Abstract
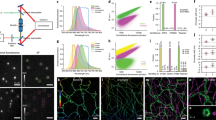
Confocal fluorescence microscopy and two-photon microscopy have become important techniques for the three-dimensional imaging of intact cells. Their lateral resolution is about 200–300 nm for visible light, whereas their axial resolution is significantly worse. By superimposing the spherical wave fronts from two opposing objective lenses in a coherent fashion in 4Pi microscopy, the axial resolution is greatly improved to ∼100 nm. In combination with specific tagging of proteins or other cellular structures, 4Pi microscopy enables a multitude of molecular interactions in cell biology to be studied. Here, we discuss the choice of appropriate fluorescent tags for dual-color 4Pi microscopy and present applications of this technique in cellular biophysics. We employ two-color fluorescence detection of actin and tubulin networks stained with fluorescent organic dyes; mitochondrial networks are imaged using the photoactivatable fluorescent protein EosFP. A further example concerns the interaction of nanoparticles with mammalian cells.
Similar content being viewed by others
References
Hell, S.W.: Far-field optical nanoscopy. Science 316, 1153–1158 (2007)
Egner, A., Hell, S.W.: Fluorescence microscopy with super-resolved optical sections. Trends Cell Biol. 15, 207–215 (2005)
Hell, S.W., Stelzer, E.H.K.: Fundamental improvement of resolution with a 4Pi-confocal fluorescence microscope using two-photon excitation. Opt. Commun. 93, 277–282 (1992)
Gustafsson, M.G., Agard, D.A., Sedat, J.W.: Sevenfold improvement of axial resolution in 3D widefield microscopy using two objective lenses. Proc. SPIE 2412, 147–156 (1995)
Gustafsson, M.G., Agard, D.A., Sedat, J.W.: I5M: 3D widefield light microscopy with better than 100 nm axial resolution. J. Microsc. 195, 10–16 (1999)
Heintzmann, R., Jovin, T.M., Cremer, C.: Saturated patterned excitation microscopy—a concept for optical resolution improvement. J. Opt. Soc. Am. A Opt. Image Sci. Vis. 19, 1599–1609 (2002)
Gustafsson, M.G.: Nonlinear structured-illumination microscopy: wide-field fluorescence imaging with theoretically unlimited resolution. Proc. Natl. Acad. Sci. U.S.A. 102, 13081–13086 (2005)
Hell, S.W., Wichmann, J.: Breaking the diffraction resolution limit by stimulated emission: stimulated emission depletion microscopy. Opt. Lett. 19, 780–782 (1994)
Hofmann, M., Eggeling, C., Jakobs, S., Hell, S.W.: Breaking the diffraction barrier in fluorescence microscopy at low light intensities by using reversibly photoswitchable proteins. Proc. Natl. Acad. Sci. U.S.A. 102, 17565–17569 (2005)
Betzig, E., Patterson, G.H., Sougrat, R., Lindwasser, O.W., Olenych, S., Bonifacino, J.S., Davidson, M.W., Lippincott-Schwartz, J., Hess, H.F.: Imaging intracellular fluorescent proteins at nanometer resolution. Science 313, 1642–1645 (2006)
Hess, S.T., Girirajan, T.P., Mason, M.D.: Ultra-high resolution imaging by fluorescence photoactivation localization microscopy. Biophys. J. 91, 4258–4272 (2006)
Rust, M.J., Bates, M., Zhuang, X.: Sub-diffraction-limit imaging by stochastic optical reconstruction microscopy (STORM). Nat. Methods 3, 793–795 (2006)
