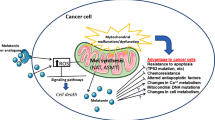
Abstract
The mitochondrial UPR (UPRmt) is rapidly gaining attention. While most studies on the UPRmt have focused on its role in aging, emerging studies suggest an important role of the UPRmt in cancer. Further, several of the players of the UPRmt in mammalian cells have well reported roles in the maintenance of the organelle. The goal of this review is to emphasize aspects of the UPRmt that have been overlooked in the current literature, describe the role of specific players of the UPRmt in the biology of the mitochondria and highlight the intriguing possibility that targeting the UPRmt in cancer may be already within reach.




Similar content being viewed by others
References
Arnold I, Langer T (2002) Membrane protein degradation by AAA proteases in mitochondria. Biochim Biophys Acta, Mol Cell Res 1592:89–96. doi:10.1016/S0167-4889(02)00267-7
Ashraf N, Zino S, MacIntyre A et al (2006) Altered sirtuin expression is associated with node-positive breast cancer. Br J Cancer 95:1056–1061. doi:10.1038/sj.bjc.6603384
Bernstein SH, Venkatesh S, Li M et al (2012) The mitochondrial ATP-dependent Lon protease: a novel target in lymphoma death mediated by the synthetic triterpenoid CDDO and its derivatives. Blood 119:3321–3329. doi:10.1182/blood-2011-02
Bhat-Nakshatri P, Wang G, Appaiah H et al (2008) AKT Alters Genome-Wide Estrogen Receptor Binding and Impacts Estrogen Signaling in Breast Cancer. Mol Cell Biol 28:7487–7503. doi:10.1128/MCB.00799-08
Bota DA, Van Remmen H, Davies KJA (2002) Modulation of Lon protease activity and aconitase turnover during aging and oxidative stress. FEBS Lett 532:103–106. doi:10.1016/S0014-5793(02)03638-4
Campbell RA, Bhat-Nakshatri P, Patel NM et al (2001) Phosphatidylinositol 3-Kinase/AKT-mediated Activation of Estrogen Receptor α. J Biol Chem 276:9817–9824. doi:10.1074/jbc.M010840200
Carroll JS, Meyer CA, Song J et al (2006) Genome-wide analysis of estrogen receptor binding sites. Nat Genet 38:1289–1297. doi:10.1038/ng1901
Chae YC, Angelin A, Lisanti S et al (2013) Landscape of the mitochondrial Hsp90 metabolome in tumours. Nat Commun 4:2139. doi:10.1038/ncomms3139
Cole A, Wang Z, Coyaud E et al (2015) Inhibition of the Mitochondrial Protease ClpP as a Therapeutic Strategy for Human Acute Myeloid Leukemia. Cancer Cell 27:864–876. doi:10.1016/j.ccell.2015.05.004
Cruz-Bermúdez A, Vallejo CG, Vicente-Blanco RJ et al (2015) Enhanced tumorigenicity by mitochondrial DNA mild mutations. Oncotarget 6:13628–13643. doi:10.18632/oncotarget.3698
Durieux J, Wolff S, Dillin A (2011) The cell-non-autonomous nature of electron transport chain-mediated longevity. Cell 144:79–91. doi:10.1016/j.cell.2010.12.016
Feeley KP, Bray AW, Westbrook DG et al (2015) Mitochondrial genetics regulate breast cancer tumorigenicity and metastatic potential HHS Public Access. Cancer Res 75:4429–4436. doi:10.1158/0008-5472.CAN-15-0074
Finley LWS, Carracedo A, Lee J et al (2011) SIRT3 Opposes Reprogramming of Cancer Cell Metabolism through HIF1α Destabilization. Cancer Cell 19:416–428. doi:10.1016/j.ccr.2011.02.014
Franco HL, Nagari A, Lee W, Correspondence K (2015) TNFα Signaling Exposes Latent Estrogen Receptor Binding Sites to Alter the Breast Cancer Cell Transcriptome. Mol Cell 58:21–34. doi:10.1016/j.molcel.2015.02.001
Germain D (2016a) Sirtuins and the Estrogen Receptor as Regulators of the Mammalian Mitochondrial UPR in Cancer and Aging. Adv Cancer Res 130:211–56. doi:10.1016/bs.acr.2016.01.004
Germain D (2016b) Mitochondrial UPR in Cancer. In: Mitochondria and Cell Death. Springer New York, New York, pp 149–167
Gertz J, Savic D, Varley KE et al (2013) Distinct Properties of Cell-Type-Specific and Shared Transcription Factor Binding Sites. Mol Cell 52:25–36. doi:10.1016/j.molcel.2013.08.037
