Abstract
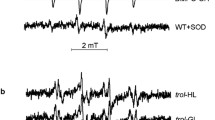
The present study for the first time describes a close relationship between a change in the states of Cyt b559, a damage to Mn complex and a rapid reduction of tyrosine D (YD) as a function of temperature in spinach thylakoid membranes. Measurements of the EPR signal of dark stable tyrosine D in heat-treated thylakoid membranes showed a gradual decay of the oxidized state of tyrosine D with the progression of temperature. Simultaneously, it leads to the conversion of high-potential Cytochrome b559 into its low-potential form. We have speculated a possible involvement of Cytochrome b559 in the primary reduction events of tyrosine D in dark at high temperature. However, rapid reduction of tyrosine D may also be due to the disassembly of the Mn clock, which causes exposure of YD to the lumen and thereby its reduction by some unknown factor. These conclusions are supported by the measurements of Mn2+ release and thermoluminescence curves of various charge pairs in heat-treated thylakoid membranes. The results reveal an important aspect on the role of Cyt b559 in PS II during temperature stress.
Similar content being viewed by others
References
Ananyev GM, Sakiyan I, Diner BA, Dismukes GC (2002) Biochemistry 41:974–980
Babcock GT, Sauer K (1973) Biochim Biophys Acta 325:483–503
Babcock GT, Sauer K (1975) Biochim Biophys Acta 376:315–328
Barra M, Haumann M, Dau H (2005) Photosynth Res 84:231–237
Bhatnagar R, Saxena P, Vora HS, Dubey VK, Sarangpani KK, Shirke ND, Bhattacharjee SK (2002) Meas Sci Technol 13:2017–2026
Blankenship RE, Babcock GT, Warden JT, Sauer K (1975) FEBS Lett 51:287–293
Buser CA, Thompson LK, Diner BA, Brudvig GW (1990) Biochemistry 29:8977–8985
Campbell KA, Peloquin JM, Diner BA, Tang XS, Chisholm DA, Britt RD (1997) J Am Chem Soc 119:4787–4788
Canaani O, Havaux M (1990) Proc Natl Acad Sci USA 87:9295–9299
Cramer WA, Whitmarsh J, Low PS (1981) Biochemistry 20:157–162
Danielsson R, Albertsson PA, Mamedov F, Styring S (2004) Biochim Biophys Acta 1608:53–61
Deák Z, Vass I, Styring S (1994) Biochim Biophys Acta 1185:65–74
Demeter S, Vass I (1984) Biochim Biophys Acta 764:24–32
Enami I, Kitamura M, Tomo T, Isokawa Y, Ohta H, Katoh S (1994) Biochim Biophys Acta 1186:52–58
Faller P, Debus RJ, Brettel K, Sugiura M, Rutherford AW, Boussac A (2001) Proc. Natl. Acad. Sci. U. S. A. 98:14368–14373
Faller P, Rutherford AW, Debus RJ (2002) Biochemistry 41:12914–12920
Ferreira KN, Iverson TM, Maghlaoui K, Barber J, Iwata S (2004) Science 303:1831–1838
Ghanotakis DF, Yocum CF, Babcock GT (1986) Photosynth Res 9:125–134
Hanley J, Deligiannakis Y, Pascal A, Faller P, Rutherford AW (1999) Biochemistry 38:8189–8195
Havaux M (1998) Isr J Chem 38:247–256
Horton P, Croze E, Smutzer G (1978) Biochim Biophys Acta 503:274–286
Jang WC, Tae GC (1996) J Biochem Mol Biol 29:58–62
Johnson GN, Boussac A, Rutherford AW (1994) Biochim Biophys Acta 1184:85–92
Kaminskaya O, Kern J, Shuvalov VA, Renger G (2005) Biochim Biophys Acta 1708:333–341
Kawamori A, Satoh J, Inui T, Satoh K (1987) FEBS Lett 217:134–138
Kok B, Forbush B, McGloin M (1970) Photochem Photobiol 11:457–475
Kouril R, Lazar D, Ilýk P, Skotnica J, Krchnak P, Nauš J (2004) Photosynth Res 81:49–66
Krieger A, Weis E, Demeter S (1993) Biochim Biophys Acta 1144:411–418
Kuwabara T, Murata N (1982) Plant Cell Physiol 23:533–539
Lazar D, Ilik P, Kruk J, Strzalka K, Naus J (2005) J Theor Biol 233:287–300
Loll B, Kern J, Saenger W, Zouni A, Biesiadka J (2005) Nature 438:1040–1044
Magnuson A, Rova M, Mamedov F, Fredriksson PO, Styring S (1999) Biochim Biophys Acta 1411:180–191
Mende D, Wiessner W, Demeter S (1996) Photosynth Res 49:277–280
Miller AF, Brudvig GW (1991) Biochim Biophys Acta 1056:1–18
Mizusawa N, Ebina M, Yamashita T (1995) Photosynth Res 45:71–77
Nash D, Miyao M, Murata N (1985) Biochim Biophys Acta 807:127–133
Pakrasi HB, Nyhus KJ, Granok H (1990) Z Naturforsch 45c:423–429
Pakrasi HB, De Ciechi P, Whitmarsh J (1991) EMBO J 10:1619–1627
Porra RJ, Thompson WA, Kriedemann PE (1989) Biochim Biophys Acta 975:384–394
Pospišil P, Haumann M, Dittmer J, Sole VA, Dau H (2003) Biophys J 84:1370–1386
Pospišil P, Šnyrychova I, Kruk J, Strzalka K, Nauš J (2006) Biochem J 397:321–327
Poulson M, Samson M, Whitmarsh J (1995) Biochemistry 34:10932–10938
Rutherford AW, Crofts AR, Inoue Y (1982) Biochim Biophys Acta 682:457–465
Rutherford AW, Govindjee, Inoue Y (1984) Proc Natl Acad Sci USA 81:1107–1111
Rutherford AW, Renger G, Koike H, Inoue Y (1985) Biochim Biophys Acta 767:548–556
Sane PV, Rutherford AW (1989) In: Govindjee, Amesz J, and Fork DC (eds) Light Emission by Plants and Bacteria. Acad. Press, New York, pp 326–361
Schreiber U, Neubauer C (1990) Photosynth Res 25:279–293
Styring S, Rutherford AW (1987) Biochemistry 26:2401–2405
Tae GS, Black MT, Cramer WA, Vallon O, Bogorad L (1988) Biochemistry 27:9075–9080
Tiwari A, Jajoo A, Bharti S, Mohanty P (2007) Photosynth Res DOI 10.1007/s11120-007-9130-z
Vass I, Styring S (1991) Biochemistry 30:830–839
Vass I, Deak Z, Jegerschold C, Styring S (1990) Biochim Biophys Acta 1018:41–46
Vermaas WFJ, Renger G, Dohnt G (1989) Biochim Biophys Acta 764:194–202
Yamane Y, Kashino Y, Koike H, Satoh K (1998) Photosynth Res 57:51–59
Zouni A, Witt HT, Kern J, Fromme P, Krauss N, Saenger W, Orth P (2001) Nature 409:739–743
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tiwari, A., Jajoo, A. & Bharti, S. Heat-induced changes in the EPR signal of tyrosine D (\( Y^{{OX}}_{D} \)): a possible role of Cytochrome b559. J Bioenerg Biomembr 40, 237–243 (2008). https://doi.org/10.1007/s10863-007-9099-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10863-007-9099-5