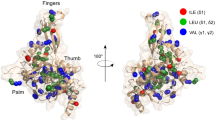
Abstract
Modification of proteins by post-translational covalent attachment of a single, or chain, of ubiquitin molecules serves as a signaling mechanism for a number of regulatory functions in eukaryotic cells. For example, proteins tagged with lysine-63 linked polyubiquitin chains are involved in error-free DNA repair. The catalysis of lysine-63 linked polyubiquitin chains involves the sequential activity of three enzymes (E1, E2, and E3) that ultimately transfer a ubiquitin thiolester intermediate to a protein target. The E2 responsible for catalysis of lysine-63 linked polyubiquitination is a protein heterodimer consisting of a canonical E2 known as Ubc13, and an E2-like protein, or ubiquitin conjugating enzyme variant (UEV), known as Mms2. We have determined the solution structure of the complex formed by human Mms2 and ubiquitin using high resolution, solution state nuclear magnetic resonance (NMR) spectroscopy. The structure of the Mms2–Ub complex provides important insights into the molecular basis underlying the catalysis of lysine-63 linked polyubiquitin chains.
Similar content being viewed by others
Abbreviations
- Ub:
-
ubiquitin
- E1:
-
ubiquitin activating enzyme
- E2:
-
ubiquitin conjugating enzyme
- E3:
-
ubiquitin ligase
- UEV:
-
ubiquitin conjugating enzyme variant
- CUE:
-
similar to yeast protein Cue1p
- UBA:
-
ubiquitin associated domain
- UIM:
-
ubiquitin interacting motif
- NMR:
-
nuclear magnetic resonance
- ITC:
-
isothermal titration calorimetry
- IPTG:
-
isopropyl ß-D-thiogalactopyranoside
- NOE:
-
nuclear Overhauser effect
References
A. Bax G.M. Clore A.M. Gronenborn (1990) J. Magn. Reson. 88 425–431
Y. Ben-Neriah (2002) Nat. Immunol. 3 20–26 Occurrence Handle10.1038/ni0102-20
V. Bernier-Villamor D.A. Sampson M.J. Matunis C.D. Lima (2002) Cell 108 345–356
S. Broomfield B.L. Chow W. Xiao (1998) Proc. Natl. Acad. Sci. USA 95 5678–5683 Occurrence Handle10.1073/pnas.95.10.5678 Occurrence Handle1998PNAS...95.5678B
J. Cavanagh A.G. Palmer P.E. Wright M. Rance (1991) J. Magn. Reson. 91 429–436
N.L. Chan C.P. Hill (2001) Nat. Struct. Biol. 8 650–652 Occurrence Handle10.1038/90337
F. Delaglio S. Grzesiek G.W. Vuister G. Zhu J. Pfeifer A. Bax (1995) J. Biomol. NMR 6 277–293 Occurrence Handle10.1007/BF00197809
W.L. DeLano (2002) DeLano Scientific San Carlos CA, USA
L. Deng C. Wang E. Spencer L. Yang A. Braun J. You C. Slaughter C. Pickart Z.J. Chen (2000) Cell 103 351–361 Occurrence Handle10.1016/S0092-8674(00)00126-4
C. Dominguez R. Boelens A.M.J.J. Bonvin (2003) J. Am. Chem. Soc. 125 1731–1737
K.H. Gardner R. Konrat M.K. Rosen L.E. Kay (1996) J. Biomol. NMR 8 351–356 Occurrence Handle10.1007/BF00410333
D.S. Garrett Y.J. Seok A. Peterkofsky G.M. Clore A.M. Gronenborn (1997) Biochemistry 36 4393–4398
M.H. Glickman A. Ciechanover (2002) Physiol. Rev. 82 373–428
Goddard, T. D. and Kneller, D. G. University of California, San Francisco
K. Haglund P.P. Fiore ParticleDi I. Dikic (2003) Trends Biochem. Sci. 28 598–603 Occurrence Handle10.1016/j.tibs.2003.09.005
L. Hicke (2001) Nat. Rev. Mol. Cell. Biol. 2 195–201 Occurrence Handle10.1038/35056583
L. Hicke H.L. Schubert C.P. Hill (2005) Nat. Rev. Mol. Cell. Biol. 6 610–621 Occurrence Handle10.1038/nrm1701
C. Hoege B. Pfander G.L. Moldovan G. Pyrowolakis S. Jentsch (2002) Nature 419 135–141 Occurrence Handle10.1038/nature00991 Occurrence Handle2002Natur.419..135H
R.M. Hofmann C.M. Pickart (1999) Cell 96 645–653 Occurrence Handle10.1016/S0092-8674(00)80575-9
R.S. Kang C.M. Daniels S.A. Francis S.C. Shih W.J. Salerno L. Hicke I. Radhakrishnan (2003) Cell 113 621–630 Occurrence Handle10.1016/S0092-8674(03)00362-3
L.E. Kay M. Ikura R. Tschudin A. Bax (1990) J. Magn. Reson. 89 496–514