Gitschlag BL, Kirby CS, Samuels DC et al (2016) Homeostatic Responses Regulate Selfish Mitochondrial Genome Dynamics in C. elegans. Cell Metab 24:91–103. doi:10.1016/j.cmet.2016.06.008
Haigis MC, Deng CX, Finley LWS et al (2012) SIRT3 is a mitochondrial tumor suppressor: A scientific tale that connects aberrant cellular ROS, the Warburg effect, and carcinogenesis. Cancer Res 72:2468–2472
Herrmann JM, Riemer J (2010) The intermembrane space of mitochondria. Antioxid Redox Signal 13:1341–1358. doi:10.1089/ars.2009.3063
Imanishi H, Hattori K, Wada R et al (2011) Mitochondrial DNA mutations regulate metastasis of human breast cancer cells. PLoS One. doi:10.1371/journal.pone.0023401
Ishikawa K, Hayashi JI (2009) Trading mtDNA uncovers its role in metastasis. Cell Adhes Migr 3:11–13
Ishikawa K, Hashizume O, Koshikawa N et al (2008a) Enhanced glycolysis induced by mtDNA mutations does not regulate metastasis. FEBS Lett 582:3525–3530. doi:10.1016/j.febslet.2008.09.024
Ishikawa K, Takenaga K, Akimoto M et al (2008b) ROS-Generating Mitochondrial DNA Mutations Can Regulate Tumor Cell Metastasis. Science (80- ) 320:661–664. doi:10.1126/science.1156906
Jin SM, Youle RJ (2013) The accumulation of misfolded proteins in the mitochondrial matrix is sensed by PINK1 to induce PARK2/Parkin-mediated mitophagy of polarized mitochondria. Autophagy. doi:10.4161/auto.26122
Kenny TC, Hart P, Ragazzi M et al (2017) Selected mitochondrial DNA landscapes activate the SIRT3 axis of the UPRmt to promote metastasis. Oncogene. doi:10.1038/onc.2017.52
Kim H-S, Patel K, Muldoon-Jacobs K et al (2010) SIRT3 is a mitochondria-localized tumor suppressor required for maintenance of mitochondrial integrity and metabolism during stress. Cancer Cell 17:41–52. doi:10.1016/j.ccr.2009.11.023
Kulawiec M, Owens KM, Singh KK (2009) Cancer cell mitochondria confer apoptosis resistance and promote metastasis. Cancer Biol Ther 8:1378–1385. doi:10.4161/cbt.8.14.8751
Lannigan DA (2003) Estrogen receptor phosphorylation. Steroids 68:1–9
Lemarie A, Grimm S (2011) Mitochondrial respiratory chain complexes: apoptosis sensors mutated in cancer? Oncogene 30:3985–4003. doi:10.1038/onc.2011.167
Lin Y-F, Schulz AM, Pellegrino MW et al (2016) Maintenance and propagation of a deleterious mitochondrial genome by the mitochondrial unfolded protein response. Nature 533:416–419. doi:10.1038/nature17989
Lombard DB, Tishkoff DX, Bao J (2011) Mitochondrial Sirtuins in the Regulation of Mitochondrial Activity and Metabolic Adaptation. Handb Exp Pharmacol 206:163–88. doi:10.1007/978-3-642-21631-2_8
Martinus RD, Garth GP, Webster TL et al (1996) Selective induction of mitochondrial chaperones in response to loss of the mitochondrial genome. Eur J Biochem 240:98–103. doi:10.1111/j.1432-1033.1996.0098h.x
Mattingly KA, Ivanova MM, Riggs KA et al (2008) Estradiol Stimulates Transcription of Nuclear Respiratory Factor-1 and Increases Mitochondrial Biogenesis. Mol Endocrinol 22:609–622. doi:10.1210/me.2007-0029
McMahon S, LaFramboise T (2014) Mutational patterns in the breast cancer mitochondrial genome, with clinical correlates. Carcinogenesis 35:1046–1054. doi:10.1093/carcin/bgu012
Mohammed H, Russell IA, Stark R et al (2015) Progesterone receptor modulates ERα action in breast cancer. Nature 523:313–317. doi:10.1038/nature14583
Nunes JB, Peixoto J, Soares P et al (2015) OXPHOS dysfunction regulates integrin- 1 modifications and enhances cell motility and migration. Hum Mol Genet 24:1977–1990. doi:10.1093/hmg/ddu612
Papa L, Germain D (2011) Estrogen receptor mediates a distinct mitochondrial unfolded protein response. J Cell Sci 124:1396–1402. doi:10.1242/jcs.078220
Papa L, Germain D (2014) SirT3 regulates the mitochondrial unfolded protein response. Mol Cell Biol 34:699–710. doi:10.1128/MCB.01337-13
Papa L, Manfredi G, Germain D (2014) SOD1, an unexpected novel target for cancer therapy. Genes Cancer 5(1-2):15–21
