Abstract
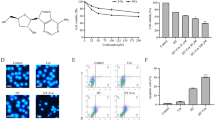
The aim of this study was to determine the effects of hyperthermia, cisplatin and their combination on mitochondrial functions such as glutamate dehydrogenase (GDH) activity and mitochondrial respiration rates, as well as survival of cultured ovarian adenocarcinoma OVCAR-3 cells. Cells treated for 1 h with hyperthermia (40 and 43 °C) or cisplatin (IC50) or a combination of both treatments were left for recovery at 37 °C temperature for 24 h or 48 h. The obtained results revealed that 43 °C hyperthermia potentiated effects of cisplatin treatment: combinatory treatment more strongly suppressed GDH activity and expression, mitochondrial functions, and decreased survival of OVCAR-3 cells in comparison to separate single treatments. We obtained evidence that in the OVCAR-3 cell line GDH was directly activated by hyperthermia (cisplatin eliminated this effect); however, this effect was followed by GDH inhibition after 48 h recovery. A combination of 43 °C hyperthermia with cisplatin induced stronger GDH inhibition in comparison to separate treatments, and negative effects exerted on GDH activity correlated with suppression of mitochondrial respiration with glutamate + malate. Cisplatin did not induce uncoupling of oxidative phosphorylation in OVCAR-3 cells but induced impairment of the outer mitochondrial membrane in combination with 43 °C hyperthermia. Hyperthermia (43 °C) potentiated cytotoxicity of cisplatin in an OVCAR-3 cell line.





Similar content being viewed by others
References
Antanaviciute I, Mildaziene V, Stankevicius E, Herdegen T, Skeberdis VA (2014) Hyperthermia differently affects connexin43 expression and gap junction permeability in skeletal myoblasts and HeLa cells Mediat Inflamm 2014:748290 https://doi.org/10.1155/2014/748290, 1, 16
Bakrin N, Classe JM, Pomel C, Gouy S, Chene G, Glehen O (2014) Hyperthermic intraperitoneal chemotherapy (HIPEC) in ovarian cancer. J Visc Surg 151:347–353. https://doi.org/10.1016/j.jviscsurg.2014.07.005
Choi YM, Kim HK, Shim W, Anwar MA, Kwon JW, Kwon HK, Kim HJ, Jeong H, Kim HM, Hwang D, Kim HS, Choi S (2015) Mechanism of cisplatin-induced cytotoxicity is correlated to impaired metabolism due to mitochondrial ROS generation. PLoS One 10:e0135083. https://doi.org/10.1371/journal.pone.0135083
Dar S, Chhina J, Mert I, Chitale D, Buekers T, Kaur H, Giri S, Munkarah A, Rattan R (2017) Bioenergetic adaptations in chemoresistant ovarian cancer cells. Sci Rep 7:8760. https://doi.org/10.1038/s41598-017-09206-0
Di Giorgio A et al (2017) Cytoreduction (Peritonectomy procedures) combined with Hyperthermic intraperitoneal chemotherapy (HIPEC) in advanced ovarian Cancer: retrospective Italian multicenter observational study of 511 cases. Ann Surg Oncol 24:914–922. https://doi.org/10.1245/s10434-016-5686-1
Dier U, Shin DH, Hemachandra LP, Uusitalo LM, Hempel N (2014) Bioenergetic analysis of ovarian cancer cell lines: profiling of histological subtypes and identification of a mitochondria-defective cell line. PLoS One 9:e98479. https://doi.org/10.1371/journal.pone.0098479
Frezza C, Gottlieb E (2009) Mitochondria in cancer: not just innocent bystanders. Semin Cancer Biol 19:4–11. https://doi.org/10.1016/j.semcancer.2008.11.008
Hsu PP, Sabatini DM (2008) Cancer cell metabolism: Warburg and beyond. Cell 134:703–707. https://doi.org/10.1016/j.cell.2008.08.021
