Abstract
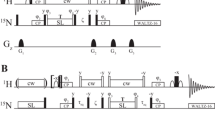
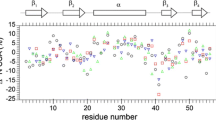
Off-resonance effects can introduce significant systematic errors in R2 measurements in constant-time Carr-Purcell-Meiboom-Gill (CPMG) transverse relaxation dispersion experiments. For an off-resonance chemical shift of 500 Hz, 15N relaxation dispersion profiles obtained from experiment and computer simulation indicated a systematic error of ca. 3%. This error is three- to five-fold larger than the random error in R2 caused by noise. Good estimates of total R2 uncertainty are critical in order to obtain accurate estimates in optimized chemical exchange parameters and their uncertainties derived from χ2 minimization of a target function. Here, we present a simple empirical approach that provides a good estimate of the total error (systematic + random) in 15N R2 values measured for the HIV protease. The advantage of this empirical error estimate is that it is applicable even when some of the factors that contribute to the off-resonance error are not known. These errors are incorporated into a χ2 minimization protocol, in which the Carver–Richards equation is used fit the observed R2 dispersion profiles, that yields optimized chemical exchange parameters and their confidence limits. Optimized parameters are also derived, using the same protein sample and data-fitting protocol, from 1H R2 measurements in which systematic errors are negligible. Although 1H and 15N relaxation profiles of individual residues were well fit, the optimized exchange parameters had large uncertainties (confidence limits). In contrast, when a single pair of exchange parameters (the exchange lifetime, τex, and the fractional population, pa), were constrained to globally fit all R2 profiles for residues in the dimer interface of the protein, confidence limits were less than 8% for all optimized exchange parameters. In addition, F-tests showed that quality of the fits obtained using τex, pa as global parameters were not improved when these parameters were free to fit the R2 profiles of individual residues. Finally, nearly the same optimized global τex, pa values were obtained, when the 1H and 15N data sets for residues in the dimer interface, were fit independently; the difference in optimized global parameters, ca. 10%, was of marginal significance according to the F-test.
Similar content being viewed by others
References
J.P. Carver R.E. Richards (1972) J. Magn. Reson. 6 89–105
M. Czisch G.C. King A. Ross (1997) J. Magn. Reson. 126 154–157 Occurrence Handle10.1006/jmre.1997.1168 Occurrence Handle9252288
D.G. Davis M.E. Perlman R.E. London (1994) J. Magn. Reson. B 104 266–275 Occurrence Handle10.1006/jmrb.1994.1084 Occurrence Handle8069484
F. Delaglio S. Grzesiek G.W. Vuister G. Zhu J. Pfeifer A. Bax (1995) J. Biomol. NMR 6 277–293 Occurrence Handle10.1007/BF00197809 Occurrence Handle8520220
R.R. Ernst G. Bodenhausen A. Wokaun (1987) Principles of Nuclear Magnetic Resonance in One and Two Dimensions Clarendon Press Oxford
D.S. Garrett R. Powers A.M. Gronenborn G.M. Clore (1991) J. Magn. Reson. 95 214–220
H. Geen R. Freeman (1991) J. Mag. Reson. 93 93–141
M.P.B. Guenneugues H. Desvaux (1999) J. Magn. Reson. 136 118–126 Occurrence Handle10.1006/jmre.1998.1590 Occurrence Handle9887297
