Abstract
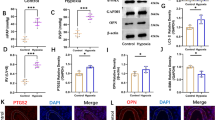
Acute myocardial infarction (AMI) is the main cause of death in the whole world. This study aimed to investigate whether forkhead box O4 (FoxO4) could negatively modulate ubiquitin specific peptidase 10 (USP10) transcription to aggravate the apoptosis and oxidative stress of hypoxia/reoxygenation (H/R)-induced cardiomyocytes through Hippo/YAP pathway. mRNA expression as well as protein expressions of USP10 and FoxO4 in H9C2 cells after H/R induction or transfection were respectively detected by Reverse transcription-quantitative (RT-q) PCR analysis and Western blot. The viability and apoptosis of H9C2 cells after H/R induction or transfection were respectively detected by CCK-8 and TUNEL assays. The expressions of lactate dehydrogenase (LDH), superoxide dismutase (SOD), glutathione peroxidase (GSH-Px) and malondialdehyde (MDA) in H9C2 cells after H/R induction or transfection were analyzed using appropriate kits and intracellular reactive oxygen species (ROS) levels were detected using a ROS Assay Kit. Dual luciferase reporter assay and Chromatin Immunoprecipitation (ChIP) have adopted to confirm the combination of USP10 and FoxO4. Western blot was also used to analyze the expression of apoptosis-related proteins and Hippo/YAP pathway-related proteins. As a result, USP10 expression was decreased in H/R-induced H9C2 cells in a time-dependent manner. USP10 overexpression increased the viability and suppressed the apoptosis and oxidative stress of H/R-induced H9C2 cells. In addition, FoxO4 modulated USP10 transcription. FoxO4 expression was increased in H9C2 cells induced by H/R. FoxO4 overexpression could reverse the protective effects of USP10 overexpression on H/R-induced H9C2 cells by regulating the Hippo/YAP signaling pathway. In conclusion, FoxO4 negatively modulated USP10 transcription to aggravate the apoptosis and oxidative stress of H/R-induced H9C2 cells via blocking Hippo/YAP pathway.







Similar content being viewed by others
Data availability
Data will be available on reasonable request.
References
Ambrosio G, Tritto I (1999) Reperfusion injury: experimental evidence and clinical implications. Am Heart J 138:S69-75. https://doi.org/10.1016/s0002-8703(99)70323-6
Cai B et al. (2016) Long noncoding RNA H19 mediates melatonin inhibition of premature senescence of c-kit(+) cardiac progenitor cells by promoting miR-675 Journal of pineal research 61:82–95 doi:https://doi.org/10.1111/jpi.12331
Chen B, Wu Z, Xu J, Xu Y (2015) Calreticulin Binds to Fas Ligand and Inhibits Neuronal Cell Apoptosis Induced by Ischemia-Reperfusion Injury Biomed Res Int 2015:895284. https://doi.org/10.1155/2015/895284
Del Re DP, Amgalan D, Linkermann A, Liu Q, Kitsis RN (2019) Fundamental Mechanisms of Regulated Cell Death and Implications for Heart Disease. Physiological Reviews 99:1765–1817. https://doi.org/10.1152/physrev.00022.2018
Fornes O et al. (2020) JASPAR 2020: update of the open-access database of transcription factor binding profiles. Nucleic Acids Res 48:D87-d92. https://doi.org/10.1093/nar/gkz1001
Gao Y et al. (2019) Usp10 modulates the hippo pathway by deubiquitinating and stabilizing the transcriptional coactivator Yorkie. Int J Mol Sci 20:6013 https://doi.org/10.3390/ijms20236013
Ge P, Luo Y, Liu CL, Hu B (2007) Protein aggregation and proteasome dysfunction after brain ischemia. Stroke 38:3230–3236. https://doi.org/10.1161/strokeaha.107.487108
Gong P et al (2019) Hippo/YAP signaling pathway mitigates blood-brain barrier disruption after cerebral ischemia/reperfusion injury. Behav Brain Res 356:8–17. https://doi.org/10.1016/j.bbr.2018.08.003
Green DR, Kroemer G (2004) The pathophysiology of mitochondrial cell death. Science 305:626–629. https://doi.org/10.1126/science.1099320