Egner, A., Geisler, C., von Middendorff, C., Bock, H., Wenzel, D., Medda, R., Andresen, M., Stiel, A.C., Jakobs, S., Eggeling, C., Schonle, A., Hell, S.W.: Fluorescence nanoscopy in whole cells by asynchronous localization of photoswitching emitters. Biophys. J. 93, 3285–3290 (2007)
Egner, A., Hell, S.W.: Fluorescence microscopy with super-resolved optical sections. Trends Cell Biol. 15, 207–215 (2005)
Gugel, H., Bewersdorf, J., Jakobs, S., Engelhardt, J., Storz, R., Hell, S.W.: Cooperative 4Pi excitation and detection yields sevenfold sharper optical sections in live-cell microscopy. Biophys. J. 87, 4146–4152 (2004)
Kano, H., Jakobs, S., Nagorni, M., Hell, S.W.: Dual-color 4Pi-confocal microscopy with 3D-resolution in the 100 nm range. Ultramicroscopy 90, 207–213 (2001)
Nagorni, M., Hell, S.W.: 4Pi-confocal microscopy provides three-dimensional images of the microtubule network with 100- to 150-nm resolution. J. Struct. Biol. 123, 236–247 (1998)
Shaner, N.C., Patterson, G.H., Davidson, M.W.: Advances in fluorescent protein technology. J. Cell Sci. 120, 4247–4260 (2007)
Wiedenmann, J., Ivanchenko, S., Oswald, F., Nienhaus, G.U.: Identification of GFP-like proteins in nonbioluminescent, azooxanthellate anthozoa opens new perspectives for bioprospecting. Mar. Biotechnol. (NY) 6, 270–277 (2004)
Wiedenmann, J., Vallone, B., Renzi, F., Nienhaus, K., Ivanchenko, S., Röcker, C., Nienhaus, G.U.: Red fluorescent protein eqFP611 and its genetically engineered dimeric variants. J. Biomed. Opt. 10, 14003 (2005)
Nienhaus, K., Renzi, F., Vallone, B., Wiedenmann, J., Nienhaus, G.U.: Exploring chromophore–protein interactions in fluorescent protein cmFP512 from Cerianthus membranaceus: X-ray structure analysis and optical spectroscopy. Biochemistry 45, 12942–12953 (2006)
Nienhaus, K., Renzi, F., Vallone, B., Wiedenmann, J., Nienhaus, G.U.: Chromophore-protein interactions in the anthozoan green fluorescent protein asFP499. Biophys. J. 91, 4210–4220 (2006)
Ando, R., Hama, H., Yamamoto-Hino, M., Mizuno, H., Miyawaki, A.: An optical marker based on the UV-induced green-to-red photoconversion of a fluorescent protein. Proc. Natl. Acad. Sci. U.S.A. 99, 12651–12656 (2002)
Ando, R., Mizuno, H., Miyawaki, A.: Regulated fast nucleocytoplasmic shuttling observed by reversible protein highlighting. Science 306, 1370–1373 (2004)
Chudakov, D.M., Belousov, V.V., Zaraisky, A.G., Novoselov, V.V., Staroverov, D.B., Zorov, D.B., Lukyanov, S., Lukyanov, K.A.: Kindling fluorescent proteins for precise in vivo photolabeling. Nat. Biotechnol. 21, 191–194 (2003)
Patterson, G.H., Lippincott-Schwartz, J.: A photoactivatable GFP for selective photolabeling of proteins and cells. Science 297, 1873–1877 (2002)
Wiedenmann, J., Ivanchenko, S., Oswald, F., Schmitt, F., Röcker, C., Salih, A., Spindler, K.D., Nienhaus, G.U.: EosFP, a fluorescent marker protein with UV-inducible green-to-red fluorescence conversion. Proc. Natl. Acad. Sci. U.S.A. 101, 15905–15910 (2004)
Wiedenmann, J., Nienhaus, G.U.: Live-cell imaging with EosFP and other photoactivatable marker proteins of the GFP family. Expert Rev. Proteomics 3, 361–374 (2006)
Nienhaus, G.U., Nienhaus, K., Hölzle, A., Ivanchenko, S., Renzi, F., Oswald, F., Wolff, M., Schmitt, F., Röcker, C., Vallone, B., Weidemann, W., Heilker, R., Nar, H., Wiedenmann, J.: Photoconvertible fluorescent protein EosFP: biophysical properties and cell biology applications. Photochem. Photobiol. 82, 351–358 (2006)
