Abstract
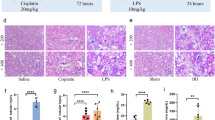
Acute kidney injury (AKI) is a serious complication of sepsis patients, but the pathogenic mechanisms underlying AKI are still largely unclear. In this view, the roles of the key component of N6-methyladenosine (m6A)-wilms tumor 1 associated protein (WTAP) in AKI progression were investigated. AKI mice model was established by using cecal ligation and puncture (CLP). AKI cell model was established by treating HK-2 cells with LPS. Cell apoptosis was analyzed by TdT-mediated dUTP Nick-End Labeling (TUNEL) staining and flow cytometry analysis. Cell viability was analyzed by 3-(4, 5-dimethyl-2-thiazolyl)-2, 5-diphenyl-2-H-tetrazolium bromide (MTT) assay. The concentrations of inflammatory factors were examined with ELISA kits. Reactive oxygen species (ROS), malondialdehyde (MDA), glutathione (GSH) and Fe2+ levels were detected with related kits. Gene expression was detected by western blot assay or quantitative real-time polymerase chain reaction (qRT-PCR) assay. The relation between WTAP and lamin B1 (LMNB1) was verified by Methylated RNA Immunoprecipitation (meRIP) assay, RIP assay, dual-luciferase reporter assay and Actinomycin D assay. CLP induced significant pathological changes in kidney tissues in mice and promoted inflammation, mitochondrial damage and ferroptosis. LMNB1 level was induced in HK-2 cells by LPS. LMNB1 knockdown promoted LPS-mediated HK-2 cell viability and inhibited LPS-mediated HK-2 cell apoptosis, inflammation, mitochondrial damage and ferroptosis. Then, WTAP was demonstrated to promote LMNB1 expression by m6A Methylation modification. Moreover, WTAP knockdown repressed LPS-treated HK-2 cell apoptosis, inflammation, mitochondrial damage and ferroptosis, while LMNB1 overexpression reversed the effects. Additionally, WTAP affected the pathways of NF-κB and JAK2/STAT3 by LMNB1. WTAP-mediated m6A promoted the inflammation, mitochondrial damage and ferroptosis in LPS-induced HK-2 cells by regulating LMNB1 expression and activating NF-κB and JAK2/STAT3 pathways.
Highlights
CLP induces cell apoptosis, inflammation, mitochondrial damage and ferroptosis in mice.
LMNB1 knockdown promotes cell viability, inhibits apoptosis, inflammation, mitochondrial damage and ferroptosis in LPS-treated HK-2 cells.
WTAP promotes LMNB1 expression through m6A Methylation modification.
WTAP-mediated m6A Methylation modification regulates LPS-induced HK-2 cell injury by regulating LMNB1 expression and NF-κB and JAK2/STAT3 pathways.






Similar content being viewed by others
Data availability
No datasets were generated or analysed during the current study.
References
Acute kidney injury (2021) Nat Rev Dis Primers 7:51. https://doi.org/10.1038/s41572-021-00291-0
Borja N, Bivona S, Peart LS, Johnson B, Gonzalez J, Barbouth D, Moore H, Guo S, Undiagnosed Disease N, Bademci G, Tekin M (2022) Genome sequencing reveals novel noncoding variants in PLA2G6 and LMNB1 causing progressive neurologic disease. Mol Genet Genomic Med 10:e1892. https://doi.org/10.1002/mgg3.1892
Cao P, Zhang W, Qiu J, Tang Z, Xue X, Feng T (2023) Gemcitabine inhibits the progression of pancreatic Cancer by restraining the WTAP/MYC chain in an m6A-dependent manner. Cancer Res Treat. https://doi.org/10.4143/crt.2022.1600
Chen X, Li J, Kang R, Klionsky DJ, Tang D (2021) Ferroptosis: machinery and regulation. Autophagy 17:2054–2081. https://doi.org/10.1080/15548627.2020.1810918
Copeland S, Warren HS, Lowry SF, Calvano SE, Remick D (2005) Inflammation, the Host Response to Injury I Acute inflammatory response to endotoxin in mice and humans. Clin Diagn Lab Immunol 12:60–67. https://doi.org/10.1128/CDLI.12.1.60-67.2005
Dillioglugil MO, Kir HM, Demir C, Ilbay G, Sahin D, Dillioglugil O, Bambal G, Mekik H, Ates N (2010) Effect of pentylenetetrazole and sound stimulation induced single and repeated convulsive seizures on the MDA, GSH and NO levels, and SOD activities in rat liver and kidney tissues. Brain Res Bull 83:356–359. https://doi.org/10.1016/j.brainresbull.2010.09.007