Hausenloy DJ et al (2017) Targeting reperfusion injury in patients with ST-segment elevation myocardial infarction: trials and tribulations. Eur Heart J 38:935–941. https://doi.org/10.1093/eurheartj/ehw145
Heallen T, Morikawa Y, Leach J, Tao G, Willerson JT, Johnson RL, Martin JF (2013) hippo signaling impedes adult heart regeneration. Development 140:4683–4690. https://doi.org/10.1242/dev.102798
Heallen T, Zhang M, Wang J, Bonilla-Claudio M, Klysik E, Johnson RL, Martin JF (2011) Hippo pathway inhibits Wnt signaling to restrain cardiomyocyte proliferation and heart size. Science 332:458–461. https://doi.org/10.1126/science.1199010
Heusch Gerd (2013) Cardioprotection: chances and challenges of its translation to the clinic. Lancet 381(9861):166–175. https://doi.org/10.1016/S0140-6736(12)60916-7
Hong G, Yan Y, Zhong Y, Chen J, Tong F, Ma Q (2019) combined ischemic preconditioning and resveratrol improved bloodbrain barrier breakdown via Hippo/YAP/TAZ signaling pathway. CNS Neurol Disord Drug Targets 18:713–722. https://doi.org/10.2174/1871527318666191021144126
Huang H, Tindall DJ (2007) Dynamic FoxO Transcription Factors J Cell Sci 120:2479–2487. https://doi.org/10.1242/jcs.001222
Huang Jianbin, Shian Wu, Barrera Jose, Matthews Krista, Pan Duojia (2005) The Hippo signaling pathway coordinately regulates cell proliferation and apoptosis by inactivating Yorkie, the Drosophila Homolog of YAP. Cell 122(3):421–434. https://doi.org/10.1016/j.cell.2005.06.007
Ibanez B et al. (2018) 2017 ESC Guidelines for the management of acute myocardial infarction in patients presenting with ST-segment elevation. Eur Heart J 39(2):119–177. https://doi.org/10.1093/eurheartj/ehx393
Jiangqiao Z, Tianyu W, Zhongbao C, Long Z, Jilin Z, Xiaoxiong M, Tao Q (2020) Ubiquitin-Specific Peptidase 10 Protects Against Hepatic Ischaemic/Reperfusion Injury via TAK1 Signalling. Front Immunol 11:506275. https://doi.org/10.3389/fimmu.2020.506275
Jochemsen AG, Shiloh Y (2010) USP10: friend and foe. Cell 140(3):308–310. https://doi.org/10.1016/j.cell.2010.01.034
Jones Steven P, Trocha Steven D, Lefer David J (2001) Cardioprotective actions of endogenous IL-10 are independent of iNOS. Am J Physiol-Heart Circ Physiol 281(1):H48–H52. https://doi.org/10.1152/ajpheart.2001.281.1.H48
Kalogeris T, Baines CP, Krenz M, Korthuis RJ (2012) Cell biology of ischemia/reperfusion injury. Int Rev Cell Mol Biol 298:229–317. https://doi.org/10.1016/b978-0-12-394309-5.00006-7
Kalogeris T, Bao Y, Korthuis RJ (2014) Mitochondrial reactive oxygen species: a double edged sword in ischemia/reperfusion vs preconditioning. Redox Biol 2:702–714. https://doi.org/10.1016/j.redox.2014.05.006
Kattoor AJ, Pothineni NVK, Palagiri D, Mehta JL (2017) Oxidative stress in atherosclerosis. Curr Atheroscler Rep 19:42. https://doi.org/10.1007/s11883-017-0678-6
Kim J et al (2017) YAP/TAZ regulates sprouting angiogenesis and vascular barrier maturation. J Clin Invest 127:3441–3461. https://doi.org/10.1172/jci93825
Kumar D, Jugdutt BI (2003) Apoptosis and oxidants in the heart. J Lab Clin Med 142:288–297. https://doi.org/10.1016/s0022-2143(03)00148-3
Liu Hao, Wang Lei, Weng Xiaodong, Chen Hui, Yang Du, Diao Changhui, Chen Zhiyuan, Liu Xiuheng (2019) Inhibition of Brd4 alleviates renal ischemia/reperfusion injury-induced apoptosis and endoplasmic reticulum stress by blocking FoxO4-mediated oxidative stress. Redox Biology 24:101195. https://doi.org/10.1016/j.redox.2019.101195
Livak Kenneth J, Schmittgen Thomas D (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods 25(4):402–408. https://doi.org/10.1006/meth.2001.1262
Ma LY, Chen WW, Gao RL, Liu LS, Hu SS (2020) China cardiovascular diseases report 2018: an updated summary. J Geriatr Cardiol 17:1–8
