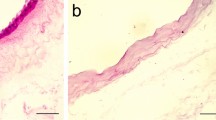
Abstract
Due to its biological properties, human amniotic membrane (hAM) is widely studied in the field of tissue engineering and regenerative medicine. hAM is already very attractive for wound healing and it may be helpful as a support for bone regeneration. However, few studies assessed its potential for guided bone regeneration (GBR). The purpose of the present study was to assess the potential of the hAM as a membrane for GBR. In vitro, cell viability in fresh and cryopreserved hAM was assessed. In vivo, we evaluated the impact of fresh versus cryopreserved hAM, using both the epithelial or the mesenchymal layer facing the defect, on bone regeneration in a critical calvarial bone defect in mice. Then, the efficacy of cryopreserved hAM associated with a bone substitute was compared to a collagen membrane currently used for GBR. In vitro, no statistical difference was observed between the conditions concerning cell viability. Without graft material, cryopreserved hAM induced more bone formation when the mesenchymal layer covered the defect compared to the defect left empty. When associated with a bone substitute, such improved bone repair was not observed. These preliminary results suggest that cryopreserved hAM has a limited potential for GBR.




Similar content being viewed by others
Abbreviations
- BMP :
-
Bone morphogenetic protein
- GBR :
-
Guided bone regeneration
- HA :
-
Hydroxy apatite
- hAM :
-
human amniotic membrane
- hAECs :
-
human amniotic epithelial cells
- hAMSCs :
-
human amniotic mesenchymal stem cells
- f-hAM :
-
fresh hAM
- Cryo-hAM :
-
Cryopreserved hAM
References
Larsson L, Decker AM, Nibali L, Pilipchuk SP, Berglundh T, Giannobile WV. Regenerative Medicine for Periodontal and Peri-implant Diseases. J Dent Res. 2016;95:255–66. https://doi.org/10.1177/0022034515618887.
Milinkovic I, Cordaro L. Are there specific indications for the different alveolar bone augmentation procedures for implant placement? A systematic review. Int J Oral Maxillofac Surg. 2014;43:606–25. https://doi.org/10.1016/j.ijom.2013.12.004.
Sculean A, Chappuis V, Cosgarea R. Coverage of mucosal recessions at dental implants. Periodontol 2000. 2017;73:134–40. https://doi.org/10.1111/prd.12178.
Rakhmatia YD, Ayukawa Y, Furuhashi A, Koyano K. Current barrier membranes: Titanium mesh and other membranes for guided bone regeneration in dental applications. J Prosthodont Res. 2013;57:3–14. https://doi.org/10.1016/j.jpor.2012.12.001.
Bottino MC, Thomas V, Schmidt G, Vohra YK, Chu T-MG, Kowolik MJ, et al. Recent advances in the development of GTR/GBR membranes for periodontal regeneration—A materials perspective. Dent Mater. 2012;28:703–21. https://doi.org/10.1016/j.dental.2012.04.022.
Francisco JC, Correa Cunha R, Cardoso MA, Baggio Simeoni R, Mogharbel BF, Picharski GL, et al. Decellularized Amniotic Membrane Scaffold as a Pericardial Substitute: An In Vivo Study. Transplant Proc. 2016;48:2845–9. https://doi.org/10.1016/j.transproceed.2016.07.026.
Sanluis-Verdes A, Sanluis-Verdes N, Manso-Revilla MJ, Castro-Castro AM, Pombo-Otero J, Fraga-Mariño M, et al. Tissue engineering for neurodegenerative diseases using human amniotic membrane and umbilical cord. Cell Tissue Bank. 2017;18:1–15. https://doi.org/10.1007/s10561-016-9595-0.
Ilic D, Vicovac L, Nikolic M, Lazic Ilic E. Human amniotic membrane grafts in therapy of chronic non-healing wounds. Br Med Bull. 2016;117:59–67. https://doi.org/10.1093/bmb/ldv053.
