Abstract
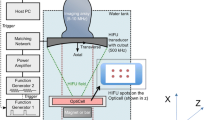
Gas-filled microbubbles attached to cell surfaces can interact with focused ultrasound to create microstreaming of nearby fluid. We directly observed the ultrasound/microbubble interaction and documented that under certain conditions fluorescent particles that were attached to the surface of live cells could be removed. Fluorescently labeled liposomes that were larger than 500 nm in diameter were attached to the surface of endothelial cells using cRGD targeting to αvβ3 integrin. Microbubbles were attached to the surface of the cells through electrostatic interactions. Images taken before and after the ultrasound exposure were compared to document the effects on the liposomes. When exposed to ultrasound with peak negative pressure of 0.8 MPa, single microbubbles and groups of isolated microbubbles were observed to remove targeted liposomes from the cell surface. Liposomes were removed from a region on the cell surface that averaged 33.1 μm in diameter. The maximum distance between a single microbubble and a detached liposome was 34.5 μm. Single microbubbles were shown to be able to remove liposomes from over half the surface of a cell. The distance over which liposomes were removed was significantly dependent on the resting diameter of the microbubble. Clusters of adjoining microbubbles were not seen to remove liposomes. These observations demonstrate that the fluid shear forces generated by the ultrasound/microbubble interaction can remove liposomes from the surfaces of cells over distances that are greater than the diameter of the microbubble.






Similar content being viewed by others
References
Bao, S., Thrall, B.D., Miller, D.L.: Transfection of a reporter plasmid into cultured cells by sonoporation in vitro. Ultrasound Med. Biol. 23(6), 953–959 (1997)
Koch, S., et al.: Ultrasound enhancement of liposome-mediated cell transfection is caused by cavitation effects. Ultrasound Med. Biol. 26(5), 897–903 (2000)
Chin, C.T., et al.: Brandaris 128: a digital 25 million frames per second camera with 128 highly sensitive frames. Rev. Sci. Instrum. 74(12), 5026–5034 (2003)
Morgan, K.E., et al.: Experimental and theoretical evaluation of microbubble behavior: effect of transmitted phase and bubble size. IEEE Trans. Ultrason. Ferroelectr. Freq. Control. 47(6), 1494–1509 (2000)
Ferrara, R.P.K., Borden, M.: Ultrasound microbubble contrast agents: fundamentals and application to gene and drug delivery. Annu. Rev. Biomed. Eng. 9, 415–447 (2007)
Elder, S.A.: Cavitation microstreaming. J. Acoust. Soc. Am. 31(1), 54–64 (1959)
Liu, J., Lewis, T.N., Prausnitz, M.R.: Non-invasive assessment and control of ultrasound-mediated membrane permeabilization. Pharm. Res. 15(6), 918–924 (1998)
Delalande, A., et al.: Sonoporation at a low mechanical index. Bubble Sci. Eng. Technol. 3(1), 3–12 (2011)
Doinikov, A.A., Haac, J.F., Dayton, P.A.: Resonance frequencies of lipid-shelled microbubbles in the regime of nonlinear oscillations. Ultrasonics 49(2), 263–268 (2009)
Ibsen, S., et al.: Sonogenetics is a non-invasive approach to activating neurons in Caenorhabditis elegans. Nat. Commun. 6, (2015)
Maxwell, A.D., et al.: Cavitation clouds created by shock scattering from bubbles during histotripsy. J. Acoust. Soc. Am. 130, 1888 (2011)
Miller, M.W., Miller, D.L., Brayman, A.A.: A review of in vitro bioeffects of inertial ultrasonic cavitation from a mechanistic perspective. Ultrasound Med. Biol. 22(9), 1131–1154 (1996)
Kodama, T., Tomita, Y.: Cavitation bubble behavior and bubble–shock wave interaction near a gelatin surface as a study of in vivo bubble dynamics. Appl. Phys. B Lasers Opt. 70, 139–149 (2000)
Li, Z.G., et al.: A Single-Cell Membrane Dynamic from Poration to Restoration by Bubble-Induced Jetting Flow. 15th International Conference on Miniaturized Systems for Chemistry and Life Sciences, p. 94-96 (2011).
