Abstract
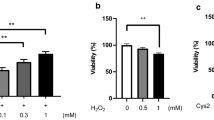
Gualou Xiebai Decoction (GXD), a classic prescription, is widely used to dealing with inflammatory diseases in China for thousands of years. Abnormal metabolic state of bile acids (BAs) is confirmed to cause intestinal epithelial barrier dysfunction. In preliminary work, we observed that GXD could decrease intestinal permeability in hyperlipidemia mice. The present study aimed to explore the protective effect of GXD on intestinal mucosa in vitro. Caco-2 cell monolayer permeability among different groups was determined by measuring the concentrations of FITC-dextran in the lower compartments and transepithelial electrical resistance (TEER). Meanwhile, mRNA and protein expressions of tight junctions (TJs) were investigated. Generation of intracellular reactive oxygen species (ROS) and the ratio of cell apoptosis induced by BAs were assessed by fluorescence probe and flow cytometry. GXD was shown to keep the cell monolayer in low permeable status, increase TEER and mRNA and protein expressions of occludin (Ocln) and zonula occluden 2 (ZO2) remarkably in cells challenged with cholic acid (CA), deoxycholic acid (DCA) and glycocholic acid (GCA). However, no significant effects were uncovered against the pathological effects of taurocholic acid (TCA). Meanwhile, generation of ROS and increased levels of apoptotic cells caused by CA, DCA and GCA were dramatically decreased by GXD, which were not observed on TCA. GXD could significantly attenuate intestinal barrier dysfunction induced by BAs via TJs regulation, oxidative stress suppression and cell apoptosis decrease, but such effects and behind mechanisms differed among different kinds of BAs.









Similar content being viewed by others
Data availability
The datasets used or analysed during the current study are available from the corresponding author on reasonable request.
Code availability
Statistical analysis was carried out with GraphPad Prism 7 and IBM SPSS 20.0 software.
References
Ajouz H, Mukherji D, Shamseddine A (2014) Secondary bile acids: an underrecognized cause of colon cancer[J]. World J Surg Oncol 12:164. https://doi.org/10.1186/1477-7819-12-164
Allam-Ndoul B, Castonguay-Paradis S, Veilleux A (2020) Gut microbiota and intestinal trans-epithelial permeability[J]. Int J Mol Sci 21(17):6402. https://doi.org/10.3390/ijms21176402
Araki Y, Fujiyama Y, Andoh A et al (2001) Hydrophilic and hydrophobic bile acids exhibit different cytotoxicities through cytolysis, interleukin-8 synthesis and apoptosis in the intestinal epithelial cell lines. IEC-6 and Caco-2 cells[J]. Scand J Gastroenterol 36(5):533–9. https://doi.org/10.1080/003655201750153430
Araki Y, Katoh T, Ogawa A et al (2005) Bile acid modulates transepithelial permeability via the generation of reactive oxygen species in the Caco-2 cell line[J]. Free Radic Biol Med 39(6):769–80. https://doi.org/10.1016/j.freeradbiomed.2005.04.026
Bernstein C, Bernstein H, Garewal H et al (1999) A bile acid-induced apoptosis assay for colon cancer risk and associated quality control studies[J]. Cancer Res 59(10):2353–7
Bhardwaj V, Gokulan RC, Horvat A et al (2017) Activation of NADPH oxidases leads to DNA damage in esophageal cells[J]. Sci Rep 7(1):9956. https://doi.org/10.1038/s41598-017-09620-4
Camilleri M (2018) Management options for irritable bowel syndrome[J]. Mayo Clin Proc 93(12):1858–1872. https://doi.org/10.1016/j.mayocp.2018.04.032
Cardoso-Silva D, Delbue D, Itzlinger A et al (2019) Intestinal barrier function in gluten-related disorders[J]. Nutrients 11(10):2325. https://doi.org/10.3390/nu11102325
Chen X, Oshima T, Shan J et al (2012) Bile salts disrupt human esophageal squamous epithelial barrier function by modulating tight junction proteins[J]. Am J Physiol Gastrointest Liver Physiol 303(2):G199-208. https://doi.org/10.1152/ajpgi.00454.2011
Chi X, Zhao X, Wang W et al (2017) Fusion expression of Occludin extracellular loops and an α-helical bundle: a new research model for tight junction[J]. PLoS One 12(4):e0175516. https://doi.org/10.1371/journal.pone.0175516
Daenen K, Andries A, Mekahli D et al (2019) Oxidative stress in chronic kidney disease[J]. Pediatr Nephrol 34(6):975–991. https://doi.org/10.1007/s00467-018-4005-4
