Abstract
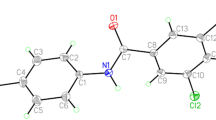
The tris-2-chloro and 2-bromotribenzylamines are prepared from aqueous ammonia and 2-chlorobenzyl chloride and 2-bromobenzyl bromide, respectively, in ethanol. Recrystallization yielded colorless cubes of each product. The crystal structures are each solved in space group P\(\bar 1\), and are isostructural. The tris-2-chloro compound, 1, has a = 7.4226(5) Å, b = 9.0825(7) Å, c = 14.529(1) Å, α = 78.279(1)∘, β = 82.389(1)∘, γ = 84.661(1)∘, and V = 948.41(12) Å3 with Z = 2, and d calc = 1.368 Mg/m3. The tris-2-bromo analog, 2, has a = 7.6569(11) Å, b = 9.0922(13) Å, c = 14.614(2) Å, α = 79.286(2)∘, β = 81.777(2)∘, γ = 85.401(2)∘, and V = 987.9(2) Å3 with Z = 2, and d calc = 1.762 Mg/m3. Lithium–halogen exchange experiments conducted in tetrahydrofuran at −78∘C using n-butyl lithium revealed that no exchange occurred for the tris-2-chloro compound, but did occur for the tris-2-bromo analog to yield tribenzylamine upon quench and work-up.
Similar content being viewed by others
References
Anderegg, G.; Wenk, F. Helv. Chim. Acta 1967, 50, 2330.
Fox, S.; Nanthakumar, A.; Wikström, M.; Karlin, K.D.; Blackburn, N.J. J. Am. Chem. Soc. 1996, 118, 24.
Ménage, S.; Zang, Y.; Hendrich, M.P.; Que, L., Jr. J. Am. Chem. Soc. 1992, 114, 7786.
Jackson, C.L.; White, J.F. Am. Chem. J. 1880–1881, 2, 315.
Koch, S.A.; Welch, M.J.; Cutler, C.S.; Quarless, D.A., Jr. PCT Int. Appl. 1999, WO 9956791.
Blessing, R. Acta Crystallogr. Sect A 1995, 51, 33.
SHELXTL V6.10; Bruker Analytical X-Ray Systems: Madison, WI, 2000.
SHELXTL V6.12; Bruker Analytical X-Ray Systems: Madison, WI.
Iwasaki, F.; Iwasaki, H. Acta Crystallogr. B 1972, 28, 3370.
Stoicheff, B.P. Tetrahedron 1962, 17, 135.
Cotton, F.A.; Wilkinson, G.; Gaus, P.L. Basic Inorganic Chemistry; Wiley: New York, 1994; pp. 60–61.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chen, Q., Buss, C.E., Young, V.G. et al. Synthesis and structural studies of tris-2-chlorobenzylamine and tris-2-bromobenzylamine. J Chem Crystallogr 35, 177–181 (2005). https://doi.org/10.1007/s10870-005-2954-5
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s10870-005-2954-5