Abstract
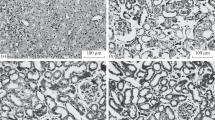
Although the pathogenesis of ischemia reperfusion (IR) injury is based on complex mechanisms, free radicals play a central role. We evaluated membrane fluidity and lipid peroxidation during pancreas transplantation (PT) performed in 12 pigs (six donors and six recipients). Fluidity was measured by fluorescence spectroscopy, and malondialdehyde (MDA) and 4-hydroxyalkenals (4-HDA) concentrations were used as an index of lipid oxidation. Pancreatic tissues were collected as follows: (A) donor, immediately before vascular clamping; (B) graft, following perfusion lavage with University of Wisconsin preservation fluid; (C) graft, after 16 h of cold ischemia; and (D) recipient, 30 min vascular postreperfusion. Fluidity and MDA and 4-HDA concentrations were similar in cases A, B, and C. However, there was significant membrane rigidity and increased lipid peroxidation after reperfusion (D). These findings suggest that reperfusion exaggerates oxidative damage and may account for the rigidity in the membranes of allografts during PT.
Similar content being viewed by others
References
Belzer FO, Southard JH (1988) Principles of solid organ preservation by cold storage. Transplantation 45:673–676
Benkoel L, Dodero F, Hardwigsen J, Mas E, Benoliel AM, Botta-Fridlund D, Le Treut YP, Chamlian A, Lombardo D (2004) Effect of ischemia-reperfusion on Na+, K+-ATPase expression in human liver tissue allograft: image analysis by confocal laser scanning microscopy. Dig Dis Sci 49:1387–1393
Benz S, Obermaier R, Wiessner R, Breitenbuch PV, Burska D, Weber H, Schnabel R, Mayer J, Pfeffer F, Nizze H, Hopt UT (2002) Effect of nitric oxide in ischemia/reperfusion of the pancreas. J Surg Res 106:46–53
Blankensteijn JD, Terpstra OT (1991) Liver preservation: the past and the future. Hepatology 13:1235–1250
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein–dye binding. Anal Biochem 72:248–254
Büsing M, Hopt UT, Qnacken M, Becket HD, Morgenroth K (1993) Morphological studies of graft pancreatitis following pancreas transplantation. Br J Surg 80:1170–1173
Busuttil RW, Tanaka K (2003) The utility of marginal donors in liver transplantation. Liver Transpl 9:651–663
Caimi G, Ferrara F, Montana M, Meli F, Canino B, Carollo C, Lo Presti R (2000) Acute ischemic stroke: polymorphonuclear leukocyte membrane fluidity and cytosolic Ca2+ concentration at baseline and after chemotactic activation. Stroke 31:1578–1582
Casillas-Ramírez A, Mosbah IB, Ramalho F, Roselló-Catafau J, Peralta C (2006) Past and future approaches to ischemia-reperfusion lesion associated with liver transplantation. Life Sci 79:1881–1894
Chen JJ, Yu BP (1994) Alterations in mitochondrial membrane fluidity by lipid peroxidation products. Free Radic Biol Med 17:411–418
Clement AB, Gimpl G, Behl C (2010) Oxidative stress resistance in hippocampal cells is associated with altered membrane fluidity and enhanced nonamyloidogenic cleavage of endogenous amyloid precursor protein. Free Radic Biol Med 48:1236–1241
Curtis MT, Gilfor D, Farber JL (1984) Lipid peroxidation increases the molecular order of microsomal membranes. Arch Biochem Biophys 235:644–649
Cutrin JC, Perrelli MG, Cavalieri B, Peralta C, Rosello-Catafau J, Poli G (2002) Microvascular dysfunction induced by reperfusion injury and protective effect of ischemic preconditioning. Free Rad Biol Med 33:1200–1208