Di Ciaula A, Garruti G, LunardiBaccetto R et al (2017) Bile acid physiology[J]. Ann Hepatol 16(Suppl. 1: s3-105):s4–s14. https://doi.org/10.5604/01.3001.0010.5493
Erlejman AG, Fraga CG, Oteiza PI (2006) Procyanidins protect Caco-2 cells from bile acid- and oxidant-induced damage[J]. Free Radic Biol Med 41(8):1247–56. https://doi.org/10.1016/j.freeradbiomed.2006.07.002
Gupta B, Liu Y, Chopyk DM et al (2020) Western diet-induced increase in colonic bile acids compromises epithelial barrier in nonalcoholic steatohepatitis[J]. FASEB J 34(5):7089–7102. https://doi.org/10.1096/fj.201902687R
He C, Deng J, Hu X et al (2019) Vitamin A inhibits the action of LPS on the intestinal epithelial barrier function and tight junction proteins[J]. Food Funct 10(2):1235–1242. https://doi.org/10.1039/c8fo01123k
Hegyi P, Maléth J, Walters JR et al (2018) Guts and Gall: Bile acids in regulation of intestinal epithelial function in health and disease[J]. Physiol Rev 98(4):1983–2023. https://doi.org/10.1152/physrev.00054.2017
Hua C, Geng Y, Chen Q et al (2018) Effects of chronic dexamethasone exposure on bile acid metabolism and cecal epithelia function in goats[J]. Domest Anim Endocrinol 65:9–16. https://doi.org/10.1016/j.domaniend.2018.05.005
Ibrahim S, Dayoub R, Krautbauer S et al (2019) Bile acid-induced apoptosis and bile acid synthesis are reduced by over-expression of Augmenter of Liver Regeneration (ALR) in a STAT3-dependent mechanism[J]. Exp Cell Res 374(1):189–197. https://doi.org/10.1016/j.yexcr.2018.11.023
Ignacio Barrasa J, Olmo N, Perez-Ramos P et al (2011) Deoxycholic and chenodeoxycholic bile acids induce apoptosis via oxidative stress in human colon adenocarcinoma cells[J]. Apoptosis 16(10):1054–67. https://doi.org/10.1007/s10495-011-0633-x
Jean-Louis S, Akare S, Ali MA et al (2006) Deoxycholic acid induces intracellular signaling through membrane perturbations[J]. J Biol Chem 281(21):14948–60. https://doi.org/10.1074/jbc.M506710200
Jiao C, Chen W, Tan X et al (2020) Ganoderma lucidum spore oil induces apoptosis of breast cancer cells in vitro and in vivo by activating caspase-3 and caspase-9[J]. J Ethnopharmacol 247:112256. https://doi.org/10.1016/j.jep.2019.112256
Kiraz Y, Adan A, KartalYandim M et al (2016) Major apoptotic mechanisms and genes involved in apoptosis[J]. Tumour Biol 37(7):8471–86. https://doi.org/10.1007/s13277-016-5035-9
Lee B, Moon KM, Kim CY (2018) Tight junction in the intestinal epithelium: its association with diseases and regulation by phytochemicals[J]. J Immunol Res 2018:2645465. https://doi.org/10.1155/2018/2645465
Li C, Zhang WY, Yu Y et al (2019) Discovery of the mechanisms and major bioactive compounds responsible for the protective effects of Gualou Xiebai Decoction on coronary heart disease by network pharmacology analysis[J]. Phytomedicine 56:261–268. https://doi.org/10.1016/j.phymed.2018.11.010
Miyoshi Y, Tanabe S, Suzuki T (2016) Cellular zinc is required for intestinal epithelial barrier maintenance via the regulation of claudin-3 and occludin expression[J]. Am J Physiol Gastrointest Liver Physiol 311(1):G105-16. https://doi.org/10.1152/ajpgi.00405.2015
Moser LA, Carter M, Schultz-Cherry S (2007) Astrovirus increases epithelial barrier permeability independently of viral replication[J]. J Virol 81(21):11937–45. https://doi.org/10.1128/jvi.00942-07
Mroz MS, Lajczak NK, Goggins BJ et al (2018) The bile acids, deoxycholic acid and ursodeoxycholic acid, regulate colonic epithelial wound healing[J]. Am J Physiol Gastrointest Liver Physiol 314(3):G378-g387. https://doi.org/10.1152/ajpgi.00435.2016
Mueller M, Castro RE, Thorell A et al (2018) Ursodeoxycholic acid: Effects on hepatic unfolded protein response, apoptosis and oxidative stress in morbidly obese patients[J]. Liver Int 38(3):523–531. https://doi.org/10.1111/liv.13562
Mukaisho KI, Kanai S, Kushima R et al (2019) Barretts’s carcinogenesis[J]. Pathol Int 69(6):319–330. https://doi.org/10.1111/pin.12804
Pavlidis P, Powell N, Vincent RP et al (2015) Systematic review: bile acids and intestinal inflammation-luminal aggressors or regulators of mucosal defence?[J]. Aliment Pharmacol Ther 42(7):802–17. https://doi.org/10.1111/apt.13333