Radke S, Chander H, Schäfer P et al (2008) Mitochondrial protein quality control by the proteasome involves ubiquitination and the protease Omi. J Biol Chem 283:12681–12685. doi:10.1074/jbc.C800036200
Ristow M, Schmeisser K (2014) Mitohormesis: promoting health and lifespan by increased levels of reactive oxygen species (ROS). Dose-Response Former Nonlinearity Biol 12:288–341. doi:10.2203/dose-response.13-035.Ristow
Santidrian AF, Matsuno-Yagi A, Ritland M et al (2013) Mitochondrial complex I activity and NAD+/NADH balance regulate breast cancer progression. J Clin Invest 123:1068–1081. doi:10.1172/JCI64264
Scarpulla RC (2011) Metabolic control of mitochondrial biogenesis through the PGC-1 family regulatory network. Biochim Biophys Acta, Mol Cell Res 1813:1269–1278
Seo JH, Rivadeneira DB, Caino MC et al (2016) The Mitochondrial Unfoldase-Peptidase Complex ClpXP Controls Bioenergetics Stress and Metastasis. PLoS Biol 14:e1002507. doi:10.1371/journal.pbio.1002507
Siegelin MD, Dohi T, Raskett CM et al (2011) Exploiting the mitochondrial unfolded protein response for cancer therapy in mice and human cells. J Clin Invest 121:1349–1360. doi:10.1172/JCI44855
Skrtic M, Sriskanthadevan S, Jhas B et al (2011) Inhibition of Mitochondrial Translation as a Therapeutic Strategy for Human Acute Myeloid Leukemia. Cancer Cell 20:674–688. doi:10.1016/j.ccr.2011.10.015
Sun M, Paciga JE, Feldman RI et al (2001) Phosphatidylinositol-3-OH Kinase (PI3K)/AKT2, activated in breast cancer, regulates and is induced by estrogen receptor alpha (ERalpha) via interaction between ERalpha and PI3K. Cancer Res 61:5985–5991. doi:10.1038/nature
Sundaresan NR, Samant SA, Pillai VB et al (2008) SIRT3 is a stress-responsive deacetylase in cardiomyocytes that protects cells from stress-mediated cell death by deacetylation of Ku70. Mol Cell Biol 28:6384–6401. doi:10.1128/MCB.00426-08
Sundaresan NR, Gupta M, Kim G et al (2009) Sirt3 blocks the cardiac hypertrophic response by augmenting Foxo3a-dependent antioxidant defense mechanisms in mice. J Clin Invest 119:2758–2771. doi:10.1172/JCI39162
Takibuchi G, Imanishi H, Morimoto M et al (2013) Polymorphic mutations in mouse mitochondrial DNA regulate a tumor phenotype. Mitochondrion 13:881–887. doi:10.1016/j.mito.2013.07.117
Tao R, Coleman MC, Pennington JD et al (2010) Sirt3-Mediated Deacetylation of Evolutionarily Conserved Lysine 122 Regulates MnSOD Activity in Response to Stress. Mol Cell 40:893–904. doi:10.1016/j.molcel.2010.12.013
Tatsuta T, Langer T (2008) Focus Quality Control Quality control of mitochondria: protection against neurodegeneration and ageing. EMBO J 27(2):306–314. doi:10.1038/sj.emboj.7601972
Taylor S, Lam M, Pararasa C et al (2015) Evaluating the evidence for targeting FOXO3a in breast cancer: a systematic review. Cancer Cell Int 15:1. doi:10.1186/s12935-015-0156-6
Vilgelm A, Lian Z, Wang H et al (2006) Akt-mediated phosphorylation and activation of estrogen receptor alpha is required for endometrial neoplastic transformation in Pten+/− mice. Cancer Res 66:3375–3380. doi:10.1158/0008-5472.CAN-05-4019
Wallace DC (2012) Mitochondria and cancer. Nat Rev Cancer 12:685–698. doi:10.1038/nrc3365
Wu C-W, Storey KB (2014) FoxO3a-mediated activation of stress responsive genes during early torpor in a mammalian hibernator. Mol Cell Biochem 390:185–195. doi:10.1007/s11010-014-1969-7
Zhao Q, Wang J, Levichkin IV et al (2002) A mitochondrial specific stress response in mammalian cells. EMBO J 21:4411–4419. doi:10.1093/emboj/cdf445
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kenny, T.C., Germain, D. From discovery of the CHOP axis and targeting ClpP to the identification of additional axes of the UPRmt driven by the estrogen receptor and SIRT3. J Bioenerg Biomembr 49, 297–305 (2017). https://doi.org/10.1007/s10863-017-9722-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10863-017-9722-z