Drachenberg CB, Odorico J, Demetris AJ, Arend L, Bajema IM, Bruijn JA, Cantarovich D, Cathro HP, Chapman J, Dimosthenous K, Fyfe-Kirschner B, Gaber L, Gaber O, Goldberg J, Honsová E, Iskandar SS, Klassen DK, Nankivell B, Papadimitriou JC, Racusen LC, Randhawa P, Reinholt FP, Renaudin K, Revelo PP, Ruiz P, Torrealba JR, Vazquez-Martul E, Voska L, Stratta R, Bartlett ST, Sutherland DE (2008) Banff schema for grading pancreas allograft rejection: working proposal by a multi-disciplinary international consensus panel. Am J Transplant 8:1237–1249
Drognitz O, Obermaier R, Von Dobschuetz E, Pisarski P, Neeff H (2009) Pancreas transplantation and ischemia-reperfusion injury: current considerations. Pancreas 38:226–227
Fernández-Cruz L, Sabater L, Gilabert IL, Ricart MJ, Sáenz A, Astudillo E (1993) Native and graft pancreatitis following combined pancreas-renal transplantation. Br J Surg 80:1429–1432
Frederiks WM, Myagkaya GL, van Veen HA, Vogels IM (1984) Biochemical and ultrastructural changes in rat liver plasma membranes after temporary ischemia. Virchows Arch B Cell Pathol Incl Mol Pathol 46:269–282
García JJ, Reiter RJ, Guerrero JM, Escames G, Yu BP, Oh CS, Muñoz-Hoyos A (1997) Melatonin prevents changes in microsomal membrane fluidity during induced lipid peroxidation. FEBS Lett 408:297–300
García JJ, Piñol-Ripoll G, Martínez-Ballarín E, Fuentes-Broto L, Miana-Mena FJ, Venegas C, Caballero B, Escames G, Coto-Montes A, Acuña-Castroviejo D (2011) Melatonin reduces membrane rigidity and oxidative damage in the brain of SAMP8 mice. Neurobiol Aging 32:2045–2054
García-Gil FA, Gonzalvo E, García JJ, Albendea CD, Güemes A, Tome-Zelaya E, Fuentes L, Santa-Clotilde E, Aso J, Bejarano C, Garrido N, García C, Gómez E, Sánchez M (2006) Lipid peroxidation in ischemia-reperfusion oxidative injury of the graft preserved in Celsior and University of Wisconsin solutions on a pig pancreas transplantation model. Transplant Proc 38:2595–2399
García-Gil FA, Albendea CD, Escartín J, Lampreave F, Fuentes-Broto L, Roselló-Catafau J, López-Pingarrón L, Reiter RJ, Alvarez-Alegret R, García JJ (2011) Melatonin prolongs graft survival of pancreas allotransplants in pigs. J Pineal Res 51:445–453
Grimm MO, Tschäpe JA, Grimm HS, Zinser EG, Hartmann T (2006) Altered membrane fluidity and lipid raft composition in presenilin-deficient cells. Acta Neurol Scand 185:27–32
Halliwel B, Gutteridge JMC (2007) Free radicals in biology and medicine. Oxford University Press, Oxford
Hamer I, Wattiaux R, Wattiaux-De Coninck S (1995) Deleterious effects of xanthine oxidase on rat liver endothelial cells after ischemia/reperfusion. Biochim Biophys Acta 1269:145–152
Irazu CE, Rajagopalan PR, Orak JK, Fitts CT, Singh I (1990) Mitochondrial membrane fluidity changes in renal ischemia. J Exp Pathol 5:1–6
Jaeschke H (2003) Molecular mechanisms of hepatic ischemia-reperfusion injury and preconditioning. Am J Physiol Gastrointest Liver Physiol 284:G15–G26
Jaeschke H, Mitchell JR (1989) Mitochondria and xanthine oxidase both generate reactive oxygen species in isolated perfused rat liver after hypoxic injury. Biochem Biophys Res Commun 160:140–147
Janero DR (1990) Malondialdehyde and thiobarbituric acid-reactivity as diagnostic indices of lipid peroxidation and peroxidative tissue injury. Free Radic Biol Med 9:515–540