Rohr MW, Narasimhulu CA, Rudeski-Rohr TA et al (2020) Negative effects of a high-fat diet on intestinal permeability: a review[J]. Adv Nutr 11(1):77–91. https://doi.org/10.1093/advances/nmz061
Sarathy J, Detloff SJ, Ao M et al (2017) The Yin and Yang of bile acid action on tight junctions in a model colonic epithelium[J]. Physiol Rep 5(10):e13294. https://doi.org/10.14814/phy2.13294
Schölmerich J, Becher MS, Schmidt K et al (1984) Influence of hydroxylation and conjugation of bile salts on their membrane-damaging properties–studies on isolated hepatocytes and lipid membrane vesicles[J]. Hepatology 4(4):661–6. https://doi.org/10.1002/hep.1840040416
Staley C, Weingarden AR, Khoruts A et al (2017) Interaction of gut microbiota with bile acid metabolism and its influence on disease states[J]. Appl Microbiol Biotechnol 101(1):47–64. https://doi.org/10.1007/s00253-016-8006-6
Suzuki T (2020) Regulation of the intestinal barrier by nutrients: the role of tight junctions[J]. Anim Sci J 91(1):e13357. https://doi.org/10.1111/asj.13357
Suzuki T, Hara H (2010) Dietary fat and bile juice, but not obesity, are responsible for the increase in small intestinal permeability induced through the suppression of tight junction protein expression in LETO and OLETF rats[J]. Nutr Metab (Lond) 7:19. https://doi.org/10.1186/1743-7075-7-19
Tang VW, Goodenough DA (2003) Paracellular ion channel at the tight junction[J]. Biophys J 84(3):1660–73. https://doi.org/10.1016/s0006-3495(03)74975-3
Wang F, Lu Z, Wang X et al (2018) Impaired vagus function in rats suppresses bile acid synthesis in the liver by disrupting tight junctions and activating Fxr-Fgf15 signaling in the intestine[J]. Biochem Biophys Res Commun 495(1):1490–1496. https://doi.org/10.1016/j.bbrc.2017.11.201
Wang Z, Litterio MC, Müller M et al (2020) (-)-Epicatechin and NADPH oxidase inhibitors prevent bile acid-induced Caco-2 monolayer permeabilization through ERK1/2 modulation[J]. Redox Biol 28:101360. https://doi.org/10.1016/j.redox.2019.101360
Xia Z, Han Y, Wang K et al (2017) Oral administration of propionic acid during lactation enhances the colonic barrier function[J]. Lipids Health Dis 16(1):62. https://doi.org/10.1186/s12944-017-0452-3
Yan LL, Zhang WY, Wei XH et al (2018) Gualou Xiebai Decoction, a traditional Chinese medicine, prevents cardiac reperfusion injury of hyperlipidemia rat via energy modulation[J]. Front Physiol 9:296. https://doi.org/10.3389/fphys.2018.00296
Zhang Y, Csanaky IL, Lehman-McKeeman LD et al (2011) Loss of organic anion transporting polypeptide 1a1 increases deoxycholic acid absorption in mice by increasing intestinal permeability[J]. Toxicol Sci 124(2):251–60. https://doi.org/10.1093/toxsci/kfr236
Zhang R, Ray JW, Jain MK et al (2017) Ileectomy-induced Bile Overaccumulation in Mouse Intestine[J]. J Vis Exp (126):55728. https://doi.org/10.3791/55728
Funding
The study was supported by the Military Science Foundation of China (no. CLB18J036), Fujian Natural Science Foundation (no. 2018J01354) and Clinical Research Fund of 900TH Hospital of PLA (no. 2020L08).
Author information
Authors and Affiliations
Contributions
Jiyuan Su and Dongmei Zhang contributed to the conception of the study;
Jiyuan Su and Zhiqing He performed the experiment;
Jiyuan Su, Zhiqing He, Zonggui Wu and Dongmei Zhang contributed significantly to analysis and manuscript preparation;
Jiyuan Su and Zhiqing He performed the data analyses and wrote the manuscript;
Yunhua Yu and Mingfang Lu helped perform the analysis with constructive discussions.
Corresponding authors
Ethics declarations
Conflicts of interest/Competing interests
The authors declare that they have no conflicts of interest.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Su, J., He, Z., Yu, Y. et al. Gualou Xiebai Decoction ameliorates increased Caco-2 monolayer permeability induced by bile acids via tight junction regulation, oxidative stress suppression and apoptosis reduction. J Bioenerg Biomembr 54, 45–57 (2022). https://doi.org/10.1007/s10863-021-09927-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10863-021-09927-y