R. Ishima P.T. Wingfield S.J. Stahl J.D. Kaufman D.A. Torchia (1998) J. Am. Chem. Soc. 120 10534–10542 Occurrence Handle10.1021/ja981546c
R. Ishima D.I. Freedberg Y.X. Wang J.M. Louis D.A. Torchia (1999) Structure 7 1047–1055 Occurrence Handle10.1016/S0969-2126(99)80172-5 Occurrence Handle10508781
R. Ishima D.A. Torchia (2003) J. Biomol. NMR 25 243–348 Occurrence Handle10.1023/A:1022851228405 Occurrence Handle12652136
R. Ishima J. Baber J.M. Louis D.A. Torchia (2004) J. Biomol. NMR 29 187–198 Occurrence Handle10.1023/B:JNMR.0000019249.50306.5d Occurrence Handle15014232
J.A. Jones P. Hodgkinson A.L. Barker P.L. Hore (1996) J. Magn. Reson. Ser. B. 113 25–34 Occurrence Handle10.1006/jmrb.1996.0151
J.A. Jones (1997) J. Magn. Reson 126 283–286 Occurrence Handle10.1006/jmre.1997.1167
E. Katoh J.M. Louis T. Yamazaki A.M. Gronenborn D.A. Torchia R. Ishima (2003) Protein Sci. 12 1376–1385 Occurrence Handle10.1110/ps.0300703 Occurrence Handle12824484
D. Korzhnev E.V. Tischenko A.S. Arseniev (2000) J. Biomol. NMR 17 231–237 Occurrence Handle10.1023/A:1008348827208 Occurrence Handle10959630
D.M. Korzhnev X. Salvatella M. Vendruscolo A.A. Di Nardo A.R. Davidson C.M. Dobson L.E. Kay (2004) Nature 430 586–590 Occurrence Handle10.1038/nature02655 Occurrence Handle15282609
J.P. Loria M. Rance A.G. Palmer SuffixIII (1999) J. Am. Chem. Soc. 121 2331–2332 Occurrence Handle10.1021/ja983961a
Louis, J.M., Ishima, R., Nesheiwat, I., Pannell, L.K., Lynch, S.M., Torchia, D.A. and Gronenborn, A.M. (2002) J. Biol. Chem.
F.A.A. Mulder N.R. Skrynnikov B. Hon F.W. Dahlquist L.E. Kay (2001) J. Am. Chem. Soc. 123 967–975 Occurrence Handle10.1021/ja003447g Occurrence Handle11456632
F.A.A. Mulder B. Hon A. Mittermaier F.W. Dahlquist L.E. Kay (2002) J. Am. Chem. Soc. 124 1443–1451 Occurrence Handle10.1021/ja0119806 Occurrence Handle11841314
Nicholson, L.K., Kay, L.E. and Torchia, D.A. (1996). In NMR Spectrocsopy and its Application to Biomedical Research. Sarkar, S.K. (Eds.), Elsevier, Amsterdam, pp. 241–279.
V.Y. Orekhov K.V. Pervushin A.S. Arseniev (1994) Eur. J. Biochem. 219 887–896 Occurrence Handle10.1111/j.1432-1033.1994.tb18570.x Occurrence Handle8112340
A.G. Palmer SuffixIII C.D. Kroenke J.P. Loria (2001) Methods in Enzymology 339 204–238 Occurrence Handle11462813
W.H. Press B.P. Flannery S.A. Teukolsky W.T. Vetterling (1988) Numerical Recipes in C Cambridge University Press Cambridge U.K
A. Ross M. Czisch G.C. King (1997) J. Magn. Reson 124 355–365 Occurrence Handle10.1006/jmre.1996.1036
N.R. Skrynnikov F.A.A. Mulder B. Hon F.W. Dahlquist L.E. Kay (2001) J. Am. Chem. Soc. 123 4556–4566 Occurrence Handle10.1021/ja004179p Occurrence Handle11457242
M. Tollinger N.R. Skrynnikov F.A.A. Mulder J.D. Forman–Kay L.E. Kay (2001) J. Am. Chem. Soc. 123 11341–11352 Occurrence Handle10.1021/ja011300z Occurrence Handle11707108
O. Trott A.G. Palmer SuffixIII (2004) J. Magn. Reson. 170 104–112 Occurrence Handle10.1016/j.jmr.2004.06.005 Occurrence Handle15324763
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ishima, R., Torchia, D.A. Error estimation and global fitting in transverse-relaxation dispersion experiments to determine chemical-exchange parameters. J Biomol NMR 32, 41–54 (2005). https://doi.org/10.1007/s10858-005-3593-z
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s10858-005-3593-z