Abstract
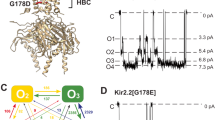
There is clear evidence that the net magnitude of negative charge at the intracellular end of inwardly rectifying potassium channels helps to generate an asymmetry in the magnitude of the current that will pass in each direction. However, a complete understanding of the physical mechanism that links these charges to current rectification has yet to be obtained. Using Brownian dynamics, we compare the conduction mechanism and binding sites in rectifying and non-rectifying channel models. We find that in our models, rectification is a consequence of asymmetry in the hydrophobicity and charge of the pore lining. As a consequence, inward conduction can occur by a multi-ion conduction mechanism. However, outward conduction is restricted, since there are fewer ions at the intracellular entrance and outwardly moving ions must cross the pore on their own. We pose the question as to whether the same mechanism could be at play in inwardly rectifying potassium channels.






Similar content being viewed by others
References
Hibino, H., Inanobe, A., Furutani, K., Murakami, S., Findlay, I., Kurachi, Y.: Inwardly rectifying potassium channels: their structure, function and physiological roles. Physiol. Rev. 90, 291–366 (2010)
Reimann, F., Ashcroft, F.M.: Inwardly rectifying potassium channels. Curr. Opin. Cell Biol. 11, 503–508 (1999)
Neusch, C., Weishaupt, J.H., Bähr, M.: Kir channels in the CNS: emerging new roles and implications for neurological diseases. Cell Tissue Res. 311, 131–138 (2003)
Yang, J., Jan, Y.N., Jan, L.Y.: Control of rectification and permeation by residues in two distinct domains in an inward rectifier K\(^{+}\) channel. Neuron 14, 1047–1054 (1995)
Xie, L.H., John, S.A., Ribalet, B., Weiss, J.N.: Regulation of gating by negative charges in the cytoplasmic pore in the Kir2.1 channel. J. Physiol. 561, 159–168 (2004)
Matsuda, H., Saigusa, A., Irisawa, H.: Ohmic conductance through the inwardly rectifying K channel and blocking by internal Mg\(^{2+}\). Nature 325, 156–159 (1987)
Guo, D., Ramu, Y., Klem, A.M., Lu, Z.: Mechanism of rectification in inward-rectifier K\(^{+}\) channels. J. Gen. Physiol. 121, 261–275 (2003)
Robertson, J.L., Palmer, L.G., Roux, B.: Long-pore electrostatics in inward rectifier potassium channels. J. Gen. Physiol. 132, 613–632 (2008)
Robertson, J.L., Palmer, L.G., Roux, B.: Multi-ion distributions in the cytoplasmic domain of inward rectifier potassium channels. Biophys. J. 103, 434–443 (2012)
Yeh, S.H., Chang, H.K., Shieh, R.C.: Electrostatics in the cytoplasmic pore produce intrinsic inward rectification in Kir2.1 channels. J. Gen. Physiol. 126, 551–562 (2005)
Clarke, O.B., Caputo, A.T., Hill, A.P., Vandenberg, J.I., Smith, B.J., Gulbis, J.M.: Domain reorientation and rotation of an intracellular assembly regulate conduction in Kir potassium channels. Cell 141, 1018–1029 (2010)
Fujiwara, Y., Kubo, Y.: Functional roles of charged amino acid residues on the wall of the cytoplasmic pore of Kir2.1. J. Gen. Physiol. 127, 401–419 (2006)
Pegan, S., Arrabit, C., Zhou, W., Kwiatkowski, W., Collins, A., Slesinger, P.A., Choe, S.: Cytoplasmic domain structures of Kir2.1 and Kir3.1 show sites for modulating gating and rectification. Nat. Neurosci. 8, 279–287 (2005)
Guo, D., Lu, Z.: Interaction mechanisms between polyamines and IRK1 inward rectifier K\(^{+}\) channels. J. Gen. Physiol. 122, 485–500 (2003)
Silver, M.R., DeCoursey, T.E.: Intrinsic gating of inward rectifier in bovine pulmonary artery endothelial cells in the presence or absence of internal Mg\(^{2+}\). J. Gen. Physiol. 96, 109–133 (1990)
Nichols, C.G., Lopatin, A.N.: Inward rectifier potassium channels. Annu. Rev. Physiol. 59, 171–191 (1997)