Jarmuszkiewicz W, Woyda-Ploszczyca A, Koziel A, Majerczak J, Zoladz JA (2015) Temperature controls oxidative phosphorylation and reactive oxygen species production through uncoupling in rat skeletal muscle mitochondria. Free Radic Biol Med 83:12–20. https://doi.org/10.1016/j.freeradbiomed.2015.02.012
Ji ZH, Peng KW, Li Y (2016) Intraperitoneal free cancer cells in gastric cancer: pathology of peritoneal carcinomatosis and rationale for intraperitoneal chemotherapy/hyperthermic intraperitoneal chemotherapy in gastric cancer. Translational Gastroenterology and Hepatology 1:69. https://doi.org/10.21037/tgh.2016.08.03
Jin L, Li D, Alesi GN, Fan J, Kang HB, Lu Z, Boggon TJ, Jin P, Yi H, Wright ER, Duong D, Seyfried NT, Egnatchik R, DeBerardinis RJ, Magliocca KR, He C, Arellano ML, Khoury HJ, Shin DM, Khuri FR, Kang S (2015) Glutamate dehydrogenase 1 signals through antioxidant glutathione peroxidase 1 to regulate redox homeostasis and tumor growth. Cancer Cell 27:257–270. https://doi.org/10.1016/j.ccell.2014.12.006
Jin L, Alesi GN, Kang S (2016) Glutaminolysis as a target for cancer therapy. Oncogene 35:3619–3625. https://doi.org/10.1038/onc.2015.447
Jozwiak Z, Leyko W (1992) Role of membrane components in thermal injury of cells and development of thermotolerance. Int J Radiat Biol 62:743–756
Jung Y, Lippard SJ (2007) Direct cellular responses to platinum-induced DNA damage. Chem Rev 107:1387–1407. https://doi.org/10.1021/cr068207j
Lee WK, Shin S, Cho SS, Park JS (1999) Purification and characterization of glutamate dehydrogenase as another isoprotein binding to the membrane of rough endoplasmic reticulum. J Cell Biochem 76:244–253
Liu G, Zhu J, Yu M, Cai C, Zhou Y, Yu M, Fu Z, Gong Y, Yang B, Li Y, Zhou Q, Lin Q, Ye H, Ye L, Zhao X, Li Z, Chen R, Han F, Tang C, Zeng B (2015) Glutamate dehydrogenase is a novel prognostic marker and predicts metastases in colorectal cancer patients. J Transl Med 13:144. https://doi.org/10.1186/s12967-015-0500-6
Lukosiute-Urboniene A, Jasukaitiene A, Silkuniene G, Barauskas V, Gulbinas A, Dambrauskas Z (2019) Human antigen R mediated post-transcriptional regulation of inhibitors of apoptosis proteins in pancreatic cancer. World J Gastroenterol 25:205–219. https://doi.org/10.3748/wjg.v25.i2.205
Mirandola L, JC M, Cobos E, Bernardini G, Jenkins MR, Kast WM, Chiriva-Internati M (2011) Cancer testis antigens: novel biomarkers and targetable proteins for ovarian cancer. Int Rev Immunol 30:127–137. https://doi.org/10.3109/08830185.2011.572504
Nauciene Z, Zukiene R, Degutyte-Fomins L, Mildaziene V (2012) Mitochondrial membrane barrier function as a target of hyperthermia. Medicina (Kaunas) 48:249–255
Plaitakis A, Zaganas I (2001) Regulation of human glutamate dehydrogenases: implications for glutamate, ammonia and energy metabolism in brain. J Neurosci Res 66:899–908. https://doi.org/10.1002/jnr.10054
Roti Roti JL (2008) Cellular responses to hyperthermia (40-46 degrees C): cell killing and molecular events. Int J Hyperth 24:3–15. https://doi.org/10.1080/02656730701769841
Shashidharan P, Clarke DD, Ahmed N, Moschonas N, Plaitakis A (1997) Nerve tissue-specific human glutamate dehydrogenase that is thermolabile and highly regulated by. ADP J Neurochem 68:1804–1811