Jaworek J, Leja-Szpak A, Bonior J, Nawrot K, Tomaszewska R, Stachura J, Sendur R, Pawlik W, Brzozowski T, Konturek SJ (2003) Protective effect of melatonin and its precursor l-tryptophan on acute pancreatitis induced by caerulein overstimulation or ischemia/reperfusion. J Pineal Res 34:40–52
Jourd'heuil D, Meddings JB (2001) Oxidative and drug-induced alterations in brush border membrane hemileaflet fluidity, functional consequences for glucose transport. Biochim Biophys Acta 1510:342–353
Khoury W, Namnesnikov M, Fedorov D, Abu-Ghazala S, Weinbroum AA (2010) Mannitol attenuates kidney damage induced by xanthine oxidase-associated pancreas ischemia-reperfusion. J Surg Res 160:163–168
Kubo S, Yamamoto K, Magata Y, Iwasakim Y, Tamaki N, Yonekura Y, Konishi J (1991) Assessment of pancreatic blood flow with positron emission tomography and oxygen-15 water. Ann Nucl Med 5:133–138
Kurose I, Wolf R, Grisham MB, Granger DN (1994) Modulation of ischemia/reperfusion-induced microvascular dysfunction by nitric oxide. Circ Res 74:376–382
Li JX, Tong CW, Xu DQ, Chan KM (1999) Changes in membrane fluidity and lipid peroxidation of skeletal muscle mitochondria after exhausting exercise in rats. Eur J Appl Physiol Occup Physiol 80:113–117
Lima-Rodríguez JR, García-Gil FA, García JJ, Rocha-Camarero G, Martín-Cancho MF, Luis-Fernández L, Crisóstomo V, Usón-Gargallo J, Carrasco-Jiménez MS (2008) Effects of premedication with tiletamine/zolazepam/ medetomidine during general anesthesia using sevoflurane/ fentanyl in swine undergoing pancreas transplantation. Transplant Proc 40:3001–3006
Mayer H, Schmidt J, Tbies J, Ryschicb E, Gebbard MM, Herfartb C, Klar E (1999) Characterization and reduction of ischemia/reperfusion injury after experimental pancreas transplantation. J Gastrointest Surg 3:162–166
Meldolesi J, Jamieson JD, Palade GE (1971) Composition of cellular membranes in the pancreas of the guinea pig. I. Isolation of membrane fractions. J Cell Biol 49:109–129
Menger MD, Pelikan S, Steiner D, Messmer K (1992) Microvascular ischemia-reperfusion injury in striated muscle: significance of "reflow-paradox". Am J Physiol 263:H1901–H1906
Miana-Mena FJ, Piedrafita E, González-Mingot C, Larrodé P, Muñoz MJ, Martínez-Ballarín E, Reiter RJ, Osta R, García JJ (2011) Levels of membrane fluidity in the spinal cord and the brain in an animal model of amyotrophic lateral sclerosis. J Bioenerg Biomembr 43:181–186
Molitoris BA, Kinne R (1987) Ischemia induces surface membrane dysfunction. Mechanism of altered Na+-dependent glucose transport. J Clin Invest 80:647–654
Muñoz-Casares FC, Padillo FJ, Briceño J, Collado JA, Muñoz-Castañeda JR, Ortega R, Cruz A, Túnez I, Montilla P, Pera C, Muntané J (2006) Melatonin reduces apoptosis and necrosis induced by ischemia/reperfusion injury of the pancreas. J Pineal Res 40:195–203
Patil DT, Yerian LM (2010) Pancreas transplant recent advances and spectrum of features in pancreas allograft pathology. Adv Anat Pathol 17:202–208
Plin C, Haddad PS, Tillement JP, Elimadi A, Morin D (2004) Protection by cyclosporin A of mitochondrial and cellular functions during a cold preservation-warm reperfusion of rat liver. Eur J Pharmacol 495:111–118
Reiter RJ, Tan D, Kim SJ, ManchesterL C, Qi W, García JJ, Cabrera JC, El-Sokkary G, Rouvier-Garay V (1999) Augmentation of indices of oxidative damage in life-long melatonin-deficient rats. Mech Ageing Dev 110:157–173