Aleksandrov, A., Velimirovic, B., Clapham, D.E.: Inward rectification of the IRK1 K\(^{+}\) channel reconstituted in lipid bilayers. Biophys. J. 70, 2680–2687 (1996)
Stanfield, P.R., Davies, N.W., Shelton, P.A., Sutcliffe, M.J., Khan, I.A., Brammer, W.J., Conley, E.C.: A single aspartate residue is involved in both intrinsic gating and blockage by Mg\(^{2+}\) of the inward rectifier, IRK1. J. Physiol. 478, 1–6 (1994)
Lopatin, A.N., Nichols, C.G.: [K\(^{+}\)] dependence of open-channel conductance in cloned inward rectifier potassium channels (IRK1, Kir2.1). Biophys. J. 71, 682–694 (1996)
Guo, D., Lu, Z.: IRK1 inward rectifier K\(^{+}\) channels exhibit no intrinsic rectification. J. Gen. Physiol. 120, 539–551 (2002)
Hilder, T.A., Chung, S.H.: Conductance properties of the inwardly rectifying channel, Kir3.2: molecular and Brownian dynamics study. Biochim. Biophys. Acta. 1828, 471–478 (2013)
Hilder, T.A., Chung, S.H.: Conduction and block of inward rectifier K\(^{+}\) channels: predicted structure of a potent blocker of Kir2.1. Biochem. 52, 967–974 (2013)
Chung, S.H., Allen, T.W., Kuyucak, S.: Modeling diverse range of potassium channels with Brownian dynamics. Biophys. J. 83, 263–277 (2002)
Hoyles, M., Kuyucak, S., Chung, S.H.: Computer simulation of ion conductance in membrane channels. Phys. Rev. E 58, 3654–3661 (1998)
Whorton, M.R., MacKinnon, R.: Crystal structure of the mammalian GIRK2 K\(^{+}\) channel and gating regulation by G proteins, PIP\(_{2}\), and sodium. Cell 147, 199–208 (2011)
Tao, X., Avalos, J.L., Chen, J., MacKinnon, R.: Crystal structure of the eukaryotic strong inward-rectifier K\(^{+}\) channel Kir2.2 at 3.1 Å resolution. Science 326, 1668–1674 (2009)
Hansen, S.B., Tao, X., MacKinnon, R.: Structural basis of PIP\(_{2}\) activation of the classical inward rectifier K\(^{+}\) channel Kir2.2. Nature 477, 495–498 (2011)
Chung, S.H., Corry, B.: Conduction properties of KcsA measured using Brownian dynamics with flexible carbonyl groups in the selectivity filter. Biophys. J. 93, 44–53 (2007)
Gordon, D., Krishnamurthy, V., Chung, S.H.: Generalized Langevin models of molecular dynamics simulations with applications to ion channels. J. Chem. Phys. 131, 134102–1–134102-13 (2009)
Allen, T.W., Kuyucak, S., Chung, S.H.: Molecular dynamics study of the KcsA potassium channel. Biophys. J. 77, 2502–2516 (1999)
Chung, S.H., Allen, T.W., Hoyles, M., Kuyucak, S.: Permeation of ions across the potassium channel: Brownian dynamics studies. Biophys. J. 77, 2517–2533 (1999)
Brelidze, T.I., Niu, X., Magelby, K.L.: A ring of eight negatively charged amino acids doubles the conductance of BK channels and prevents inward rectification. Proc. Natl. Acad. Sci. USA 100, 9017–9022 (2003)
Nishida, M., MacKinnon, R.: Structural basis of inward rectification: cytoplasmic pore of the G protein-gated inward rectifier GIRK1 at 1.8 Å resolution. Cell 111, 957–965 (2002)
Xie, L.H., John, S.A., Weiss, J.N.: Spermine block of the strong inward rectifier potassium channel Kir2.1: dual roles of surface charge screening and pore block. J. Gen. Physiol. 120, 53–66 (2002)
Acknowledgments
Silvie Ngo provided excellent technical assistance, for which we are grateful. We thank Michael Thomas for his scientific advice. This work was supported by the NCI National Facility at the Australian National University. We gratefully acknowledge the support from the Australian Research Council through a Discovery Early Career Researcher Award, and the National Health and Medical Council.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Hilder, T.A., Corry, B. & Chung, SH. Multi-ion versus single-ion conduction mechanisms can yield current rectification in biological ion channels. J Biol Phys 40, 109–119 (2014). https://doi.org/10.1007/s10867-013-9338-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10867-013-9338-4