Nienhaus, K., Nienhaus, G.U., Wiedenmann, J., Nar, H.: Structural basis for photo-induced protein cleavage and green-to-red conversion of fluorescent protein EosFP. Proc. Natl. Acad. Sci. U.S.A. 102, 9156–9159 (2005)
Schneider, M., Barozzi, S., Testa, I., Faretta, M., Diaspro, A.: Two-photon activation and excitation properties of PA-GFP in the 720–920-nm region. Biophys. J. 89, 1346–1352 (2005)
Tsutsui, H., Karasawa, S., Shimizu, H., Nukina, N., Miyawaki, A.: Semi-rational engineering of a coral fluorescent protein into an efficient highlighter. EMBO Rep. 6, 233–238 (2005)
Ivanchenko, S., Glaschick, S., Röcker, C., Oswald, F., Wiedenmann, J., Nienhaus, G.U.: Two-photon excitation and photoconversion of EosFP in dual-color 4Pi confocal microscopy. Biophys. J. 92, 4451–4457 (2007)
Ivanchenko, S., Röcker, C., Oswald, F., Wiedenmann, J., Nienhaus, G.U.: Targeted green-to-red photoconversion of EosFP, a fluorescent marker protein. J. Biol. Phys. 31, 249–259 (2005)
Bestvater, F., Spiess, E., Stobrawa, G., Hacker, M., Feurer, T., Porwol, T., Berchner-Pfannschmidt, U., Wotzlaw, C., Acker, H.: Two-photon fluorescence absorption and emission spectra of dyes relevant for cell imaging. J. Microsc. 208, 108–115 (2002)
Blab, G.A., Lommerse, O.H.M., Cognet, L., Harms, G.S., Schmidt, T.: Two-photon excitation action cross-sections of the autofluorescent proteins. Chem. Phys. Lett. 350, 71–77 (2001)
Dickinson, M.E., Simbuerger, E., Zimmermann, B., Waters, C.W., Fraser, S.E.: Multiphoton excitation spectra in biological samples. J. Biomed. Opt. 8, 329–338 (2003)
Xu, C., Webb, W.W.: Measurement of two-photon excitation cross sections of molecular fluorophores with data from 690 to 1050 nm. J. Opt. Soc. Am. B 13, 481–491 (1996)
Kirkpatrick, S.M., Naik, R.R., Stone, M.O.: Nonlinear saturation and determination of the two-photon absorption cross section of green fluorescent protein. J. Phys. Chem. B. 105, 2867–2873 (2001)
Schönle, A.: Imspector Image Acquisition and Analysis Software, v0.1. http://www.imspector.de (2006)
Clendenon, J.L., Phillips, C.L., Sandoval, R.M., Fang, S., Dunn, K.W.: Voxx: a PC-based, near real-time volume rendering system for biological microscopy. Am. J. Physiol. Cell Physiol. 282, C213–218 (2002)
Baddeley, D., Carl, C., Cremer, C.: 4Pi microscopy deconvolution with a variable point-spread function. Appl. Opt. 45, 7056–7064 (2006)
Lang, M.C., Engelhardt, J., Hell, S.W.: 4Pi microscopy with linear fluorescence excitation. Opt. Lett. 32, 259–261 (2007)
Stracke, F., Weiss, B., Lehr, C.M., Konig, K., Schaefer, U.F., Schneider, M.: Multiphoton microscopy for the investigation of dermal penetration of nanoparticle-borne drugs. J. Invest. Dermatol. 126, 2224–2233 (2006)
Gwinn, M.R., Vallyathan, V.: Nanoparticles: health effects—pros and cons. Environ. Health Perspect. 114, 1818–1825 (2006)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Glaschick, S., Röcker, C., Deuschle, K. et al. Axial Resolution Enhancement by 4Pi Confocal Fluorescence Microscopy with Two-Photon Excitation. J Biol Phys 33, 433–443 (2007). https://doi.org/10.1007/s10867-008-9084-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10867-008-9084-1