L.E. Kay P. Keifer T. Saarinen (1992) J. Am. Chem. Soc. 114 10663–10665 Occurrence Handle10.1021/ja00052a088
L.E. Kay G.Y. Xu A.U. Singer D.R. Muhandiram J.D. Forman-Kay (1993) J. Magn. Reson. B 101 333–337
R.A. Laskowski M.W. MacArthur D.S. Moss J.M. Thorton (1993) J. Appl. Crystallogr. 26 283–290 Occurrence Handle10.1107/S0021889892009944
P. Lavigne J.R. Bagu R. Boyko L. Willard C.F. Holmes B.D. Sykes (2000) Protein Sci. 9 252–264
T.M. Logan E.T. Olejniczak R.X. Xu S.W. Fesik (1993) J. Biomol. NMR 3 225–231 Occurrence Handle10.1007/BF00178264
B.A. Lyons G.T. Montelione (1993) J. Magn. Reson. B 101 206–209
S. McKenna J. Hu T. Moraes W. Xiao M.J. Ellison L. Spyracopoulos (2003a) Biochemistry 42 7922–7930 Occurrence Handle10.1021/bi034480t
S. McKenna T. Moraes L. Pastushok C. Ptak W. Xiao L. Spyracopoulos M.J. Ellison (2003b) J. Biol. Chem. 278 13151–13158 Occurrence Handle10.1074/jbc.M212353200
S. McKenna L. Spyracopoulos T. Moraes L. Pastushok C. Ptak W. Xiao M.J. Ellison (2001) J. Biol. Chem. 276 40120–40126 Occurrence Handle10.1074/jbc.M102858200
T. Miura W. Klaus B. Gsell C. Miyamoto H. Senn (1999) J. Mol. Biol. 290 213–228 Occurrence Handle10.1006/jmbi.1999.2859
T.F. Moraes R.A. Edwards S. McKenna L. Pastushok W. Xiao J.N. Glover M.J. Ellison (2001) Nat. Struct. Biol. 8 669–673 Occurrence Handle10.1038/90373
D.R. Muhandiram L.E. Kay (1994) J. Magn. Reson. Ser. B 103 203–216 Occurrence Handle10.1006/jmrb.1994.1032
D. Neri T. Szyperski G. Otting H. Senn K. Wuthrich (1989) Biochemistry 28 7510–7516 Occurrence Handle10.1021/bi00445a003
O. Pornillos S.L. Alam R.L. Rich D.G. Myszka D.R. Davis W.I. Sundquist (2002) Embo. J. 21 2397–2406 Occurrence Handle10.1093/emboj/21.10.2397
G. Prag S. Misra E.A. Jones R. Ghirlando B.A. Davies B.F. Horazdovsky J.H. Hurley (2003) Cell 113 609–620 Occurrence Handle10.1016/S0092-8674(03)00364-7
S. Sambrook E.F. Fritch T. Maniatis (1989) Molecular Cloning: A Laboratory Manual Cold Spring Harbor Laboratory Press New York, NY
J.D. Schnell L. Hicke (2003) J. Biol. Chem. 278 35857–35860 Occurrence Handle10.1074/jbc.R300018200
C.M. Slupsky C.M. Kay F.C. Reinach L.B. Smillie B.D. Sykes (1995) Biochemistry 34 7365–7375
W.I. Sundquist H.L. Schubert B.N. Kelly G.C. Hill J.M. Holton C.P. Hill (2004) Mol. Cell 13 783–789 Occurrence Handle10.1016/S1097-2765(04)00129-7
K.A. Swanson R.S. Kang S.D. Stamenova L. Hicke I. Radhakrishnan (2003) Embo. J. 22 4597–4606 Occurrence Handle10.1093/emboj/cdg471
C. Tsui A. Raguraj C.M. Pickart (2005) J. Biol. Chem. 280 19829–19835 Occurrence Handle10.1074/jbc.M414060200
A.P. VanDemark R.M. Hofmann C. Tsui C.M. Pickart C. Wolberger (2001) Cell 105 711–720 Occurrence Handle10.1016/S0092-8674(01)00387-7
G.W. Vuister A. Bax (1992) J. Magn. Reson. 98 428–435
C. Wang L. Deng M. Hong G.R. Akkaraju J. Inoue Z.J. Chen (2001) Nature 412 346–351 Occurrence Handle2001Natur.412..346W
M. Wittekind L. Mueller (1993) J. Magn. Reson. B 101 201–205
W. Xiao S.L. Lin S. Broomfield B.L. Chow Y.F. Wei (1998) Nucleic Acid Res. 26 3908–3914
T. Yamazaki W. Lee C.H. Arrowsmith D.R. Muhandiram L.E. Kay (1994) J. Am. Chem. Soc. 116 11655–11666
D.W. Yang Y. Zheng D.J. Liu D.F. Wyss (2004) J. Am. Chem. Soc. 126 3710–3711
C. Zwahlen P. Legault S.J.F. Vincent J. Greenblatt R. Konrat L.E. Kay (1997) J. Am. Chem. Soc. 119 6711–6721 Occurrence Handle10.1021/ja970224q
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lewis, M.J., Saltibus, L.F., Hau, D.D. et al. Structural Basis for Non-Covalent Interaction Between Ubiquitin and the Ubiquitin Conjugating Enzyme Variant Human MMS2. J Biomol NMR 34, 89–100 (2006). https://doi.org/10.1007/s10858-005-5583-6
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s10858-005-5583-6