Ma Shuxian, Dong Zhenming (2019) Melatonin attenuates cardiac reperfusion stress by Improving OPA1-Related mitochondrial fusion in a Yap–Hippo pathway–dependent manner. J Cardiovasc Pharmacol 73(1):27–39. https://doi.org/10.1097/FJC.0000000000000626
Meier P, Finch A, Evan G (2000) Apoptosis in development. Nature 407:796–801. https://doi.org/10.1038/35037734
Mia Masum M, Chelakkot-Govindalayathil Ayshwarya L, Singh Manvendra K (2016) Targeting NF2-Hippo/Yap signaling pathway for cardioprotection after ischemia/reperfusion injury. Ann Translat Med 4(24):545–545. https://doi.org/10.21037/atm.2016.11.85
Nishihara Masahiro, Miura Tetsuji, Miki Takayuki, Tanno Masaya, Yano Toshiyuki, Naitoh Kazuyuki, Ohori Katsuhiko, Hotta Hiroyuki, Terashima Yoshiaki, Shimamoto Kazuaki (2007) Modulation of the mitochondrial permeability transition pore complex in GSK-3β-mediated myocardial protection. J Molec Cell Cardiol 43(5):564–570. https://doi.org/10.1016/j.yjmcc.2007.08.010
Protection of rats spinal cord ischemia-reperfusion injury by inhibition of MiR-497 on inflammation and apoptosis: Possible role in pediatrics (2016). Biomed Pharmacotherapy 81:337–344
Singh A et al (2016) Hippo signaling mediators Yap and Taz are required in the Epicardium for coronary vasculature development. Cell Rep 15:1384–1393. https://doi.org/10.1016/j.celrep.2016.04.027
Soncini Chiara, Berdo Ingrid, Draetta Giulio (2001) Ras–GAP SH3 domain binding protein (G3BP) is a modulator of USP10, a novel human ubiquitin specific protease. Oncogene 20(29):3869–3879. https://doi.org/10.1038/sj.onc.1204553
Tian Li, Cao Wenjiang, Yue Rujing, Yuan Yong, Guo Xinhong, Qin Dongmei, Xing Jianguo, Wang Xinchun (2019) Pretreatment with Tilianin improves mitochondrial energy metabolism and oxidative stress in rats with myocardial ischemia/reperfusion injury via AMPK/SIRT1/PGC-1 alpha signaling pathway. J Pharmacol Sci 139(4):352–360. https://doi.org/10.1016/j.jphs.2019.02.008
van der Horst A, Burgering BM (2007) Stressing the role of FoxO proteins in lifespan and disease. Nat Rev Mol Cell Biol 8:440–450. https://doi.org/10.1038/nrm2190
Wang Lei, Dongchuan Wu, Zongrong Xu (2019) USP10 protects against cerebral ischemia injury by suppressing inflammation and apoptosis through the inhibition of TAK1 signaling. Biochem Biophysic Res Commu 516(4):1272–1278. https://doi.org/10.1016/j.bbrc.2019.06.042
Wang Z, Shen X, He X, Ge Z, Cardiology DO, Hospital T (2017) Effect of exercises with sports bracelet combined with WeChat-based education on acute myocardial infarction patients. J Nurs Sci
Yang M, Chen J, Zhao J, Meng M (2014) Etanercept attenuates myocardial ischemia/reperfusion injury by decreasing inflammation and oxidative stress. PLoS One 9:e108024. https://doi.org/10.1371/journal.pone.0108024
Yu L et al (2018) FoxO4 promotes myocardial ischemia-reperfusion injury: the role of oxidative stress-induced apoptosis. Am J Transl Res 10:2890–2900
Yuan J, Luo K, Zhang L, Cheville JC, Lou Z (2010) USP10 regulates p53 localization and stability by deubiquitinating p53. Cell 140:384–396. https://doi.org/10.1016/j.cell.2009.12.032
Zhang X (2020) Effect of collaborative care mode on self-care ability and cardiac function in patients with acute myocardial infarction with PCI Chinese General Practice Nursing 18:4605-4607
Zhao Bin, Tumaneng Karen, Guan Kun-Liang (2011) The Hippo pathway in organ size control, tissue regeneration and stem cell self-renewal. Nat Cell Biol 13(8):877–883. https://doi.org/10.1038/ncb2303
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Huang, J., Liu, Y., Wang, M. et al. FoxO4 negatively modulates USP10 transcription to aggravate the apoptosis and oxidative stress of hypoxia/reoxygenation-induced cardiomyocytes by regulating the Hippo/YAP pathway. J Bioenerg Biomembr 53, 541–551 (2021). https://doi.org/10.1007/s10863-021-09910-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10863-021-09910-7