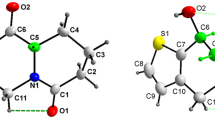
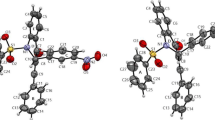
White plates of 2,3-diphenylquinoxaline crystallize in the monoclinic space group P21/n with Z = 4 and a = 6.0325(3) Å, b = 10.9516(6) Å, c = 22.5985(13) Å, and β = 95.107(2)°. The phenyl rings in 2,3-diphenylquinoxaline form torsion angles of 36.88(5)° and 53.32(4)° with the plane defined by the quinoxaline moiety. Yellow plates of 2,3-diphenylbenzo[g]quinoxaline crystallize in the monoclinic space group C2/c with Z = 8 and a = 25.0621(16) Å, b = 7.7190(5) Å, c = 21.2225(14) Å, and β = 123.674(2)°. The phenyl rings in 2,3-diphenylbenzo[g]quinoxaline form torsion angles of 46.89(3)° and 43.42(3)° with the plane defined by the benzo[g]quinoxaline moiety. Packing in 2,3-diphenylquinoxaline can best be described as following the herringbone motif; whereas the packing in 2,3-diphenylbenzo[g]quinoxaline crystals can also be described as herringbone—albeit a herringbone pattern made up of head-to-tail oriented neighbors.




Similar content being viewed by others
References
(a) Salzner, U.; Lagowski, J.B.; Pickup, P.G.; Poirier, R.A. J. Org. Chem. 1999, 64, 7419. (b) Pomerantz, M.; Gu, X.; Zhang, S.X. Macromolecules 2001, 34, 1817. (c) Alvarez, J.; Sun, L.; Crooks, R.M. Chem. Mater. 2002, 14, 3995. (d) Zotti, G.; Zecchin, S.; Schiavon, G.; Berlin, A.; Pagani, G.; Borgonovo, M.; Lazzaroni, R. Chem. Mater. 1997, 9, 2876–2886. (e) Zotti, G.; Salmaso, R.; Gallazzi, M. C.; Marin, R. A. Chem. Mater. 1997, 9, 791–795. (f) Kaufmann, Th.; Ghanem, M.; Otter, R. Chem. Ber. 1982, 115, 459. (g) Lukeš, V.; Breza, M.; Végh, D.; Hrdlovič, P.; Krajèovič, J.; Laurinc, V. Synth. Metals 2001, 124, 279. (h) Hrdlovič, P.; Krajèovič, J.; Végh, D. J. Photochem. Photobiol. 2001, 144, 73–81. (i) Yamamoto, T.; Lee, B.-L.; Kokubo, H.; Kishida, H.; Hirota, K.; Wakabayashi, T.; Okamoto, H. Macromol. Rapid Commun. 2003, 24, 440.
(a) Voloshin, Y.Z.; Belov, A.S.; Lebedev, A.Y.; Varzatskii, O.A.; Antipin, M.Y.; Starikova, Z.A.; Kron, T.E. Russ. Chem. Bull. 2004, 53, 1218. (b) Toshima, K.; Ozawa, T.; Kimura, T.; Matsumura, S. Bioorg. Med. Chem. Lett. 2004, 14, 2777. (c) Aguirre, G.; Cerecetto, H.; Di Maio, R.; Gonzalez, M.; Alfaro, M.E.M.; Jaso, A.; Zarranz, B.; Ortega, M.A.; Aldana, I.; Monge-Vega, A. Bioorg. Med. Chem. Lett. 2004, 14, 3835. (d) Ammar, Y.A.; Ismail, M.M.F.; El-Gaby, M.S.A.; Zahran, M.A. Indian J. Chem. 2002, 41B, 1486.
(a) Crundwell, G.; Sayers, D.; Herron, S.R.; Kantardjieff, K. Acta Cryst. 2003, E59, 314. (b) Crundwell, G.; Linehan, J.; Updegraff III, J.B.; Zeller, M.; Hunter, A.D. Acta Cryst. 2004, 60, o656.
(a) Raj, S.S.S.; Fun, H.-K.; Chen, X.-F.; Zhu, X.-H.; You, X.-Z. Acta Cryst. 1999, C55, 2035. (b) Jung, O.-S.; Park, S.H.; Kim, Y.J.; Lee, Y.-A.; Jang, H.G.; Lee, U. Inorg. Chim. Acta 2001, 312, 93. (c) Bu, X.-H.; Liu, H.; Du, M.; Wong, K.M.-C.; Yam, V.W.-W.; Shionoya, M. Inorg. Chem. 2001, 40, 4143. (d) Liu, H.; Du, M. J. Mol. Struct. 2002, 607, 143.
Escuer, A.; Vincente, R.; Comas, T.; Ribas, J.; Gomez, M.; Solans, X.; Gatteschi, D.; Zanchini, C. Inorg. Chim. Acta 1991, 181, 51. This cobalt complex is the sole example of 2,3-di-2-pyridylquinoxaline binding through a pyridyl ring and benzo[g]quinoxaline. (Other structures have shown bidentate binding via both pyridyl rings.)
(a) Scott, S.M.; Gordon, K.C.; Burell, A.K. J. Chem Soc., Dalton Trans. 1999, 2669. (b) Gordon, K.C.; Al-Obaidi, A.H.R.; Jayaweera, P.M.; McGarvey, J.J.; Malone, J.F.; Bell, S.E.J. J. Chem. Soc., Dalton Trans. 1996, 1591. (c) Heirtzler, F.; Dias, S.; Neuburger, M. Eur. J. Inorg. Chem. 2004, 685.