Ding X, Hou L, Zhang H, Chen Z, Liu Z, Gong J, Tang Z, Hu R (2022) EIF3C promotes Lung Cancer Tumorigenesis by regulating the APP/HSPA1A/LMNB1 Axis. Dis Markers 2022(9464094). https://doi.org/10.1155/2022/9464094
Farrar A (2018) Acute kidney Injury. Nurs Clin North Am 53:499–510. https://doi.org/10.1016/j.cnur.2018.07.001
Hu Z, Zhang H, Yang SK, Wu X, He D, Cao K, Zhang W (2019) Emerging Role of Ferroptosis in Acute Kidney Injury. Oxid Med Cell Longev 2019:8010614. https://doi.org/10.1155/2019/8010614
Hu C, Zhang B, Zhao S (2023) METTL3-mediated N6-methyladenosine modification stimulates mitochondrial damage and ferroptosis of kidney tubular epithelial cells following acute kidney injury by modulating the stabilization of MDM2-p53-LMNB1 axis. Eur J Med Chem 259:115677. https://doi.org/10.1016/j.ejmech.2023.115677
Huang Y, Zhang L, Liu T, Liang E (2023) LMNB1 targets FOXD1 to promote progression of prostate cancer. Exp Ther Med 26:513. https://doi.org/10.3892/etm.2023.12212
Jia G, Fu Y, Zhao X, Dai Q, Zheng G, Yang Y, Yi C, Lindahl T, Pan T, Yang YG, He C (2011) N6-methyladenosine in nuclear RNA is a major substrate of the obesity-associated FTO. Nat Chem Biol 7:885–887. https://doi.org/10.1038/nchembio.687
Kaushal GP, Shah SV (2014) Challenges and advances in the treatment of AKI. J Am Soc Nephrol 25:877–883. https://doi.org/10.1681/ASN.2013070780
Kozan R, Sare M, Yilmaz TU, Yuksel S, Senes M, Cayci AB, Bozkurt S (2018) Effectiveness of new parameters in the evaluation of pneumoperitoneum-related acute kidney injury in rats. Turk J Med Sci 48:1278–1284. https://doi.org/10.3906/sag-1602-124
Li CM, Li M, Zhao WB, Ye ZC, Peng H (2021) Alteration of N6-Methyladenosine RNA profiles in Cisplatin-Induced Acute kidney Injury in mice. Front Mol Biosci 8:654465. https://doi.org/10.3389/fmolb.2021.654465
Li H, Li C, Zhang B, Jiang H (2023) Lactoferrin suppresses the progression of colon cancer under hyperglycemia by targeting WTAP/m(6)A/NT5DC3/HKDC1 axis. J Transl Med 21:156. https://doi.org/10.1186/s12967-023-03983-1
Liang NN, Zhao Y, Guo YY, Zhang ZH, Gao L, Yu DX, Xu DX, Xu S (2022) Mitochondria-derived reactive oxygen species are involved in renal cell ferroptosis during lipopolysaccharide-induced acute kidney injury. Int Immunopharmacol 107:108687. https://doi.org/10.1016/j.intimp.2022.108687
Liu J, Yue Y, Han D, Wang X, Fu Y, Zhang L, Jia G, Yu M, Lu Z, Deng X, Dai Q, Chen W, He C (2014) A METTL3-METTL14 complex mediates mammalian nuclear RNA N6-adenosine methylation. Nat Chem Biol 10:93–95. https://doi.org/10.1038/nchembio.1432
Liu J, Xia X, Huang P (2020) xCT: a critical molecule that Links Cancer metabolism to Redox Signaling. Mol Ther 28:2358–2366. https://doi.org/10.1016/j.ymthe.2020.08.021
Maekawa H, Inoue T, Ouchi H, Jao TM, Inoue R, Nishi H, Fujii R, Ishidate F, Tanaka T, Tanaka Y, Hirokawa N, Nangaku M, Inagi R (2019) Mitochondrial damage causes inflammation via cGAS-STING signaling in Acute kidney Injury. Cell Rep 29:1261–1273e1266. https://doi.org/10.1016/j.celrep.2019.09.050
Menon S, Symons JM, Selewski DT (2023) Acute kidney Injury. Pediatr Rev 44:265–279. https://doi.org/10.1542/pir.2021-005438
Moore PK, Hsu RK, Liu KD (2018) Management of Acute kidney Injury: Core Curriculum 2018. Am J Kidney Dis 72:136–148. https://doi.org/10.1053/j.ajkd.2017.11.021
Ni L, Yuan C, Wu X (2022) Targeting ferroptosis in acute kidney injury. Cell Death Dis 13:182. https://doi.org/10.1038/s41419-022-04628-9
Ni L, Bai R, Zhou Q, Yuan C, Zhou LT, Wu X (2022a) The correlation between ferroptosis and m6A methylation in patients with acute kidney Injury. Kidney Blood Press Res 47:523–533. https://doi.org/10.1159/000524900