Ricci E, Vanosi G, Lindenmair A, Hennerbichler S, Peterbauer-Scherb A, Wolbank S, et al. Anti-fibrotic effects of fresh and cryopreserved human amniotic membrane in a rat liver fibrosis model. Cell Tissue Bank. 2013;14:475–88. https://doi.org/10.1007/s10561-012-9337-x.
Hao Y, Ma DH, Hwang DG, Kim WS, Zhang F. Identification of antiangiogenic and antiinflammatory proteins in human amniotic membrane. Cornea. 2000;19:348–52.
Kubo M, Sonoda Y, Muramatsu R, Usui M. Immunogenicity of human amniotic membrane in experimental xenotransplantation. Invest Ophthalmol Vis Sci. 2001;42:1539–46.
Kang JW, Koo HC, Hwang SY, Kang SK, Ra JC, Lee MH, et al. Immunomodulatory effects of human amniotic membrane-derived mesenchymal stem cells. J Vet Sci. 2012;13:23–31.
Parolini O, Alviano F, Bagnara GP, Bilic G, Bühring H-J, Evangelista M, et al. Concise review: isolation and characterization of cells from human term placenta: outcome of the first international Workshop on Placenta Derived. Stem Cells Stem Cells Dayt Ohio. 2008;26:300–11. https://doi.org/10.1634/stemcells.2007-0594.
Grzywocz Z, Pius-Sadowska E, Klos P, Gryzik M, Wasilewska D, Aleksandrowicz B, et al. Growth factors and their receptors derived from human amniotic cells in vitro. Folia Histochem Cytobiol Pol Acad Sci Pol Histochem Cytochem Soc. 2014;52:163–70. https://doi.org/10.5603/FHC.2014.0019.
Go YY, Kim SE, Cho GJ, Chae S-W, Song J-J. Differential effects of amnion and chorion membrane extracts on osteoblast-like cells due to the different growth factor composition of the extracts. PLoS ONE. 2017;12:e0182716. https://doi.org/10.1371/journal.pone.0182716.
Malhotra C, Jain AK. Human amniotic membrane transplantation: Different modalities of its use in ophthalmology. World J Transplant. 2014;4:111–21. https://doi.org/10.5500/wjt.v4.i2.111.
Allen CL, Clare G, Stewart EA, Branch MJ, McIntosh OD, Dadhwal M, et al. Augmented dried versus cryopreserved amniotic membrane as an ocular surface dressing. PLoS ONE. 2013;8:e78441. https://doi.org/10.1371/journal.pone.0078441.
Laurent R, Nallet A, Obert L, Nicod L, Gindraux F. Storage and qualification of viable intact human amniotic graft and technology transfer to a tissue bank. Cell Tissue Bank. 2014;15:267–75. https://doi.org/10.1007/s10561-014-9437-x.
Soncini M, Vertua E, Gibelli L, Zorzi F, Denegri M, Albertini A, et al. Isolation and characterization of mesenchymal cells from human fetal membranes. J Tissue Eng Regen Med. 2007;1:296–305. https://doi.org/10.1002/term.40.
Lindenmair A, Wolbank S, Stadler G, Meinl A, Peterbauer-Scherb A, Eibl J, et al. Osteogenic differentiation of intact human amniotic membrane. Biomaterials. 2010;31:8659–65. https://doi.org/10.1016/j.biomaterials.2010.07.090.
Barboni B, Mangano C, Valbonetti L, Marruchella G, Berardinelli P, Martelli A, et al. Synthetic bone substitute engineered with amniotic epithelial cells enhances bone regeneration after maxillary sinus augmentation. PLoS ONE. 2013;8:e63256. https://doi.org/10.1371/journal.pone.0063256.
Lawson VG. Oral Cavity Reconstruction Using Pectoralis Major Muscle and Amnion. Arch Otolaryngol - Head Neck Surg. 1985;111:230–3. https://doi.org/10.1001/archotol.1985.00800060054006.
