed that the mentioned compounds crystallize in the space groups P1- (triclinic crystal system; unit cell parameters: a = 8.087(2) Å, b = 9.241(3) Å, c = 10.911(3) Å, α = 93.77(3)°, β = 102.51(3)°, γ = 106.44(2)°, V = 756.4(4) Å3 and Z = 2) and P212121 (orthorhombic crystal system; unit cell parameters: a = 4.9274(9) Å, b = 14.725(3) Å, c = 17.866(4) Å, α = β = γ = 90°, V = 1296.3(5) Å3 and Z = 4), respectively. The analyses of crystal structures and gas phase conformations, inferred from single X-ray crystallographic and molecular modeling experiments, respectively, showed that the changes in π delocalization of the farmacoactive formal 3-amino-2-nitro-acrylic acid derivatives might explain the observed significant difference of the antimicrobial and antioxidant activities and spectral properties of two 4-arylamino-3-nitro-coumarin derivatives.
Graphical Abstract
In this work we considered the possibility that the changes in the π-delocalization of the pharmacoactive 4-(naphthalene-1-ylamino)- and 4-phenylamino-3-nitro-chromen-2-ones, inferred from X-ray analysis, might explain significant alterations of the antioxidant and antimicrobial activities (and spectral ones) of two coumarin derivatives.







Similar content being viewed by others
References
Radulović N, Stojanović G, Vukićević R, Dekić V, Dekić B, Palić R (2006) Monatsh Chem 137:1477
Savel’ev V, Pryanishnikova N, Artamonova O, Fedina I, Zagorevskii V (1975) Khim-Farm Zh 9:10
Savel’ev V, Artamonova O, Troitskaya V, Vinokurov V, Zagorevskii V (1973) Khim Geterotsikl Soedin 7:885
Tabaković K, Tabaković I, Trkovnik M, Trinajstić N (1983) Liebigs Ann Chem 11:1901
Enraf-Nonius CAD4 Software, Version 5.0 (1989) Enraf-Nonius, Delft, The Netherlands
CAD-4 Express Software (1994) Enraf-Nonius, Delft, The Netherlands
Sheldrick GM (1997) SHELXS97, Program for the solution of crystal structures. University of Göttingen, Germany
Sheldrick GM (1997) SHELXL97, Program for the refinement of crystal structures. University of Göttingen, Germany
Spek A (1990) Acta Cryst A46:C34
Spek A (1998) PLATON, a multipurpose: crystallographic tool. Utrecht University, Utrecht, The Netherlands
Farrugia L (1999) J Appl Crystallogr 32:837
Nardelli M (1995) J Appl Crystallogr 28:659
Farrugia L (1997) J Appl Crystallogr 30:565
NCCLS (National Committee for Clinical Laboratory Standards) (1997) Performance standards for antimicrobial disk susceptibility test, 6th edn. Approved Standard M2-A6, Wayne, PA
Prieto P, Pineda M, Aguilar M (1999) Anal Biochem 269:337
Drozd V, Knyazev V, Nam N, Yufit D, Struchkov Y, Stankevich I, Chistyakov A, Lezina V, Mozhaeva T, Saveljev V (1992) Tetrahedron 48:469
Rodios N, Bojilova A, Terzis A, Raptopoulou C (1994) J Heterocycl Chem 31:1129
Drozd V, Nam N, Knyazev V, Yufit D, Struchkov Y, Savel’ev V (1992) Russ J Org Chem 28:1496
Smith-Palmer A, Stewart J, Fyfe L (1998) Lett Appl Microbiol 26:118
Rodriguez E, Towers G, Mitchell J (1976) Phytochemistry 15:1573
Quang D, Hashimoto T, Stadler M, Radulović N, Asakawa Y (2005) Planta Med 71:1058
Acknowledgments
The authors acknowledge the Ministry of Science and Technological Development of Serbia for financial support (project number 172061).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Radulović, N.S., Bogdanović, G.A., Blagojević, P.D. et al. Could X-Ray Analysis Explain for the Differing Antimicrobial and Antioxidant Activity of Two 2-Arylamino-3-Nitro-Coumarins?. J Chem Crystallogr 41, 545–551 (2011). https://doi.org/10.1007/s10870-010-9918-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10870-010-9918-0