Ping XL, Sun BF, Wang L, Xiao W, Yang X, Wang WJ, Adhikari S, Shi Y, Lv Y, Chen YS, Zhao X, Li A, Yang Y, Dahal U, Lou XM, Liu X, Huang J, Yuan WP, Zhu XF, Cheng T, Zhao YL, Wang X, Rendtlew Danielsen JM, Liu F, Yang YG (2014) Mammalian WTAP is a regulatory subunit of the RNA N6-methyladenosine methyltransferase. Cell Res 24:177–189. https://doi.org/10.1038/cr.2014.3
Schumann U, Shafik A, Preiss T (2016) METTL3 gains R/W Access to the Epitranscriptome. Mol Cell 62:323–324. https://doi.org/10.1016/j.molcel.2016.04.024
Stiekema M, Ramaekers FCS, Kapsokalyvas D, van Zandvoort M, Veltrop RJA, Broers JLV (2021) Super-resolution Imaging of the A- and B-Type Lamin networks: a comparative study of different fluorescence labeling procedures. Int J Mol Sci 22. https://doi.org/10.3390/ijms221910194
Vijayan A (2021) Tackling AKI: prevention, timing of dialysis and follow-up. Nat Rev Nephrol 17:87–88. https://doi.org/10.1038/s41581-020-00390-3
Wan L, Liu J, Huang C, Zhu Z, Li F, Sun G, Wang K, Li S, Ma X, Chen X, Yuan W (2022) Role of m6A modification and novel circ_0066715/ miR-486-5p/ ETS1 axis in rheumatoid arthritis macrophage polarization progression. Aging 14:10009–10026. https://doi.org/10.18632/aging.204439
Wang X, Bi X, Yang K, Huang Y, Liu Y, Zhao J (2020) ROS/p38MAPK-induced lamin B1 accumulation promotes chronic kidney disease-associated vascular smooth muscle cells senescence. Biochem Biophys Res Commun 531:187–194. https://doi.org/10.1016/j.bbrc.2020.07.020
Yu JH, Kim KH, Kim H (2006) Suppression of IL-1beta expression by the Jak 2 inhibitor AG490 in cerulein-stimulated pancreatic acinar cells. Biochem Pharmacol 72:1555–1562. https://doi.org/10.1016/j.bcp.2006.07.008
Yuan X, Bao Q, Lu B, Xiang C, Hou X, Wei W (2023) WTAP affects intracranial aneurysm progression by regulating m6A methylation modification. Open Med (Wars) 18:20230818. https://doi.org/10.1515/med-2023-0818
Zheng G, Dahl JA, Niu Y, Fedorcsak P, Huang CM, Li CJ, Vagbo CB, Shi Y, Wang WL, Song SH, Lu Z, Bosmans RP, Dai Q, Hao YJ, Yang X, Zhao WM, Tong WM, Wang XJ, Bogdan F, Furu K, Fu Y, Jia G, Zhao X, Liu J, Krokan HE, Klungland A, Yang YG, He C (2013) ALKBH5 is a mammalian RNA demethylase that impacts RNA metabolism and mouse fertility. Mol Cell 49:18–29. https://doi.org/10.1016/j.molcel.2012.10.015
Zhou Y, Xu W, Zhu H (2019) CXCL8((3–72)) K11R/G31P protects against sepsis-induced acute kidney injury via NF-kappaB and JAK2/STAT3 pathway. Biol Res 52:29. https://doi.org/10.1186/s40659-019-0236-5
Zhu H, Wang X, Wang X, Liu B, Yuan Y, Zuo X (2020) Curcumin attenuates inflammation and cell apoptosis through regulating NF-kappaB and JAK2/STAT3 signaling pathway against acute kidney injury. Cell Cycle 19:1941–1951. https://doi.org/10.1080/15384101.2020.1784599
Funding
This work was not received funds.
Author information
Authors and Affiliations
Contributions
All authors made substantial contribution to conception and design, acquisition of the data, or analysis and interpretation of the data; take part in drafting the article or revising it critically for important intellectual content; gave final approval of the revision to be published; and agree to be accountable for all aspect of the work.
Corresponding authors
Ethics declarations
Ethical approval
The present study was approved by the ethical review committee of The Eighth Affiliated Hospital, Sun Yat-sen University. Written informed consent was obtained from all enrolled patients.
Consent to participate
Not applicable.
Consent for publication
Patients agree to participate in this work.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Huang, F., Wang, Y., Lv, X. et al. WTAP-mediated N6-methyladenosine modification promotes the inflammation, mitochondrial damage and ferroptosis of kidney tubular epithelial cells in acute kidney injury by regulating LMNB1 expression and activating NF-κB and JAK2/STAT3 pathways. J Bioenerg Biomembr (2024). https://doi.org/10.1007/s10863-024-10015-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10863-024-10015-0