Gomes MF, da Silva dos Anjos MJ, de Oliveira Nogueira T, Guimarães SAC. Histologic Evaluation of the Osteoinductive Property of Autogenous Demineralized Dentin Matrix on Surgical Bone Defects in Rabbit Skulls Using Human Amniotic Membrane for Guided Bone Regeneration. Int J Oral Maxillofac Implants. 2001;16:563.
Tsugawa J, Komaki M, Yoshida T, Nakahama K, Amagasa T, Morita I. Cell-printing and transfer technology applications for bone defects in mice. J Tissue Eng Regen Med. 2011;5:695–703. https://doi.org/10.1002/term.366.
Semyari H, Rajipour M, Sabetkish S, Sabetkish N, Abbas FM, Kajbafzadeh A-M. Evaluating the bone regeneration in calvarial defect using osteoblasts differentiated from adipose-derived mesenchymal stem cells on three different scaffolds: an animal study. Cell Tissue Bank. 2015;17:69–83. https://doi.org/10.1007/s10561-015-9518-5.
Wu P-H, Chung H-Y, Wang J-H, Shih J-C, Kuo MY-P, Chang P-C, et al. Amniotic membrane and adipose-derived stem cell co-culture system enhances bone regeneration in a rat periodontal defect model. J Formos Med Assoc Taiwan Yi Zhi. 2015;115:186–94. https://doi.org/10.1016/j.jfma.2015.02.002.
Li W, Ma G, Brazile B, Li N, Dai W, Butler JR, et al. Investigating the Potential of Amnion-Based Scaffolds as a Barrier Membrane for Guided Bone Regeneration. Langmuir ACS J Surf Colloids. 2015;31:8642–53. https://doi.org/10.1021/acs.langmuir.5b02362.
Amemiya T, Nishigaki M, Yamamoto T, Kanamura N. Experiences of preclinical use of periodontal ligament-derived cell sheet cultured on human amniotic membrane. J Oral Tissue Eng. 2008;6:106–12.
Iwasaki K, Komaki M, Yokoyama N, Tanaka Y, Taki A, Honda I, et al. Periodontal regeneration using periodontal ligament stem cell-transferred amnion. Tissue Eng Part A. 2014;20:693–704. https://doi.org/10.1089/ten.TEA.2013.0017.
Kumar A, Chandra RV, Reddy AA, Reddy BH, Reddy C, Naveen A. Evaluation of clinical, antiinflammatory and antiinfective properties of amniotic membrane used for guided tissue regeneration: A randomized controlled trial. Dent Res J. 2015;12:127–35.
Kiany F, Moloudi F. Amnion membrane as a novel barrier in the treatment of intrabony defects: a controlled clinical trial. Int J Oral Maxillofac Implants. 2015;30:639–47.
Kothiwale SV, Anuroopa P, Gajiwala AL. A clinical and radiological evaluation of DFDBA with amniotic membrane versus bovine derived xenograft with amniotic membrane in human periodontal grade II furcation defects. Cell Tissue Bank. 2009;10:317–26. https://doi.org/10.1007/s10561-009-9126-3.
Afshar A, Ghorbani M, Ehsani N, Saeri MR, Sorrell CC. Some important factors in the wet precipitation process of hydroxyapatite. Mater Des. 2003;24:197–202. https://doi.org/10.1016/S0261-3069(03)00003-7.
Catros S, Guillemot F, Lebraud E, Chanseau C, Perez S, Bareille R, et al. Physico-chemical and biological properties of a nano-hydroxyapatite powder synthesized at room temperature. IRBM. 2010;31:226–33. https://doi.org/10.1016/j.irbm.2010.04.002.
Guduric V, Metz C, Siadous R, Bareille R, Levato R, Engel E, et al. Layer-by-layer bioassembly of cellularized polylactic acid porous membranes for bone tissue engineering. J Mater Sci Mater Med. 2017;28:78. https://doi.org/10.1007/s10856-017-5887-6.
