Abstract
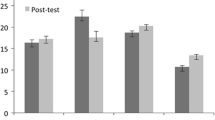
We sought to examine how patients’ treatment decisions incorporate potentially conflicting information from standard clinical indicators (e.g., tumor size) and genomic tests for breast cancer recurrence risk. Participants were 77 early stage breast cancer survivors who previously received genomic testing. They read six hypothetical vignettes that varied recurrence risk indicated by standard tests (low or high risk) coupled with the genomic test (low, intermediate or high risk). For each vignette, women reported their perceived recurrence risk and treatment preferences. Test results indicating high recurrence risk increased perception of risk and preference for chemotherapy (p < .001 for all). Perceived risk explained (i.e., mediated) the effect of test results on chemotherapy preferences. When test results conflicted, women gave more weight to genomic over standard test results. Hypothetical genomic test results had the intended effect of influencing women’s perceptions of recurrence risk and interest in chemotherapy.



Similar content being viewed by others
References
Albain, K. S., Barlow, W. E., Shak, S., Hortobagyi, G. N., Livingston, R. B., Yeh, I. T., et al. (2010). Prognostic and predictive value of the 21-gene recurrence score assay in postmenopausal women with node-positive, oestrogen-receptor-positive breast cancer on chemotherapy: A retrospective analysis of a randomised trial. The lancet Oncology, 11, 55–65.
Baron, R. M., & Kenny, D. A. (1986). The moderator-mediator variable distinction in social psychological research: Conceptual, strategic, and statistical considerations. Journal of Personality and Social Psychology, 51, 1173–1182.
Brewer, N. T., Cuite, C. L., Herrington, J. E., & Weinstein, N. D. (2007). Risk compensation and vaccination: Can getting vaccinated cause people to engage in risky behaviors? Annals of Behavioral Medicine, 34, 95–99.
Brewer, N. T., Edwards, A. S., O’Neill, S. C., Tzeng, J. P., Carey, L. A., & Rimer, B. K. (2009). When genomic and standard test results diverge: Implications for breast cancer patients’ preference for chemotherapy. Breast Cancer Research and Treatment, 117, 25–29.
Dowsett, M., Cuzick, J., Wale, C., Forbes, J., Mallon, E. A., Salter, J., et al. (2010). Prediction of risk of distant recurrence using the 21-gene recurrence score in node-negative and node-positive postmenopausal patients with breast cancer treated with anastrozole or tamoxifen: A TransATAC study. Journal of Clinical Oncology, 28, 1829–1834.
Gianni, L., Zambetti, M., Clark, K., Baker, J., Cronin, M., Wu, J., et al. (2005). Gene expression profiles in paraffin-embedded core biopsy tissue predict response to chemotherapy in women with locally advanced breast cancer. Journal of Clinical Oncology, 23, 7265–7277.
Gurmankin, A. D., Domchek, S., Stopfer, J., Fels, C., & Armstrong, K. (2005). Patients’ resistance to risk information in genetic counseling for BRCA1/2. Archives of Internal Medicine, 165, 523–529.
Guttmacher, A. E., Collins, F. S., Guttmacher, A. E., & Collins, F. S. (2002). Genomic medicine—A primer. New England Journal of Medicine, 347, 1512–1520.
Habel, L. A., Shak, S., Jacobs, M. K., Capra, A., Alexander, C., Pho, M., et al. (2006). A population-based study of tumor gene expression and risk of breast cancer death among lymph node-negative patients. Breast Cancer Research, 8, R25.
Harris, L., Fritsche, H., Mennel, R., Norton, L., Ravdin, P., Taube, S., et al. (2007). American society of clinical oncology 2007 update of recommendations for the use of tumor markers in breast cancer. Journal of Clinical Oncology, 25, 5287–5312.
Lerman, C., Hughes, C., Croyle, R. T., Main, D., Durham, C., Snyder, C., et al. (2000). Prophylactic surgery decisions and surveillance practices one year following BRCA1/2 testing. Preventive Medicine, 31, 75–80.