Sundaram, J., Mellein, B.R., Mitragotri, S.: An experimental and theoretical analysis of ultrasound-induced permeabilization of cell membranes. Biophys. J. 84, 3087–3101 (2003)
Prentice, P., et al.: Membrane disruption by optically controlled microbubble cavitation. Nat. Phys. 1(2), 107–110 (2005)
Skyba, D.M., et al.: Direct in vivo visualization of intravascular destruction of microbubbles by ultrasound and its local effects on tissue. Circulation 98(4), 290–293 (1998)
Stieger, S.M., et al.: Enhancement of vascular permeability with low-frequency contrast-enhanced ultrasound in the chorioallantoic membrane model1. Radiology 243(1), 112–121 (2007)
Ohl, C.-D., et al.: Surface cleaning from laser-induced cavitation bubbles. Appl. Phys. Lett. 89(7), 074102 (2006)
Cui, W., et al.: Neural progenitor cells labeling with microbubble contrast agent for ultrasound imaging in vivo. Biomaterials 34(21), 4926–4935 (2013)
Klein, N.J., et al.: Alteration in glycosaminoglycan metabolism and surface charge on human umbilical vein endothelial cells induced by cytokines, endotoxin and neutrophils. J. Cell Sci. 102, 821–832 (1992)
Rerat, V., et al.: αvβ3 integrin-targeting Arg-Gly-asp (RGD) peptidomimetics containing oligoethylene glycol (OEG) spacer. J. Med. Chem. 52, 7029–7043 (2009)
Trikha, M., et al.: Multiple roles for platelet GPIIb/IIIa and v 3 integrins in tumor growth, angiogenesis, and metastasis. Cancer Res. 62, 2824–2833 (2002)
Fåhraeus, R., Lane, D.P.: The p16INK4a tumour suppressor protein inhibits αvβ3 integrin-mediated cell spreading on vitronectin by blocking PKC-dependent localization of αvβ3 to focal contacts. EMBO J. 18(8), 2106–2118 (1999)
Maubant, S., et al.: Blockade of αvβ3 and αvβ5 integrins by RGD mimetics induces anoikis and not integrin-mediated death in human endothelial cells. Blood 108(9), 3035–3044 (2006)
Wiewrodt, R., et al.: Size-dependent intracellular immunotargeting of therapeutic cargoes into endothelial cells. Blood 99(3), 912–922 (2002)
Rose, G.: A separable and multipurpose tissue culture chamber. Tex. Rep. Biol. Med. 12(4), 1074 (1954)
Ibsen, S., et al.: The behavior of lipid debris left on cell surfaces from microbubble based ultrasound molecular imaging. Ultrasonics 54, 2090–2098 (2014)
Wang, D.S., et al.: Cationic versus neutral microbubbles for ultrasound-mediated gene delivery in cancer. Radiology 264(3), 721–732 (2012)
Borden, M.A., et al.: DNA and polylysine adsorption and multilayer construction onto cationic lipid-coated microbubbles. Langmuir 23(18), 9401–9408 (2007)
Nomikou, N., et al.: Studies on neutral, cationic and biotinylated cationic microbubbles in enhancing ultrasound-mediated gene delivery in vitro and in vivo. Acta Biomater. 8(3), 1273–1280 (2012)
Schutt, E., Pelura, T., Hopkins, R.: Osmotically-stabilized microbubble ultrasound contrast agents. Acad. Radiol. 3, S188–S190 (1996)
Schutt, E., et al.: Injectable microbubbles as contrast agents for diagnostic ultrasound imaging: the key role of perfluorochemicals. Angew. Chem. Int. Ed. 42, 3218–3235 (2003)
Ibsen, S., et al.: A novel nested liposome drug delivery vehicle capable of ultrasound triggered release of its payload. J. Control. Release 155(3), 358–366 (2011)
Ibsen, S., Benchimol, M., Esener, S.: Fluorescent microscope system to monitor real-time interactions between focused ultrasound, echogenic drug delivery vehicles, and live cell membranes. Ultrasonics 53(1), 178–184 (2013)
Stride, E., Saffari, N.: Microbubble ultrasound contrast agents: a review. Proc. Inst. Mech. Eng. Part H: J. Eng. Med. 217, 429–447 (2003)
Lehenkari, P.P., Horton, M.A.: Single integrin molecule adhesion forces in intact cells measured by atomic force microscopy. Biochem. Biophys. Res. Commun. 259, 645–650 (1999)
Marrink, S.-J., et al.: Adhesion forces of lipids in a phospholipid membrane studied by molecular dynamics simulations. Biophys. J. 74, 931–943 (1998)
van der Meer, S.M., et al.: Microbubble spectroscopy of ultrasound contrast agents. J. Acoust. Soc. Am. 121, 648 (2007)
Miller, D.L., Thomas, R.M.: Ultrasound contrast agents nucleate inertial cavitation in vitro. Ultrasound Med. Biol. 21(8), 1059–1065 (1995)
Schutt, C., et al.: The influence of distance between microbubbles on the fluid flow produced during ultrasound exposure. J. Acoust. Soc. Am. 136(6), 3422-3430 (2014)
Collis, J., et al.: Cavitation microstreaming and stress fields created by microbubbles. Ultrasonics 50(2), 273–279 (2010)
Tho, P., Manasseh, R., Ooi, A.: Cavitation microstreaming patterns in single and multiple bubble systems. J. Fluid Mech. 576, 191–233 (2007)
Lauterborn, W., Kurz, T.: Physics of bubble oscillations. Rep. Prog. Phys. 73(10), 106501 (2010)
Funding
The study was supported by Grant Numbers T32 CA121938, R25 CA153915 NCI, and 5U54CA119335-05 from the National Cancer Institute. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Cancer Institute or the National Institutes of Health. Support was also provided by the UCSD Cancer Center Specialized Support Grant P30 CA23100.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Electronic supplementary material
ESM 1
(PDF 7 kb)
Rights and permissions
About this article
Cite this article
Ibsen, S., Mora, R., Shi, G. et al. Removal of ligand-bound liposomes from cell surfaces by microbubbles exposed to ultrasound. J Biol Phys 43, 493–510 (2017). https://doi.org/10.1007/s10867-017-9465-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10867-017-9465-4