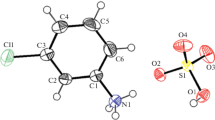
The crystal structure of the compound bis [2,4-diamino-5-(p-chlorophenyl)-6-ethyl pyrimidinium hydrogen sulfate] monohydrate was studied by X-ray diffraction methods. The compound crystallises in orthorhombic system, space group P212121, a=18.531(2) Å, b=16.190(2) Å, c=10.777(2) Å, V=3233.3(8), Z=4. The asymmetric unit shows the presence of two crystallographically independent pyrimethamine molecules, two hydrogen sulfate anions, and a water molecule. The hydrogen sulfate anions form a cyclic hydrogen bonded motif R2 2(8) with the 2-amino pyrimidine of the respective protonated pyrimethamine cations, through N–H···O hydrogen bonds. Thus the sulfate anions mimic the role of carboxylate anions observed in many aminopyrimidine-carboxylate interactions. This motif self assembles through DDAA array of quadruple hydrogen bonds, N–H···Cl, O–H···O and C–H···O hydrogen bonds.




Similar content being viewed by others
References
Macreadie, I.; Ginsberg, H.; Sirawaraporn, W.; Tilley, L. Parasitol. Today 2000, 16, 438–444.
Sethuraman, V.; Muthiah, P. T. Acta Crystallogr. Sect. E 2002, 58, o817–o818.
Hemamalini, M.; Muthiah, P. T.; Sridhar, B.; Rajaram, R. K. Acta Crystallogr. Sect. E 2005, 61, o1480–o1482.
Balasubramani, K.; Muthiah, P. T.; Rychlewska, U.; Plutecka, A. Acta Crystallogr. Sect. C 2005, 61, o586–o588.
Stanley, N.; Sethuraman, V.; Muthiah, P. T.; Luger, P.; Weber, M. Cryst. Growth Des. 2002, 631–635.
Onada, A.; Yamada, Y.; Doi, M.; Okamura, T.; Ueyama, N. Inorg. Chem. 2001, 40, 516–521.
Jacobson, C.K.; Quiocho, F.A. J. Mol. Biol. 1988, 204, 783–787.
Pflugrath, J.W.; Quiocho, F.A. Nature (London) 1985, 314, 257–260.
Muthiah, P.T.; Umadevi, B.; Stanley, N.; Shui, X.; Eggleston, D.S. Acta Crystallogr. Sect. E 2001, 57, o1179–o1182.
Hemamalini, M.; Muthiah, P.T.; Rychlewska, U.; Plutecka, A. Acta Crystallogr. Sect. C 2005, 61, o95–o97.
Muthiah, P.T.; Hemamalini, M.; Bocelli, G.; Cantoni, A. Acta Crystallogr. Sect. E 2004, 60, o2038–2040.
Cruickshank, D.W.J. J. Chem. Soc. 1961, 5486–5504.
Sansom, C.E.; Schwalbe, C.H.; Lambert, P.A.; Griffin, R.J.; Stevens, M.F.G. Biochim. Biophys. Acta 1989, 995, 21–27.
Lynch, D.E.; Jones, G.D. Acta Crystallogr. Sect. B 2004, 60, 748–764.
Etter, M.C. Acc. Chem. Res. 1990, 23, 120–126.
Kuyper, L.F. Crystallographic and Modeling Methods in Molecular Design; Bugg, C.E.; Ealick, S.E. New York: Springer Verlag, 1990; pp. 56–79.
Quesada, A.; Marchal, A.; Low, J.N.; Glidewell, C. Acta. Crystallogr. Sect. C 2003, 59, o102–o104.
Sethuraman, V.; Stanley, N.; Muthiah, P.T.; Sheldrick, W.S.; Winter, M.; Luger, P.; Weber, M. Cryst. Growth Des. 2003, 5, 823–828.
Hunter, C.A. Chem. Soc. Res. 1994, 23, 101–109.
Sheldrick, G.M. SHELXS-97 and SHELXL-97. University of Gottingen: Germany, 1997.
Spek, A.L. Acta Crystallogr. Sect. A 1990, 46, C-34.
Siemens. XSCANS. Version 2.1 Siemens Analytical X- ray Instruments Inc., Madison, Wisconsin, USA, 1994.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary material
CCDC-293607 contains the supplementary crystallographic data for this paper. These data can be obtained free of charge at www.ccdc.cam.ac.uk/conts/retrieving.html [or from the Cambridge Crystallographic Data Centre (CCDC), 12 Union Road, Cambridge CB2 IEZ, UK; fax: C44 (0) 1223-336033; e-mail: deposit@ccdc.cam.ac.uk].
Rights and permissions
About this article
Cite this article
Devi, P., Muthiah, P.T., Bocelli, G. et al. Hydrogen bonding patterns in bis(pyrimethamine hydrogen sulfate) monohydrate. J Chem Crystallogr 36, 857–861 (2006). https://doi.org/10.1007/s10870-006-9145-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10870-006-9145-x