Arai N, Tsuno H, Okabe M, Yoshida T, Koike C, Noguchi M, et al. Clinical application of a hyperdry amniotic membrane on surgical defects of the oral mucosa. J Oral Maxillofac Surg J Am Assoc Oral Maxillofac Surg. 2012;70:2221–8. https://doi.org/10.1016/j.joms.2011.09.033.
Gurinsky B. A novel dehydrated amnion allograft for use in the treatment of gingival recession: an observational case series. J Implant & Adv Clin Dent. 2009;1:124–30.
Maral T, Borman H, Arslan H, Demirhan B, Akinbingol G, Haberal M. Effectiveness of human amnion preserved long-term in glycerol as a temporary biological dressing. Burns. 1999;25:625–35. https://doi.org/10.1016/S0305-4179(99)00072-8.
Hennerbichler S, Reichl B, Pleiner D, Gabriel C, Eibl J, Redl H. The influence of various storage conditions on cell viability in amniotic membrane. Cell Tissue Bank Int J Bank Eng Transplant Cells Tissues 2007:1.
Kesting MR, Loeffelbein DJ, Steinstraesser L, Muecke T, Demtroeder C, Sommerer F, et al. Cryopreserved human amniotic membrane for soft tissue repair in rats. Ann Plast Surg. 2008;60:684–91. https://doi.org/10.1097/SAP.0b013e31814fb9d2.
Si J, Dai J, Zhang J, Liu S, Gu J, Shi J, et al. Comparative investigation of human amniotic epithelial cells and mesenchymal stem cells for application in bone tissue engineering. Stem Cells Int. 2015;2015:565732. https://doi.org/10.1155/2015/565732.
Laurent R, Nallet A, de Billy B, Obert L, Nicod L, Meyer C, et al. Fresh and in vitro osteodifferentiated human amniotic membrane, alone or associated with an additional scaffold, does not induce ectopic bone formation in Balb/c mice. Cell Tissue Bank. 2017;18:17–25. https://doi.org/10.1007/s10561-016-9605-2.
Gindraux F, Rondot T, de Billy B, Zwetyenga N, Fricain J-C, Pagnon A, et al. Similarities between induced membrane and amniotic membrane: Novelty for bone repair. Placenta. 2017. https://doi.org/10.1016/j.placenta.2017.06.340.
Bunyaratavej P, Wang HL. Collagen membranes: a review. J Periodontol. 2001;72:215–29. https://doi.org/10.1902/jop.2001.72.2.215.
Zhou H, Lee J. Nanoscale hydroxyapatite particles for bone tissue engineering. Acta Biomater. 2011;7:2769–81. https://doi.org/10.1016/j.actbio.2011.03.019.
McKay WF, Peckham SM, Badura JM. A comprehensive clinical review of recombinant human bone morphogenetic protein-2 (INFUSE Bone Graft). Int Orthop. 2007;31:729–34. https://doi.org/10.1007/s00264-007-0418-6.
Lee C-H, Jin MU, Jung H-M, Lee J-T, Kwon T-G. Effect of dual treatment with SDF-1 and BMP-2 on ectopic and orthotopic bone formation. PLoS ONE. 2015;10:e0120051. https://doi.org/10.1371/journal.pone.0120051.
Ben-David D, Srouji S, Shapira-Schweitzer K, Kossover O, Ivanir E, Kuhn G, et al. Low dose BMP-2 treatment for bone repair using a PEGylated fibrinogen hydrogel matrix. Biomaterials. 2013;34:2902–10. https://doi.org/10.1016/j.biomaterials.2013.01.035.
Notodihardjo FZ, Kakudo N, Kushida S, Suzuki K, Kusumoto K. Bone regeneration with BMP-2 and hydroxyapatite in critical-size calvarial defects in rats. J Cranio-Maxillo-Fac Surg Publ Eur Assoc Cranio-Maxillo-Fac Surg. 2012;40:287–91. https://doi.org/10.1016/j.jcms.2011.04.008.