Goodwin, K.V.; Pennington, W.T.; Petersen, J.D. Acta Cryst. 1990, C46, 898.
Moliner, N.; Munoz, M.C.; Van Koningsbruggen, P.J.; Real, J.A. Inorg. Chim. Acta 1998, 274, 1.
Choi, Y.-Y.; Wong, W.-T. J. Chem. Soc., Dalton Trans. 1999, 331.
Waterland, M.R.; Simpson, T.J.; Gordon, K.C.; Burrell, A.K. J. Chem. Soc., Dalton Trans. 1998, 185.
Rasmussen, S.C.; Richter, M.M.; Yi, E.; Place, H.; Brewer, K.L. Inorg. Chem. 1990, 29, 3926.
(a) Lalrempuia, R.; Govindaswamy, P.; Mozharivskyj, Y.A.; Kollipara, M.R. Polyhedron, 2004, 1069. (b) Lalrempuia, R.; Kollipara, M.R. Polyhedron, 2003, 3155. (c) Rillema, D.P.; Taghdiri, D.G.; Jones, D.S.; Keller, C.D.; Worl, L.A.; Meyer, T.J.; Levy, H.A. Inorg. Chem. 1987, 26, 578.
Bock, H.; John, A.; Näther, C.; Ruppert, K. Helv. Chim. Acta 1994, 77, 1505.
Naskar, J.P.; Chowbury, S.; Drew, M.G.B.; Datta, D. New J. Chem. 2002, 26, 170.
Naskar, J.P.; Hati, S.; Datta, D.; Tocher, D.A. Chem. Comm. 1997, 1319.
Foss, P.C.D.; Crundwell, G.; Updegraff, J.B., III; Zeller, M.; Hunter, A.D. Acta Cryst. 2004, E60, o1281.
Williamson, K.L. Macroscale and Microscale Organic Experiments; D.C. Heath and Company: Lexington, Massachusetts, 1989.
SMART: Bruker Axs Inc., 5465 East Cheryl Parkway, Madison, WI 53711–5373, USA, 2000.
Saint V-7.06A Bruker Axs Inc., 5465 East Cheryl Parkway, Madison, WI 53711–5373, USA, 2000.
SADABS-Bruker Nonius area detector scaling and absorption correction—V2.10 Bruker Axs Inc., 5465 East Cheryl Parkway, Madison, WI 53711-5373, USA, 2000.
Shelxtl V-6.14 Bruker Axs Inc., 5465 East Cheryl Parkway, Madison, WI 53711–5373, USA, 2000.
Farrugia, L.J. J. Appl. Cryst. 1997, 30, 565.
Spek, A.L. Acta Cryst. 1990, A46, C34. Spek, A.L. Platon Vakgroep Algemene Chemie, University of Utrecht, Utrecht, The Netherlands, 2002.
Allen, F.H. Acta Cryst. 2002, B58, 380.
(a) Belizzi, M.; Foss, P.C.D.; Pelto, R.; Crundwell, G.; Bruckner, C.; Updegraff III, J.B.; Zeller, M.; Hunter, A.D. Z. Kryst. NCS, 2004, 129. (b) Purushothaman, B.; Varghese, B.; Bhyrappa, P. Acta Cryst. 2001, C57, 252.
Escuer, A.; Vicente, R.; Comas, T.; Ribas, J.; Gomez, M.; Solans, X. Inorg. Chim. Acta 1990, 177, 161.
Sessler, J.L.; Berthon-Gelloz, G.; Gale, P.A.; Camiolo, S.; Anslyn, E.V.; Anzenbacker, P., Jr.; Furuta, H.; Kirkovitz, G.J.; Lynch, V.M.; Maeda, H.; Morosini, P.; Scherer, M.; Shriver, J.; Zimmerman, R.S. Polyhedron 2003, 22, 2963.
Wu, J.-Z.; Xu, Z.-G.; Li, Y.; Zhang, W.-G.; Zeng, H.-P.; Xu, X.; Zhou, Z.-Y. J. Chem. Cryst. 2002, 32, 75.
Kanoktanaporn, S.; MacBride, J.A.H.; King, T.J. J. Chem. Res. 1980, 406, 4901.
Acknowledgments
The authors would like to thank Dr Charles F. Campana of Bruker AXS for the data collections. S. Cantalupo and Dr Crundwell would like to thank Dr Amy R. Howell and Dr Martha Morton from the University of Connecticut for summer mentoring and assisting in the acquisition of 1H and 13C spectra, respectively. The authors would like to acknowledge an NSF MRI Grant (0420322) and American Chemical Society Petroleum Research Fund Grant for support of this research (#38867-B5 m) as well as support from an NIH Area Grant (No. 1 R15 AI057408-01), the assistance of the University of Connecticut REU program (CHE0354012), CCSU-AAUP research grants, and CCSU Faculty-Student research grants.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Cantalupo, S.A., Salvati, H., McBurney, B. et al. Crystal structures and packing motifs of 2,3-diphenylquinoxaline and 2,3-diphenylbenzo[g]quinoxaline. J Chem Crystallogr 36, 17–24 (2006). https://doi.org/10.1007/s10870-005-9010-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10870-005-9010-3