Lo, S. S., Mumby, P. B., Norton, J., Rychlik, K., Smerage, J., Kash, J., et al. (2010). Prospective multicenter study of the impact of the 21-gene recurrence score assay on medical oncologist and patient adjuvant breast cancer treatment selection. Journal of Clinical Oncology, 28, 1671–1676.
Morris, S. R., & Carey, L. A. (2007). Gene expression profiling in breast cancer. Current Opinion in Oncology, 19, 547–551.
O’Neill, S. C., Brewer, N. T., Lillie, S. E., Morrill, E. F., Dees, E. C., Carey, L. A., et al. (2007). Women’s interest in gene expression analysis for breast cancer recurrence risk. Journal of Clinical Oncology, 25, 4628–4634.
Oratz, R., Kim, B., Chao, C., Skrzypczak, S., Ory, C., Bugarini, R., et al. (2011). Physician survey of the effect of the 21-gene recurrence score assay results on treatment recommendations for patients with lymph node-positive, estrogen receptor-positive breast cancer. Journal of Oncology Practice, 7, 94–99.
Oratz, R., Paul, D., Cohn, A. L., & Sedlacek, S. M. (2007). Impact of a commercial reference laboratory test recurrence score on decision making in early-stage breast cancer. Journal of Oncology Practice, 3, 182–186.
Paik, S. (2007). Development and clinical utility of a 21-gene recurrence score prognostic assay in patients with early breast cancer treated with tamoxifen. The Oncologist, 12, 631–635.
Paik, S., Shak, S., Tang, G., Kim, C., Baker, J., Cronin, M., et al. (2004). A multigene assay to predict recurrence of tamoxifen-treated, node-negative breast cancer. New England Journal of Medicine, 351, 2817–2826.
Paik, S., Tang, G., Shak, S., Kim, C., Baker, J., Kim, W., et al. (2006). Gene expression and benefit of chemotherapy in women with node-negative, estrogen receptor–positive breast cancer. Journal of Clinical Oncology, 24, 3726–3734.
Richman, A. R., Tzeng, J. P., Carey, L. A., Retèl, V. P., & Brewer, N. T. (2011). Knowledge of genomic testing among early-stage breast cancer patients. Psycho-Oncology, 20, 28–35.
Schwartz, L. M., Woloshin, S., Black, W. C., & Welch, H. G. (1997). The role of numeracy in understanding the benefit of screening mammography. Annals of Internal Medicine, 127, 966–972.
Sparano, J. A., & Paik, S. (2008). Development of the 21-gene assay and its application in clinical practice and clinical trials. Journal of Clinical Oncology, 26, 721–728.
Stokes, M. E., Davis, C. S., & Koch, G. G. (2000). Categorical data analysis using the SAS ® system (2nd ed.). Cary: SAS Institute, Inc.
Tzeng, J. P., Mayer, D., Richman, A. R., Lipkus, I., Han, P. K., Valle, C. G., et al. (2010). Women’s experiences with genomic testing for breast cancer recurrence risk. Cancer, 116, 1992–2000.
Van De Vijver, M. J., He, Y. D., Van’t Veer, L. J., Dai, H., Hart, A. A. M., Voskuil, D. W., et al. (2002). A gene-expression signature as a predictor of survival in breast cancer. New England Journal of Medicine, 347, 1999–2009.
Acknowledgments
We are grateful to the physicians and nurses of the University of North Carolina Breast Center for their assistance with this study. Most importantly, we thank the women who participated in this study. We thank Alice Richman and Janice Tzeng for their work on the study. The study received generous financial support from the American Cancer Society (MSRG-06-259-01-CPPB). Jessica T. DeFrank was funded by the UNC Cancer Care and Quality Training Program (NCI R25 Grant, CA116339).
Conflict of interest
The authors have no financial disclosures or conflicts of interest to declare.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
DeFrank, J.T., Carey, L.A. & Brewer, N.T. Understanding how breast cancer patients use risk information from genomic tests. J Behav Med 36, 567–573 (2013). https://doi.org/10.1007/s10865-012-9449-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10865-012-9449-6