Muschler GF, Raut VP, Patterson TE, Wenke JC, Hollinger JO. The design and use of animal models for translational research in bone tissue engineering and regenerative medicine. Tissue Eng Part B Rev. 2010;16:123–45. https://doi.org/10.1089/ten.TEB.2009.0658.
Gomes PS, Fernandes MH. Rodent models in bone-related research: the relevance of calvarial defects in the assessment of bone regeneration strategies. Lab Anim. 2011;45:14–24. https://doi.org/10.1258/la.2010.010085.
Smith DM, Cray JJ, Weiss LE, Dai Fei EK, Shakir S, Rottgers SA, et al. Precise control of osteogenesis for craniofacial defect repair: the role of direct osteoprogenitor contact in BMP-2-based bioprinting. Ann Plast Surg. 2012;69:485–8. https://doi.org/10.1097/SAP.0b013e31824cfe64.
Pellegrini G, Pagni G, Rasperini G Surgical Approaches Based on Biological Objectives: GTR versus GBR Techniques. Int J Dent 2013;2013:1–13. https://doi.org/10.1155/2013/521547.
Acknowledgements
La Fondation des gueules cassées for financial support. Ray Cooke (professional proof-reader) for copyediting the manuscript. Patrick Guitton for his contribution to design the figures.
Author contributions
MF (1st author): Contributed to conception and design of the experimental studies (both in vitro and in vivo studies); Contributed to data acquisition, analysis, and data interpretation; Drafted the manuscript; Critically revised the manuscript; Gave final approval; Agrees to be accountable for all aspects of work ensuring integrity and accuracy. OC(2nd author): X-ray analyzes; Gave final approval; Agrees to be accountable for all aspects of work ensuring integrity and accuracy. JK(3rd author): Obtained hydroxyapatite particles; Critically revised the manuscript; Gave final approval; Agrees to be accountable for all aspects of work ensuring integrity and accuracy. FG (4th author): Contributed to conception and design of the experimental in vivo studies; Critically revised the manuscript; Gave final approval; Agrees to be accountable for all aspects of work ensuring integrity and accuracy. SB (5th author): contributed to collection of human placentas; Critically revised the manuscript; Gave final appoval; Agrees to be accountable for all aspects of work ensuring integrity and accuracy RB (6th author): contributed to conception and design of in vitro studies; Critically revised the manuscript; Gave final appoval; Agrees to be accountable for all aspects of work ensuring integrity and accuracyZI (57th author): Contributed to collection of human placentas; Critically revised the manuscript; Gave final approval, Agrees to be accountable for all aspects of work ensuring integrity and accuracy. JCF (last co-author): Contributed to conception and design of experimental studies (both in vitro and in vivo studies); Contributed to analysis, and interpretation of the data; Drafted the manuscript; Critically revised the manuscript; Gave final approvalAgrees to be accountable for all aspects of work ensuring integrity and accuracy. CB (Last co- author); Contributed to conception and design of experimental studies (both in vitro and in vivo studies); Contributed to data acquisition, analysis, and data interpretation; Drafted the manuscript; Gave final approval; Agrees to be accountable for all aspects of work ensuring integrity and accuracy.
Funding
Fondation des gueules cassées.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Ethics approval
The present study was approved by the French Ethics Committee (agreement APAFIS n 2685-20l5111012075358 v4).
Additional information
Jean-Christophe Fricain and Claudine Boiziau are co-directed.
Rights and permissions
About this article
Cite this article
Fénelon, M., Chassande, O., Kalisky, J. et al. Human amniotic membrane for guided bone regeneration of calvarial defects in mice. J Mater Sci: Mater Med 29, 78 (2018). https://doi.org/10.1007/s10856-018-6086-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10856-018-6086-9