Abstract
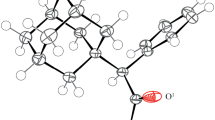
The title compound (C46H72N4O2, M r = 713.08) [systematic name: 1-isopropyl-3-((1R,4aS,10aR)-7-isopropyl-1, 4a-dimethyl-1,2,3,4,4a,9,10,10a-octahydrophenanthren-1-yl)urea] is a new derivative of dehydroabietic acid. It crystallized in the orthorhombic system, space group P2121 21 with cell dimensions of a = 9.5950(19) Å, b = 18.740(4) Å, c = 24.654(5) Å, V = 4,433.0(16) Å3, Z = 4, D c = 1.068 g/cm3, λ = 0.71073 Å, μ = 0.07 mm−1, F(000) = 1,568, final R = 0.0777 and wR = 0.1731 for 2,048 observed reflections with I > 2σ(I). Two crystallorgraphically independent molecules with different conformations co-exist in the structure. Each molecule exhibit classic tricyclic hydrophenanthrene structure, the two cyclohexane rings form a trans ring junction with chair and half-chair conformations, respectively. The crystal is stabilized by intermolecule hydrogen bonds and the molecules are in a head–tail zigzag arrangement.
Graphical Abstract
The title compound (C46H72N4O2, M r = 713.08) [systematic name: 1-isopropyl-3-((1R,4aS,10aR)-7-isopropyl-1, 4a-dimethyl-1,2,3,4,4a,9,10,10a-octahydrophenanthren-1-yl)urea] is a new derivative of dehydroabietic acid. It crystallized in the orthorhombic system, space group P2121 21 with cell dimensions of a = 9.5950(19) Å, b = 18.740(4) Å, c = 24.654(5) Å, V = 4,433.0(16) Å3, Z = 4, D c = 1.068 g/cm3, λ = 0.71073 Å, μ = 0.07 mm−1, F(000) = 1,568, final R = 0.0777 and wR = 0.1731 for 2,048 observed reflections with I > 2σ(I). Two crystallorgraphically independent molecules with different conformations co-exist in the structure. Each molecule exhibit classic tricyclic hydrophenanthrene structure, the two cyclohexane rings form a trans ring junction with chair and half-chair conformations, respectively. The crystal is stabilized by intermolecule hydrogen bonds and the molecules are in a head–tail zigzag arrangement.





Similar content being viewed by others
References
Wang HH, Liu B, Liu XQ, Zhang JW, Xian M (2008) Green Chem 10:1190
Chyu CF, Lin HC, Kuo YH (2005) Chem Pharm Bull 53:11
Rao XP, Song ZQ, He L (2008) Heteroa Chem 19(5):512
Savluchinske FS, Gigante B, Roseiro JC, Marcelo-Curto MJ (1999) J Microbiol Methods 35:201
Fernandez MA, Tornos MP, Garcia MD, Heras B, Villar AM, Saenz MT (2001) J Pharm Pharmacol 53:867
Sepulveda B, Astudillo L, Rodriguez J, Yanez T, Theoduloz C, Schmeda G (2005) Pharma Res 52:429
Rao XP, Song ZQ, Radbil B, Radbil A (2006) Acta Cryst E62:o5301
Rao XP, Song ZQ, Jia WH, Shang SB (2007) Acta Cryst E63:o3886
Qiu F, Hong Q, Jiang D, Zhu J, Huang LL (2008) Acta Cryst E64:o889
Rao XP, Song ZQ, Gong Y, Yao XJ, Shang SB (2006) Acta Cryst E62:o3450
Jiang D, Hong M, Qiu F, Zhu Jand Huang L (2007) Acta Cryst E63:o3623
Rao XP, Song ZQ, He L, Jia WH (2008) Chem Pharm Bull 56(11):1575
Sheldrick GM (1996) SADABS. University of Göttingen, Germany
Sheldrick GM (1997) SHELXL97 and SHELXS97. University of Göttingen, Germany
Sheldrick GM (2008) Acta Cryst A64:112
Acknowledgments
This research was financially supported by grants from National Natural Science Foundation of China (no. 30771690) and Forestry Commonwealth Industry Special Foundation of China (no. 200704008).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rao, XP., Wu, Y., Song, ZQ. et al. Crystal Structure of Unsymmetrically N-nor-dehydroabietyl-N′-isopropyl Urea. J Chem Crystallogr 40, 328–331 (2010). https://doi.org/10.1007/s10870-009-9656-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10870-009-9656-3