Silkuniene G, Zukiene R, Nauciene Z, Degutyte-Fomins L, Mildaziene V (2018) Impact of gender and age on hyperthermia-induced changes in respiration of liver mitochondria. Medicina (Kaunas) 54. https://doi.org/10.3390/medicina54040062
Sözgen K, Cekic SD, Tutem E, Apak R (2006) Spectrophotometric total protein assay with copper(II)-neocuproine reagent in alkaline medium Talanta 68:1601–1609. https://doi.org/10.1016/j.talanta.2005.08.043
Sugarbaker PH, Van der Speeten K (2016) Surgical technology and pharmacology of hyperthermic perioperative chemotherapy. J Gastrointest Oncol 7:29–44. https://doi.org/10.3978/j.issn.2078-6891.2015.105
Sukovas A et al (2017) Response of OVCAR-3 cells to cisplatin and hyperthermia: does hyperthermia really matter? Anticancer Res 37:5011–5018. https://doi.org/10.21873/anticanres.11915
Trumbeckaite S, Cesna V, Jasukaitiene A, Baniene R, Gulbinas A (2018) Different mitochondrial response to cisplatin and hyperthermia treatment in human AGS, Caco-2 and T3M4 cancer cell lines. J Bioenerg Biomembr 50:329–338. https://doi.org/10.1007/s10863-018-9764-x
Tsujimoto Y, Nakagawa T, Shimizu S (2006) Mitochondrial membrane permeability transition and cell death. Biochim Biophys Acta 1757:1297–1300. https://doi.org/10.1016/j.bbabio.2006.03.017
van Driel WJ, Koole SN, Sonke GS (2018) Hyperthermic intraperitoneal chemotherapy in ovarian Cancer N. Engl J Med 378:1363–1364. https://doi.org/10.1056/NEJMc1802033
Warburg O (1956) On the origin of cancer cells. Science 123:309–314
White MG, Saleh O, Nonner D, Barrett EF, Moraes CT, Barrett JN (2012) Mitochondrial dysfunction induced by heat stress in cultured rat CNS neurons. J Neurophysiol 108:2203–2214. https://doi.org/10.1152/jn.00638.2011
Yang SJ, Huh JW, Hong HN, Kim TU, Cho SW (2004) Important role of Ser443 in different thermal stability of human glutamate dehydrogenase isozymes. FEBS Lett 562:59–64. https://doi.org/10.1016/S0014-5793(04)00183-8
Yang L, Moss T, Mangala LS, Marini J, Zhao H, Wahlig S, Armaiz-Pena G, Jiang D, Achreja A, Win J, Roopaimoole R, Rodriguez-Aguayo C, Mercado-Uribe I, Lopez-Berestein G, Liu J, Tsukamoto T, Sood AK, Ram PT, Nagrath D (2014) Metabolic shifts toward glutamine regulate tumor growth, invasion and bioenergetics in ovarian cancer. Mol Syst Biol 10:728. https://doi.org/10.1002/msb.20134892
Yuan L, Sheng X, Willson AK, Roque DR, Stine JE, Guo H, Jones HM, Zhou C, Bae-Jump VL (2015) Glutamine promotes ovarian cancer cell proliferation through the mTOR/S6 pathway. Endocr Relat Cancer 22:577–591. https://doi.org/10.1530/ERC-15-0192
Zukiene R, Nauciene Z, Ciapaite J, Mildaziene V (2010) Acute temperature resistance threshold in heart mitochondria: febrile temperature activates function but exceeding it collapses the membrane barrier. Int J Hyperth 26:56–66. https://doi.org/10.3109/02656730903262140
Acknowledgements
This research was funded by a grant (No. SEN-01/2015) from the Research Council of Lithuania.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors confirm that this article content has no conflict of interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Sukovas, A., Silkuniene, G., Trumbeckaite, S. et al. Hyperthermia potentiates cisplatin cytotoxicity and negative effects on mitochondrial functions in OVCAR-3 cells. J Bioenerg Biomembr 51, 301–310 (2019). https://doi.org/10.1007/s10863-019-09805-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10863-019-09805-8