Rice-Evans C, Burdon R (1993) Free radical-lipid interactions and their pathological consequences. Prog Lipid Res 32:71–110
Schulak JA, Mayes JT, Iricik DE (1990) Combined kidney and pancreas transplantation. A safe and effective treatment for diabetic nephropathy. Arch Surg 125:881–885
Shin G, Sugiyama M, Shoji T, Kagiyama A, Sato H, Ogura R (1989) Detection of mitochondrial membrane damages in myocardial ischemia with ESR spin labeling technique. J Mol Cell Cardiol 21:1029–1036
Singer SJ, Nicolson GL (1972) The fluid mosaic model of the structure of cell membranes. Science 175:720–731
Sollinger HW, Stratta RJ, D'Alessandro AM, Kalayoglu M, Pirsch JD, Belzer FO (1988) Experience with simultaneous pancreas-kidney transplantation. Ann Surg 208:475–483
Southard JH (2004) The right solution for organ preservation. North Am Pharmacother 2:1–4
Sumimoto K, Matsura T, Oku JI, Fukuda Y, Yamada K, Dohi K (1996) Protective effect of UW solution on postischemic injury in rat liver: suppression of reduction in hepatic antioxidants during reperfusion. Transplantation 62:1391–1398
Sutherland DER (1997) Pancreas transplantation as a treatment for diabetes: indications and outcome. Curr Ther Endocrinol Metab 6:496–499
Tiedge M, Lortz S, Drinkgern J, Lenzen S (1997) Relation between antioxidant enzyme gene expression and antioxidative defense status of insulin-producing cells. Diabetes 46:1733–1742
Vajdova K, Graf R, Clavien PA (2002) ATP-supplies in the cold-preserved liver: a long-neglected factor of organ viability. Hepatology 36:1543–1552
Vareesangthip K, Nimmannit S, Chuawattana D, Mingkam S, Hanlakorn P, Ong-Aj-Yooth L (2001) Impairment of erythrocyte membrane fluidity in cyclosporine-treated renal transplant patients. Transplant Proc 33:1198–1200
Vollmar B, Janata J, Yamauchi JI, Menger MD (1999) Attenuation of microvascular reperfusion injury in rat pancreas transplantation by L-arginine. Transplantation 67:950–955
Witzigmann H, Ludwig S, Armann B, Gäbel G, Teupser D, Kratzsch J, Pietsch UC, Tannapfel A, Geissler F, Hauss J, Uhlmann D (2003) Endothelin-A receptor blockade reduces ischemia/reperfusion injury in pig pancreas transplantation. Ann Surg 238:264–274
Wrobel A, Kaminska D, Klinger M (2003) Electron paramagnetic resonance study of erythrocyte membrane fluidity in renal transplant recipients. Transplant Proc 35:2230–2232
Yu BP, Suescun EA, Yang SY (1992) Effect of age-related lipid peroxidation on membrane fluidity and phospholipase A2: modulation by dietary restriction. Mech Ageing Dev 65:17–33
Zaouali MA, Ben Abdennebi H, Padrissa-Altés S, Mahfoudh-Boussaid A, Roselló-Catafau J (2010) Pharmacological strategies against cold ischemia reperfusion injury. Expert Opin Pharmacother 11:537–555
Zhou XM, Cao YL, Dou DQ (2006) Protective effect of ginsenoside-Re against cerebral ischemia/reperfusion damage in rats. Biol Pharm Bull 29:2502–2505
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
García-Gil, F.A., Albendea, C.D., López-Pingarrón, L. et al. Altered cellular membrane fluidity levels and lipid peroxidation during experimental pancreas transplantation. J Bioenerg Biomembr 44, 571–577 (2012). https://doi.org/10.1007/s10863-012-9459-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10863-012-9459-7